Research

The chemical synthesis of functional organic molecules underpins many key advances in human medicine, crop protection, biotechnology, and material science. Hence, the development of efficient, cost-effective routes to carbon-based molecules is an important, contemporary scientific challenge. Work in the Shipman Group is focused on the development of new methods for the construction of organic compounds alongside a variety of application-driven projects. We are especially interested in new uses of strained (3- and 4- membered) heterocycles. Projects include:
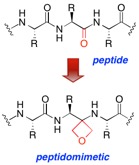
Oxetane Based Peptidomimetics
Peptides and their larger counterparts, proteins, are assembled by linking together the proteinogenic amino acids through identical, repeating amide (peptide) bonds. These oligomeric structures produce incredible levels of structural diversity that ultimately enables great variety in biological function. Whilst peptide bonds are chemically very stable at neutral pH, they are often labile in vivo due to the action of peptidases and proteases. Chemical modification of a natural peptide can increase its bioavailability, enzymatic stability, prolong half-lives and increase activity and/or selectivity for the target receptor. Peptide bond isosteres are an especially important type of peptidomimetic that can aid in the interrogation of the structure or function of a peptide or protein by altering the chemical properties of one or more amino acid residues. The resulting perturbation enables structure-function relationships within native peptides and proteins to be revealed through access to protein structural space not available to natural α-amino acids. We are studying a new type of peptide bond isostere, in which the heterocyclic oxetane nucleus is used as a replacement for the carbonyl group of the peptide bond.1 Methods for the synthesis of other oxetane containing heterocycles are also being explored.2
For a poster detailing some of our findings, click here.![]()
This project is supported by the Leverhulme Trust.
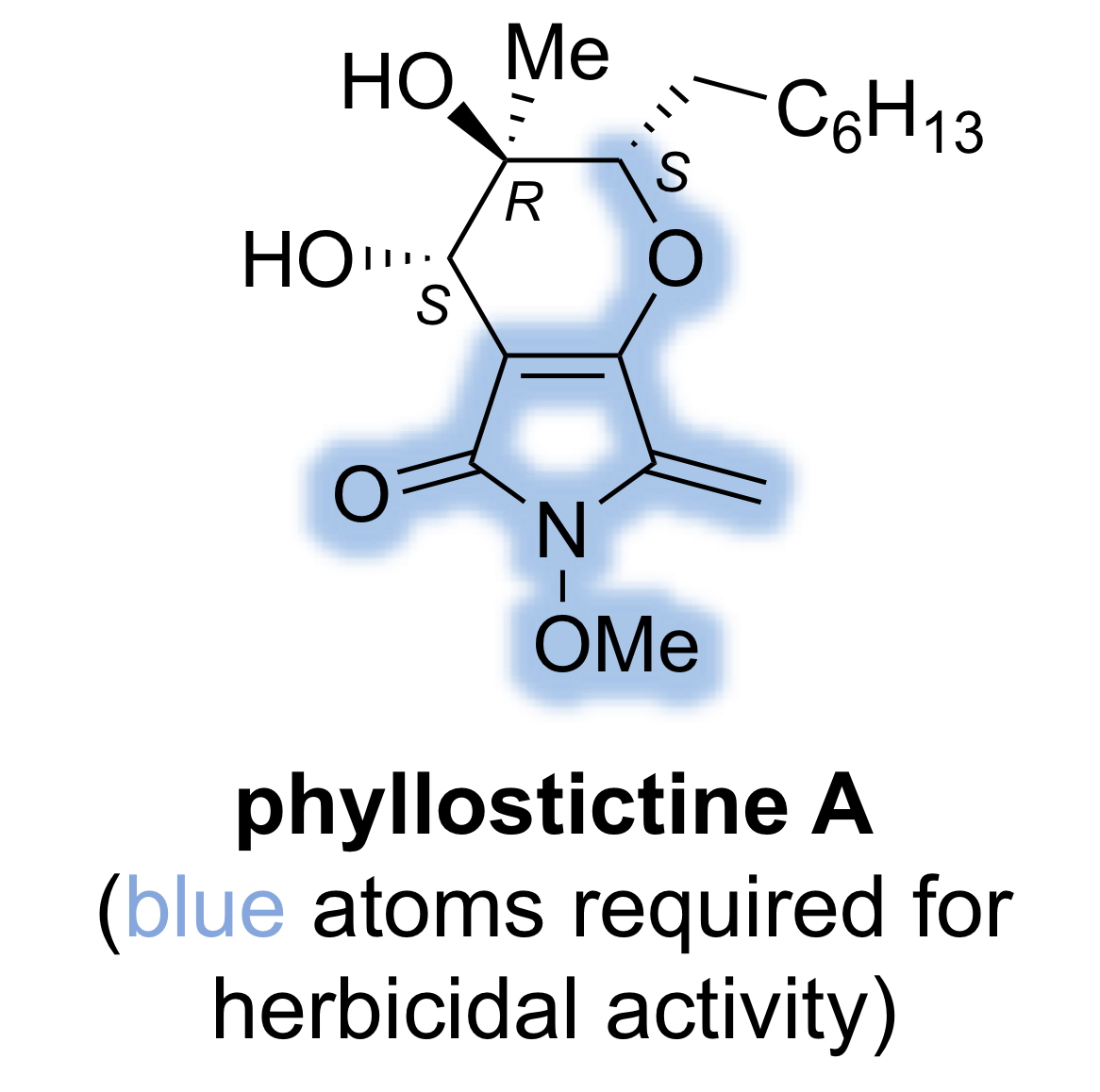
Crop Protection Agents Based on Natural Products
