The Current Status of Platorchestia Platensis in the United Kingdom
Rebekah Simpson[1], Marine Biology and Ecology Research Centre, School of Marine Science and Engineering, University of Plymouth
Abstract
Platorchestia platensis are a cryptogenic amphipod found on shores worldwide. Outside of the UK, P. platensis outcompetes the native species, reducing biodiversity resulting in strandline not being decomposed. Originally recorded in the United Kingdom (UK) in 1979, P. platensis had not been found since. Within this study timed searches were undertaken to document the distribution and abundance of amphipod species around Kent, in order to establish whether P. platensis was present in the UK. The natural history associated with the amphipod communities at each site was also documented. In 2010, this study rediscovered P. platensis after 31 years at three sites on the north Kent coast, where it was reproducing successfully and found to coexist with the native species. The P. platensis found were most morphologically similar to a Danish P. platensis population, suggesting possible transport by ballast water. The UK P. platensis may since have adapted in geographical isolation to become a new sub-species; however, genetic tests are needed to confirm this. More research is also needed to assess whether P. platensis continues to coexist with the native species or whether in time P. platensis will outcompete the native species, as it has done elsewhere.
Keywords: Amphipod, Gammarid, Talitrid, Invasive, Orchestia gammarellus, Kent
Introduction
Alien or invasive species are defined as 'non-indigenous species whose introduction causes or is likely to cause economic or environmental harm, reducing biodiversity' (ISAC, 2006: 1). Worldwide, marine invasions are considered the leading cause of marine extinctions and biotic homogenisation (Sala et al., 2000: 1770-74; Lockwood and McKinney, 2001: 1-259; Drake and Lodge, 2007: 121-31). One invasive species is a semi-terrestrial talitrid from the order Amphipoda (Crustacea), called Platorchestia platensis (Bousfield, 1982: 26-30).
Despite this invasive species having worldwide distribution very little is known about its invasive history (Iwasa, 1939: 255-69; Backlund, 1945: 1-236; Dahl, 1946:1-53; Persson, 2001: 201-10). Originally found in Uruguay (Serejo and Lowry, 2008:189-94), it is unclear whether P. platensis originated from here as the site is close to the international port of Buenos Aires, which played an essential role as a connecting port between the New World and Europe. As with many amphipods, dispersal capacity is naturally low (Friend and Richardson, 1986: 25-48) so amphipods tend to disperse transoceanic in ballast water (Lacey, 2009) or more locally on floating strandline (Stock and Biernbaum, 1994: 795-811). In the UK P. platensis was originally recorded in the Thames Estuary by Wildish and Lincoln (1979), P. platensis has not been found in the UK since, despite intermediate studies.
P. platensis are a cryptogenic, cosmopolitan species, distributed in temperate and tropical regions around the world (Behbehani and Croker, 1982: 611-20), including North Western Europe (Dahl, 1946:1-53), Sweden (Backlund, 1945: 1-236; Dahl, 1946:1-53), Poland (Spicer and Janas, 2006: 321-29), the Netherlands (Karlbrink, 1969:327-34), Tenerife (Andersson, 1962: 211-18), the Canary Islands (Andersson, 1962: 211-18), Uruguay (Serejo and Lowry, 2008:189-194), Israel (Morino and Ortal, 1995, 68: 824-32), India (Chilton, 1921:519-58), Korea (Jo, 1988: 153-179), Japan (Ruffo, 1949: 58; Morino, 1978: 824-32), Hawaii and other Pacific Islands (Stephensen, 1935: 1-20), the Atlantic coast of Canada and the USA, Bermuda (Kunkel, 1910: 1-115), the West Indies (Bousfield, 1973: 883-98) and South America (Stebbing, 1906: 806).
Amphipods are of great ecological importance in the marine environment as they function as key decomposers in a number of different communities, as well as acting as a food source for a wide range of animals including fish, birds, invertebrate and mammals (Morritt and Spicer, 1998: 1965-82; Reid, 1947: 1-25). When the natural amphipod community is altered, such as by the presence of an invasive competitor, excess strandline results. This is because P. platensis cannot break down the wrack as efficiently alone as a diverse multitude of amphipod species.
The abundance of P. platensis shows high spatial and temporal variation, but has been found to dominate over other amphipod species all year round in all habitats (Behbehani and Croker, 1982: 611-20). This could be because P. platensis were found to be omnivorous, eating anything and everything digestible (Morino, 1978: 824-32), have a bivoltine life cycle (two broods of offspring per year) and were nocturnal, avoiding low relative humidity's and daytime predators (Little, 1983). When P. platensis outcompetes other species this results in negative beach aesthetics, with knock-on effects on tourism while altering the balance of nutrients available for use by other organisms in the intertidal ecosystem. The aim of this study was therefore to investigate whether P. platensis are present in the UKby mapping the abundance and distribution of amphipod species present in Kent.
Methodology
Amphipod Distribution
30-minute timed surveys were undertaken at a site every 10km around Kent at low tide, including Wildish and Lincoln's (1979) original site. On pebbly beaches, the strandline was displaced every 3m to elicit jumping behaviours in any amphipods present and on sandy beaches a 0.5m2hole was dug every 3m along the spring strandline to record burrowing amphipods. Amphipods were caught by hand and put in sampling pots containing 70% industrial methylated spirit (IMS) for later identification and counting. 0.2ml Rose of Bengal was added to each sampling pot and left to settle for 10 minutes, as the IMS had bleached the amphipods translucent making the appendages difficult to use for identification. Identification was carried out under low power magnification (15x) using Spicer and Janas's (2006) classification system.
Natural History
The fauna and flora found in the strandline, beach profile (dissipative vs reflective), neap vs spring tide, the weather, possible anthropogenic impacts on the site, air temperature and substrate type were recorded.
Morphological comparison
Selected life history characteristics of P. platensis were explored by examining the morphology of any P. platensis found. The characteristics measured included the sex, determined by the presence of oostegite in females and developed gnathopod 2 in males, and total body length measured as the dorsal length from the tip of the rostrum to the tip of the telson (Stock and Biernbaum 1994: 795-811, Scapini et al. 1999: 659-62). The numbers of segments on antennae 2 in female P. platensis were counted to assess whether the females had reproduced prior to sampling (Morino, 1978, 68: 824-32), and scanning electron microscopy was used on gnathopod 2 in male P. platensis in order to compare the UK species to other P. platensis species worldwide.
Total body length was chosen as it varies between population, age, reproductive cycle and sex; therefore the relative growth of morphological characters can be related to different life strategies (Scapini et al. 1999: 659-62). The relationship between the body length of adult P. platensis and the native species, O. gammarellus, was investigated using a two-sample t-test in Minitab statistical software, as an indication of whether P. platensis are out-competing O. gammarellus. P. platensis could do this by adopting a better life strategy, using different quantities of nutrients for growth and metabolic functions (Backlund, 1945: 1-236).
By comparing the P. platensis from the UK under scanning electron microscope to other published material conclusions can be drawn as to whether a distinct sub-species was present based on its morphological characteristics. The morphology of male gnathopod 2 in P. platensis found in the UK was compared to other species of P. platensis from around the world, as the female gnathopod 2 in UK P. platensis showed no morphological differences. The other species used for comparison were Polish P. platensis (Stock and Biernbaum, 1994: 795-811), Danish P. platensis (Stock and Biernbaum, 1994: 795-811), P. monodi (a tropical variation from the Ascension Islands), Orchestia platensis japonica later renamed P. japonica found in Japan (Iwasa, 1939: 255-69) and China (Hou and Li, 2003: 2442-48) and P. platensis from Israel (Morino and Ortal, 1995: 824-32). In this study P. monodi and P. japonica are treated as two separate species as the only differences occur on antennae 2 and in the number of outer marginal spines along the inner ramus of uropod 1 (four associated with P. monodi as opposed to three associated with P. japonica) (Morino and Ortal, 1995, 68: 824-32).
Prior to scanning electron microscopy the amphipods were air dried for 12 hours, placed on carbon-infiltrated minitabs on clean stubs and labelled using a permanent marker. The males' gnathopods were then dissected from their body using a razor blade. The white backing on the carbon-infiltrated minitabs was then removed and the gnathopod 2 placed on. An Emitech k550 Sputter Gold Coated Unit was used for 10 minutes to coat the appendages in gold dust before using JEOL JSM-560 OLV SEM to take photographs.
Distribution over time
By comparing past data on amphipod community compositions around Kent, a detailed account of the current status of P. platensis in the UK can be provided. As Reid (1947: 1-25) does not state specifically where the Orchestia spp. were found or which species were found in Kent, data were used from Wildish (1969: 288-90, 1982: 3071-74; 1987: 571-83), Wildish and Lincoln (1979:199-200) and Spicer (1996; 1998) to create multi-layered maps using CorelDraw software showing the distribution of amphipod populations including P. platensis over the last 43 years.
Results
Amphipod Distribution
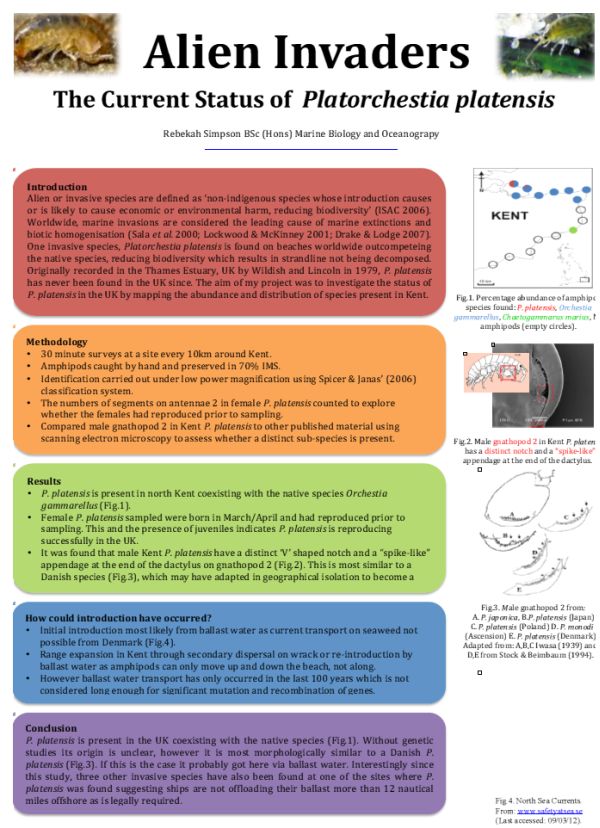
P. platensis were present at three of the sixteen sites studied: Whitstable (one female), Minster (two males and one female) and Leysdown-on-sea (two juveniles) on the north Kent coast (Fig.1). Orchestia gammarellus were also present from Ramsgate round to Sheerness, co-existing with P. platensis at the three sites (Fig.1). Chaetogammarus marinus were found in extreme abundance 100mm below the shingle at Dover, within the ferry terminal (Fig.1). On the south coast of Kent no amphipods were found, despite recurrent visits (Fig.1).