Natural products produced by the pathways of secondary metabolism in a range of organisms possess much greater structural diversity than compounds produced by man. They are often endowed with potent activity for specific biological targets together with useful physicochemical properties. Consequently, natural products provide an excellent starting point for the discovery of new classes of human medicines and agrochemicals that operate by novel mechanisms of action. In 2008, Evidente et al reported the isolation and structural elucidation of a highly novel class of natural herbicide produced by the fungus Phyllosticta ciirsi.3 Four compounds were isolated called phyllostictines A-D. The structure of these natural products was revised in 2017 by Cox and Trenti.4 This project, in collaboration with colleagues in the School of Life Sciences, is focused on uncovering the molecular basis of the herbicidal activity. We have recently acheived the first total synthesis of phyllostictine A (PA), the most potent member of this new family of herbicides, and identified the heterocyclic subunit responsible for the weed-killing activity.5
This project has received funding from the European Union’s Horizon 2020 research and innovation programme under the Marie Sklodowska-Curie grant agreement No 705079, EPSRC (EP/K031783/1) and BBSRC.



Useful New Bond Forming Reactions of Small Ring Heterocycles
Several projects are being pursued which rely on relief of ring strain to promote useful new bond-forming processes. These include:
- New routes to vicinal diamines, important building blocks in medicinal chemistry and catalysis, based upon catalytic asymmetric additions to 3-methylene-1,2-diazetidines;6
- Multi-component reactions and cycloadditions of highly strained methyleneaziridines;7
- Telescoped reactions of aziridines and related heterocycles in flow.
References:
- N. H. Powell, G. J. Clarkson, R. Notman, P. Raubo, N. G. Martin, M. Shipman, Chem. Commun. 2014, 50, 8797. [Abstract]
- J. V. Geden, B. O. Beasley, G. J. Clarkson, M. Shipman, J. Org. Chem., 2013, 78, 12243. [Open Access] (b) B. O. Beasley, G. J. Clarkson, M. Shipman, Tetrahedron Lett., 2012, 53, 2951. [Abstract]
- A. Evidente, A. Cimmino, A. Andolfi, M.Vurro, M. C. Zonno, C. L. Cantrell, A. Motta, Tetrahedron 2008, 64, 1612.
- F. Trenti and R. J. Cox, J. Nat. Prod., 2017, 80, 1235.
- M. Riemer, V. V. Uzunova, N. Riemer, G. J. Clarkson, N. Pereira, R. Napier and M. Shipman, Chem. Commun. 2018, 10.1039/C8CC03349H
-
(a) G. P. Iacobini, D. W. Porter, M. Shipman, Chem. Commun. 2012, 9852. [Abstract] (b) M. J. Brown, G. J. Clarkson, G. G. Inglis, M. Shipman, Org. Lett., 2011, 13, 1686. [Abstract]
- (a) K. Griffin, C. Montagne, C. T. Hoang, G. J. Clarkson, M. Shipman,
Org. Biomol. Chem., 2012, 10, 1032. [Abstract] (b) C. C. A. Cariou, G. J. Clarkson, M. Shipman, J. Org. Chem. 2008, 73, 9762.[Abstract]