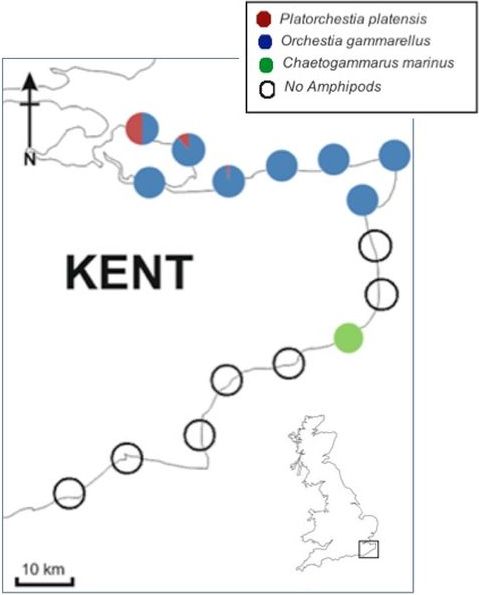
Figure 1: Pie charts show the percentage abundance and distribution of Amphipoda found around Kent.
Natural History
The following animals found in wrack beds alongside P. platensis were identified using Hayward et al. (1996, 1-325). These were Littorina neritoides, Xantho sp., Cerastroderma edule, Crepidula fornicata, Chartella papyraceae, Chlamys sp., Solen marginatus,Nucella sp., Chlorochroa sayi, mussel shell (unidentified=U), oyster shells (U), flies (U), barnacles (U), spiders (U), maggots (U) and beetle (U). Small beetles may be present here as they prey upon Talitridae in the strandline (Bate and Westwood, 1863). The organic matter found within the strandline consisted of Corallina sp., Ascophyllum nodosum, Sargassum muticum, Ulva lactuca, Fucus spiralis, F. vesiculosus, Gelidium latifolium and Enteromorpha intestinalis.
P. platensis were found on pebbly beaches as well as on beaches where the texture of the substrate was 'oaty' due to fine sand and large quantities of broken shell (self defined). These beaches were both dissipative and reflective in the intertidal, displaying both wide and thin surf zones, though all beaches with P. platensis present displayed plunging breakers. P. platensis inhabited both; thick, degraded and thin, patchy continuous strandline (self defined).
Anthropogenic impacts noted which could affect the distribution and dispersal of P. platensis both up and along the shore include beach huts less than 2m from the upper strandline, main roads less than 20m from beach, groynes, continuous sea wall, skate park less than 50m from beach, well-used car parks both above and on the beach at high-water strandline, sailing club, busy caravan park, shellfish farms, seaside resorts less than 100m from beach, jet skiers, horse riders, kite surfers, three wind farms offshore, busy shipping lane offshore, well used for angling and dog walking.
During sampling (June-August) the weather remained overcast and windy with occasional sunshine resulting in an average air temperature of 19ºC. Other points to note include that in heavy rain P. platensis were extremely active and that in the 'oaty' substrate P. platensis were less energetic and jumpy than on pebbly beaches. Instead of exhibiting jumping behaviours the amphipods curled up making them easier to catch, these were also much lighter in colour.
Morphological Comparison
Male P. platensis were found to be 30.9% longer than females; however, it was unclear whether these results are significant due to the limited number of P. platensis found. The average total body length of P. platensis females was 8.3mm and for males was 12.0mm. P. platensis were found to have an average total body length 13.5% larger than O. gammarellus (p= 0.41, 196 d.f.). In O. gammarellus, males were found to be 18.9% bigger on average than females (p< 0.001, 191 d.f.). The mean total body length for O. gammarellus females was 9.5mm and for males was 11.7mm. The population of C. marinus found showed no statistical significance between total body length and sex using a Mann-Whitney U test (p> 0.050, 34 d.f.), implying that this species does not exhibit sexual dimorphism. The sex ratio of male: female for P. platensis was 50:50, O. gammarellus was 75:118 and C. marinus 8:27 respectively.
It was ascertained that the two female P. platensis collected were born in March/April and had reproduced prior to sampling, with one having one brood and the second having two. This suggests that P. platensis was reproducing successfully in the UK, a hypothesis which was further supported by the presence of juvenile P. platensis.
It can be seen that the UK P. platensis had a distinct notch in the centre of the palm of the male gnathopod 2 with a "spike-like" appendage at the end of the dactylus (Fig.2), unlike the lectotype which displays a sinuous palm, without a definite notch (Serejo and Lowry, 2008:189-94).

Figure 2: Male gnathopod 2 in P. platensis from Kent, UK noticing the distinct notch in the palm of the gnathopod 2 and "spike-like" appendage at the end of the dactylus.
This notch can be seen varying in intensity on male gnathopod 2 from the heavily notched palm of P. platensis found in Kent and Denmark (Stock and Biernbaum, 1994: 795-811) to the less pronounced depressions of P. platensis, Poland, the further reduced P. monodi from Ascension (Stock and Biernbaum, 1994: 795-811) and the sinuous palm with notches absent for P. japonica in Japan and China (Iwasa, 1939: 255-69; Hou and Li, 2003, 2442-48) illustrating a continuum of form (Fig.3).
The palm of gnathopod 2 was broadly convex allowing the dactyl to follow the curvature of the palm (Bate, 1862) (Fig.2). Figure 3 further illustrates this continuum of form in regard to the dactylus of gnathopod 2 in males, with a slender, distinct, "finger-like" projection apparent on the tip of the dactylus of P. platensis from the UK and Denmark (Stock and Biernbaum, 1994: 795-811) to a shorter "finger-like" appendage for P. platensis from Poland and no projection in P. japonica, P. monodi or P. platensis from Japan(Lacey, 2009).
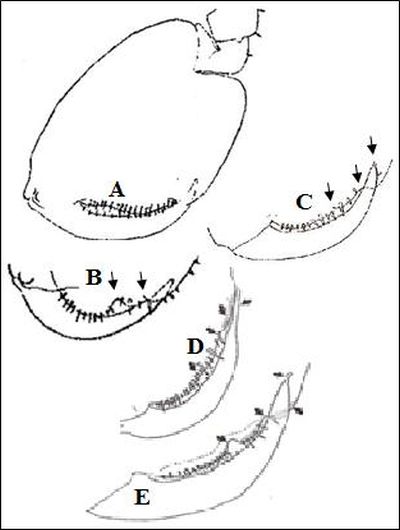
Figure 3: Platorchestia sp. male gnathopod 2; (A) P. japonica; (B) P. platensis, Japan; (C) P. platensis, Poland, (D) P. monodi from Ascension, (E) P. platensis from Denmark. A, B, C adapted from: Iwasa (1939) and D, E adapted from: Stock and Biernbaum (1994).
The P. platensis population from the UK seem to be most similar to P. platensis from Japan due to the distinct 'V' shaped notch in the palm of male gnathopod 2, as the notch in P. platensis from Denmark seems to be more 'U' than 'V' shaped. Despite this, P. platensis from Japan has no "spike-like" appendage at the end of the dactylus suggesting that the Kent population of P. platensis was either a Danish species which has adapted in geographical isolation or a new sub-species, however molecular genetic tests are needed to confirm this.
Distribution over time
In 1947, Reid (1947: 1-25) conducted a study of British Talitridae, recording O. gammarellus, O. mediterranea and Talorchestia deshayesii around the majority of the British coast, with T. brito distributed along the south coast and O. bottae recorded from the shore of the River Thames at Isleworth Ferry, Richmond, Oxford, River Yare, Brundall and Norfolk. Reid (1947: 1-25) adds that although Orchestia platensis (now named P. platensis ) was not present, it could arrive to Britain at any time, as it was present on the coasts of Germany, Denmark and Sweden.
In 1968, Wildish found a new species Orchestia remyi roffensis, renamed by Wildish (1969: 288-90) as O. roffensis, which had not been previously recorded in Britain (Reid, 1947: 1-25). The closest known species to that found were O. remyi, the lectotype of which were described from only two males, four females and one juvenile from a freshwater cave in Bonifacio, St. Bathelémy, Corsica (Wildish, 1969: 288-90), although no such species have been found anywhere in the world since so the results are dubious. Wildish and Lincoln (1979:199-200) also found another species for the first time in Britain, namely P. platensis. The type locality of this finding was Seasalter, Kent, although after sampling this site extensively during this study no P. platensis were found. P. platensis were however found 3km along the coast at Whitstable.
Wildish (1987: 571-83) stated that O. gammarellus, O. roffensis, O. mediterranea, O. aestuarensis and O. cavimara have been found in the Medway Estuary tabulating their distribution limits using mean salinity. However, Wildish fails to give descriptions of the sampling sites, simply providing old ordinance survey map grid references so the distribution cannot be accurately mapped below. Despite this, it can be assumed that O. cavimara occurs upstream of the River Medway as it was commonly found in freshwater ecosystems in Britain (Curry et al., 1972: 55-56) and that O. aestruarensis was found lower downstream but still within the Medway Estuary as it appears to be limited to estuarine locations (Wildish, 1987: 571-83). As O. gammarellus, O. roffensis and O. mediterranea are have been found on fully marine shores (Wildish, 1987: 571-83), it can be assumed that these were found along the north coast of Kent in the Thames Estuary. Regrettably, the Medway Estuary was not sampled, as most of Wildish's (1987: 571-83) original sites have been built upon and was inaccessible. Spicer (2010) attempted to re-sample these sites in 1996, finding only O. gammarellus in the Medway Estuary, Orchestia mediterranea around the Isle of Sheppey and Talitrus saltator on the north coast of Kent. Spicer (2010) continued his sampling in 1998 further up the south west coast to Essex, where he found only O. gammarellus.
These results suggest a dramatic shift in the community composition in the south east of England over the last 43 years with, O. mediterranea, T. saltator, O. aestuarensis, O. roffensis, O. cavimara and P. platensis suffering extirpation, with P. platensis reappearing 32 years later probably due to reintroduction. O. gammarellus appears to be abundant around the southeast over the last 15 years, despite not being recorded prior to 1996 (Fig.4).
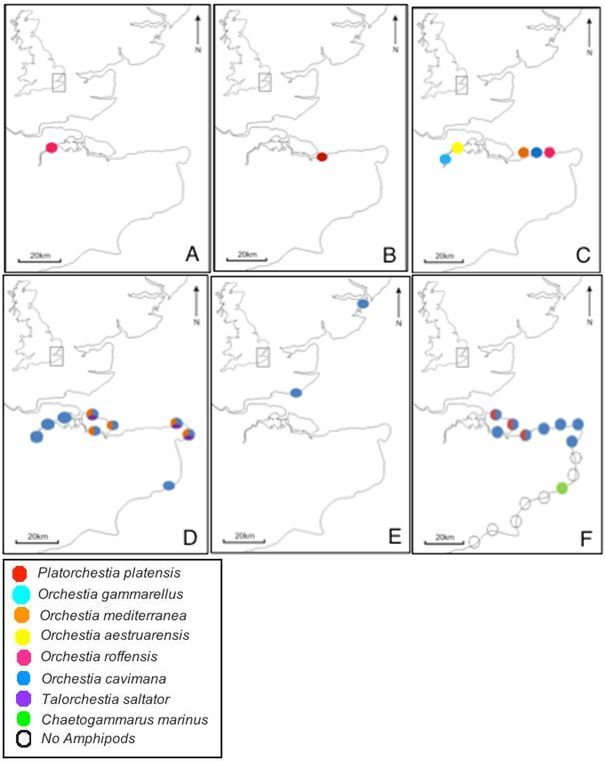
Figure 4: Pie charts showing number of species found and their distribution over time (A-F). Samples collected by Wildish (1969) (A), Wildish and Lincoln (1979) (B), Wildish (1987) (C), Spicer (2010) data collected in 1996 (D), Spicer (2010) data collected in 1998 (E) and this study's results (F).
Discussion
Amphipod Distribution
The abundance of P. platensis differs between sites and over time, which supports the finding that P. platensis populations demonstrate spatial and temporal variation (Behbehani and Croker, 1982: 611-20). P. platensis was only found at the three sites closest to the River Thames. This spatial distribution could be because P. platensis requires lower optimum water quality than O. gammarellus (MacNeil et al., 2009: 232-41). As the River Thames effluent contains higher organic matter and pollutants than the open ocean, it makes sense for P. platensis to survive better further up the estuary. Another factor, which could explain the distribution seen, would be the salinity, measured as 0.5-30ppt in the lower reaches of the River Thames (Environment Agency, 2011). Normant et al. (2007: 101-11) found that amphipods significantly alter the ratio between their resting and active metabolic rates in stressful salinities due to the maintenance energy needed for osmoregulation (increase in sub-normal and decrease in supra-normal). However, Normant et al. (2007: 101-11) concludes that single factors such as salinity cannot determine an invaders success, with Persson (2001: 201-10) stating that salinity does not affect brood size.
Behbehani and Croker (1982: 611-20) also found that P. platensis dominated all year round in all habitats, which does not concur with the findings in Kent where O. gammarellus either co-exists or out-competes P. platensis in number of individuals sampled. Elsewhere in the world, P. platensis has been found to out-compete O. gammarellus by enduring lower salinities (Persson, 2001: 201-10) and having a higher rate of reproduction (Dahl, 1946:1-53). Dahl (1946:1-53) predicted that P. platensis would be competitively superior to O. gammarellus due to its higher rate of reproduction (Backlund, 1945: 1-236; Dahl, 1946:1-53). Persson (2001: 201-210) supports this, stating that P. platensis has a longer period of reproduction, higher rate of juvenile development and larger broods for females of equal size when compared with O. gammarellus. P. platensis can also tolerate higher (30%) water loss (Morritt and Spicer, 1998: 1965-82; Humberto and Garcés, 1988: 1-10) and was a more active jumper than O. gammarellus, so was better at coping with desiccation and escaping from predators (Karlbrink, 1969:327-34).Behbehani and Croker (1982: 611-20) state that the mobility, aggregation and aggressiveness of P. platensis assist the species in establishing and maintaining populations in wrack habitats. This aggressiveness may explain how P. platensis out-competes O. gammarellus (Backlund, 1945: 1-236; Dahl, 1946:1-53; Persson, 2001: 201-10; Bock, 1967: 405-28; Karlbrink, 1969:327-34).
Persson (2001: 201-10) proposed that resource exploitation, interference competition, predation and hybridisation cause the exclusion or displacement of O. gammarellus, while Dick et al. (1993: 79-88) suggests differential cannibalism and mutual predation cause this displacement. However this displacement occurs, the outcome of the interactions between introduced and native species may also be balanced by environmental factors, giving different results in different areas (Dick et al., 1997: 209-16; Dick and Platvoet, 2000: 977-83). For example, Dahl (1946:1-53), Den Hartog (1963:40-67) and Karlbrink (1969:327-34) reported the occurrence of sympatric populations of the two species in the Netherlands and Sweden. Environmental factors may therefore explain why P. platensis had not outcompeted O. gammarellus in the UK.
Natural History
Semi-terrestrial species behaviourally select damp substrates and high humidity microclimates to inhabit, such as wrack beds, to avoid dehydration (Humberto and Garcés, 1988: 1-10). The micro-distribution of P. platensis was therefore dependent on wrack occurrence (Morino, 1978:245-67). Wrack tends to build up on sheltered, protected beaches as it was not washed away. For this reason, P. platensis was found on protected beaches (Morino and Ortal, 1995, 68: 824-32) as the strandline was disturbed and washed away less frequently.
Unlike most Talitridae, P. platensis have been observed in a variety of habitats; wrack beds on pebbly, shingle and sandy shores, estuaries, salt marshes, mangroves, beneath dead leaves on the upper shore (Persson, 2001: 201-10; Iwasa, 1939: 255-69; Karlbrink, 1969:327-34; Bousfield, 1958: 883-98; Teigsmark, 1981: 65), in coastal defences on the upper shore and up to altitudes of 792m (Spicer, 2010). This supports the findings that P. platensis can exist on both pebbly and 'oaty' substrates consisting of fine sand and large quantities of broken shell.
At all the beaches where P. platensis were found, plunging breakers were found meaning that the bathymetry offshore leading up to the beach was steep, as otherwise the wave would break further out to sea. This was significant as it means that P. platensis can be transported secondarily, along the shore via floating wrack successfully.
It was thought that C. marinus may have been transported to the site within the ferry port of Dover by ballast water because the beach was extremely sheltered within the ferry port making current transport highly unlikely.
No amphipods were found on the south or east coasts of Kent as the coastline was exposed to strong south-westerly winds and rapid currents amplified by the narrowing of the English Channel; this results in the beaches' fine sand substrate (Bousfield, 1958: 883-98) with very little strandline to provide the amphipods with shelter and nutrition.
The sampling method used captures only a rough approximation of the quantitative composition of a community affecting the reliability of results. This was because older amphipods were bigger and tended to curl up to play dead instead of jumping away like the energetic juveniles, so they were easier to catch leading to a possible sampling bias. Furthermore, at one of the three sites site where P. platensis were recorded, a dense population of P. platensis were found in the last 8min of sampling leading to a false representation of the abundance of P. platensis at the site.
Morphological Comparison
No significant difference was found in total body length between P. platensis and O. gammarellus, suggesting that the number of individuals sampled may have been too small, as the literature states that P. platensis is the smaller species. Normant et al. (2007: 101-11) found that individuals of smaller body mass have a higher metabolic rate due to larger absolute heat production rates. The metabolic rate of an organism is defined as the rate at which an organism transforms energy and materials into biological structure and function (Atkinson, 2011). Another invasive amphipod species, Gammarus tigrinus, uses this to its advantage to increase colonisation success, with the females increasing their metabolic rates to cope with the energetic demands of a bivoltine life cycle. They do this by producing eggs in their ovaries while simultaneously developing embryos in their marsupium (Normant et al., 2007: 101-11). P. platensis also exhibit bivoltine life cycles and were more active than O. gammarellus, so theoretically would metabolise consumed resources quicker, needing to feed more frequently.
Both P. platensis and O. gammarellus males were found to be longer than females which supports Humberto et al. (1991, 54: 1-10), who add that mature males also have conspicuously more robust antennae 2, enlarged gnathopod 2 and penile papilla at the base of the 7thpereopods. This difference in size was probably due to sexual dimorphism, which is well known among crustaceans (Normant et al., 2007: 101-111).
The sex ratio of male: female in O. gammarellus was 75:118 and for C. marinus 8:27, suggesting that there were more females than males in amphipod populations. This was thought to be because increasing the number of reproducing individuals increases the reproductive potential of a population (Wildish and Lincoln, 1979:199-200; Humberto et al., 1991: 1-10).
Distribution over time
It is believed that P. platensis reached the UK coastline primarily through ballast water transport (Simpson, 2011: 278-92).Karlbrink (1969:327-34) said that ballast water was the most likely vector of transport, from the Dutch Wadden Sea to the Thames Estuary as both these sites were in close proximity to international ports. Most taxa with a planktonic phase in their life cycle have been found in ballast water, as were all major marine habitat and trophic groups (Carlton and Geller, 1993:76-82). It was thought that the high salinity tolerance of P. platensis allows it to survive ballast changes both mid-ocean and in coastal waters (Lacey, 2009). Persson (2001: 201-10) demonstrating that P. platensis had an LT50 value (mean time taken for 50% of population to die) of 15-22 days in low salinity of 0.5ppt, no mortality after 5 days in high salinity of 51ppt and had an LT50 value of 117 hours in 35ppt. However, although this method would provide a clear link between primary and secondary sites of P. platensis, further research is needed.
It was interesting to note that although no P. platensis were recorded at Wildish and Lincoln's (1979:199-200) original site, P. platensis were recorded 3km east along the Kent coast. This suggests that P. platensis could have become locally extinct at Wildish and Lincoln's (1979:199-200) original site at some point in the last 31 years, but either dispersed to the secondary site before hand or was later reintroduced to the second site, most probably by ballast water.
As amphipods cannot move along the coast due to gaps in strandline, it was unclear whether the P. platensis was introduced to the three sites by ballast water, secondary dispersal from one site of ballast water introduction or re-introduction at one site by either ballast water or wrack transport over varying time scales. Transport due to ballast water has only occurred within the past 100 years with the rapid expansion of human travel and exploration (Carlton, 2011:265-73). This was not generally considered enough time for significant mutation and recombination of genes (Lacey, 2009). While it may be that given time speciation will occur to the degree that the populations form discrete species, this has not yet been evidenced (Lacey, 2009). Further investigation should compare molecular genetics, combined with experimental study into the ability of intra-population reproduction, and the morphology and viability of the offspring (Carlton, 2011:265-73).
Only a small percentage of the original population survive transportation, reducing the genotypic variation in the original population, further reducing during the initial stages of colonisation leading to genetic bottleneck events (Carlton, 2011:265-73). Selective forces such as predation, competition and disease would cause variation of form and physiology from the source populations with secondary dispersal increasing the likelihood of this (Lacey, 2009). As P. platensis can only move vertically up or down the beach profile and not horizontally along the beach, the only method of transport alongshore was on floating wrack (Spicer, 2010). Carlton (2011:265-73) stated that the range expansions of invasive species can be predictable along natural corridors such as in the case of P. platensis along coastlines. The likelihood of successful colonisation increases positively to the number of individuals being secondarily dispersed the frequency of their dispersal and therefore the propagule pressure on a secondary site (MacNeil et al., 2009).
Clark (2011) stated that many invasive species have been transported to the UK through the shellfish industry, either on or in the shellfish themselves during transplantation, on spat or on farming equipment. Clark (2011) also suggested that this could be a possible method of invasion by P. platensis in the Thames Estuary due to the intensive shellfish farms around Whitstable. In the UK strict protocols have been put in place to log the transport of shellfish to and from shellfish farms. The shellfish of which must be depurated for at least 48 hours prior to sale and which originate from the same seed stock to reduce the risk of contamination by disease or epiphytes (Clark, 2011). It would be interesting to investigate further whether shellfish have been transplanted to the Whitstable area from areas where P. platensis have been found.
Conclusion
P. platensis were found in the UK reproducing successfully and coexisting with the native species O. gammarellus. P. platensis were most likely to have been transported here in ballast water, expanding their range through secondary dispersal from the original site via floating wrack or through recurrent ballast water introductions. This study also investigated the abundance and distribution, both current and historical, of amphipod species in southeast England, finding that amphipod populations in the southeast were in a constant state of flux with O. gammarellus most prevalent.
The implications of this study are that if like elsewhere in Europe P. platensis outcompetes the native species O. gammarellus there could be negative effects on tourism to the southeast and reduce biological diversity decreasing the amount of available nutrients within the strandline to other organisms.
As our knowledge and understanding of the number, diversity and transport vectors of biological invasions in the marine environment are limited further monitoring of amphipod populations in Kent is needed. It would also be interesting to investigate the presence of P. platensis further up the River Medway, River Thames and north along the east coast of Essex to clarify P. platensis distribution. Genetic research could also clarify the taxonomy of Platorchestia spp., which would answer whether the P. platensis found in the UK is indeed a new sub-species and also tell us the UK populations origins.
Acknowledgments
I wish to thank R. Ticehurst for the use of sampling equipment, M. J. Simpson for transportation and financial support during sampling, J. I. Spicer for species verification, critically reviewing the text and for his detailed subject knowledge, G. Harper for training on the scanning electron microscope, N. Lacey for help identifying morphological characteristics and sourcing primary literature and S. Pitman for assistance using CorelDraw.
List of Illustrations
Figure 1: Pie charts showing the percentage abundance and distribution of Amphipoda found around Kent. Showing Platorchestia platensis (red), Orchestia gammarellus (blue), Chaetogammarus marinus (green) with a black circle indicating an absence of amphipods. Created by the author.
Figure 2: Male gnathopod 2 in P. platensis from Kent, UK noticing the distinct notch in the palm of the gnathopod 2 and "spike-like" appendage at the end of the dactylus. Created by the author.
Figure 3: Platorchestia sp. male gnathopod 2; (A) P. japonica; (B) P. platensis, Japan; (C) P. platensis, Poland, (D) P. monodi from Ascension, (E) P. platensis from Denmark. A, B, C adapted from: Iwasa (1939) and D, E adapted from: Stock and Biernbaum (1994). Created by the author.
Figure 4: Pie charts showing number of species found and their distribution over time. O. gammarellus (blue), P. platensis (red), O. mediterranea (orange), T. saltator (purple), O. aestuarensis (yellow), O. roffensis (pink), O. cavimana (dark blue), C. marinus (green) and empty circles denoting the absence of amphipods. Samples collected by; Wildish (1969) (A), Wildish and Lincoln (1979) (B), Wildish (1987) (C), Spicer (2010) data collected in 1996 (D), Spicer (2010) data collected in 1998 (E) and this study's results (F). Created by the author.
Appendices
Appendix 1 - Amphipod Distribution
Number of O. gammarellus, P. platensis and C. marius at each site.
| Site name | Number of O. gammarellus | Number of P. platensis | Number of C. marius |
|---|---|---|---|
| Camber sands | 0 | 0 | 0 |
| Dungeness | 0 | 0 | 0 |
| St Mary's Bay | 0 | 0 | 0 |
| Dymchurch | 0 | 0 | 0 |
| Folkestone | 0 | 0 | 0 |
| Dover | 0 | 0 | 34 |
| Deal | 0 | 0 | 0 |
| Sandwich bay | 0 | 0 | 0 |
| Ramsgate | 1 | 0 | 0 |
| Margate | 3 | 0 | 0 |
| Reculver | 61 | 0 | 0 |
| Whitstable | 40 | 1 | 0 |
| The Swale-Isle of Harty | 54 | 0 | 0 |
| Leysdown on sea | 16 | 2 | 0 |
| Minster | 2 | 2 | 0 |
| Allhallows | 17 | 0 | 0 |
Appendix 2 - Natural History
The fauna and flora found in the strandline, the beach substrate and profile, any anthropogenic impacts, the weather and air temperature at every site visited
| Site name | Fauna | Flora | Strandline | Substrate | Beach Type | Anthropogenic Impacts | Weather | Air Temp (*C) |
|---|---|---|---|---|---|---|---|---|
| Camber sands | Bryozoan, seabirds | Sargassum muticum, Fucus ceranoides, Fucus spiralis, Enteromorpha intestinalis, Ulva lactuca | Very few small patches | Sand | Spilling breakers, Wide surf zone, big dunes back of beach-good succession, Dissipative | Well used, tourism, dog walkers, well trodden, many car parks, cafes, wind surfers groynes burger bar, 'dredged' by tractor | Torrential rain | 18.5 |
| Dungeness | Seabirds | Fixed grasses | NA | Fine sand Large pebbles on lower shore | Spilling breakers wide surf zone, v dissipative | NA | Overcast | 19 |
| St Mary's Bay | Mussels | U. lactuca, F. ceranoides | NA | Fine shingle on upper shore Sand | Spilling breakers dissipative beach | Groynes, seawall, well used car park, ice cream shop | Overcast | 19 |
| St Mary's Bay 2 | Razor clam, mussels, scallops, oyster, crab moult, flies | S. muticum, U. lactuca, F. vesiculosus, Ascophylum nodosum, filamentous green algae, F. ceranoides | Continuous but dry | Lots of burrows, sand falls away in layers when dug up | Spilling breakers dissipative beach | Groynes, seawall, well used car park, ice cream shop | Overcast | 19 |
| Dymchurch | Mussel beds in and on boulders, anenome, Arenicola marina, Patella vulgata, barnacles, Littorina nigrolineata | F. ceranoides, U. lactuca, E. intestinalis | NA | Sand and a few pebbles | Spilling breakers, wide surf zone, dissipative beach | Seawall, large boulders groynes well fortified as next to MOD range | Cloudy | 19 |
| Dymchurch 2 | Barnacles, mussels, scallops | filamentous red and green algae, A. nodosum, F. ceranoides, F. vesiculosus, F. spiralis | NA | Sand and a few pebbles | Spilling breakers, wide surf zone, dissipative beach | seawall, large boulders groynes well fortified as next to MOD range | Overcast | 18.5 |
| Folkestone | Mussels, limpets, beach fleas, wasps, L. obtusata, L. nigrolineata, barnacles | Mastercarpus stellatus, Chondrus crispus, U. lactuca, F. vesiculosus, filamentous red algae, F. serratus, Palmaria palmata, Laminaria saccharina, L. digitata, E. intestinalis, Spongomorpha arcta | Medium thick, fresh and degraded. Algae, very slimy- maybe algae breaks down produce sugars, explains the wasps? | Shingle upper to pebbles lower beach | Reflective sheltered beach very calm sea spilling breakers | Groynes, seawall, beach huts boulders to protect the coast in front of groynes beach is cleaned, close to town, developed for public as open space café toilets play park | Breeze but hot and few clouds | 19 |
| Folkestone 2 | Juvenile spirobis, beach fleas, seabirds, mussels, encrusting bryozoan. | F. serratus, epiphytic filamentous red, small Palmatia epiphyte, P. palmata U. lactuca, M. stellatus | Shingle upper to pebbles lower beach | Reflective beach v calm sea spilling breakers | As above | |||
| Dover | Seabirds, mussel seed, epiphytic bryozoan on L. digitatata | L. digitata, F. spiralis, Ceramium sp., Calliblepharis ciliata, U. lactuca | NA | Rocky shingle | Dissipative plunging breakers | Sea wall, in inner harbour, groynes, sailing club, built up area, main road<5om from sea | Overcast and quite windy | 19 |
| Dover 2 | Palmonid shrimp | NA | Rocky shingle | Dissipative plunging breakers | sea wall, in inner harbour, groynes, sailing club, built up area, main road<5om from sea, Main shipping harbour | Overcast | 21.5 | |
| Deal | Seabirds, Chartella papyraceae | NA | NA | Pebbly shingle | 50:50 reflective: dissipative plunging breakers, wide surf zone, long expanse of beach | Pier, main road <100m from sea, well used, very close to main shipping channel for dover | Breeze mild | 21 |
| Deal 2 | Dog whelk eggs Urosalpinx cinerea, C. papyraceae | S. muticum, F. vesiculosus, A. nodosum, P. pamata, U. lactuca | Minimal | Pebbly shingle | 50:50 reflective: dissipative plunging breakers, wide surf zone, long expanse of beach | As above | Clear sunny | 17.5 |
| Sandwich bay | C. papyraceae, barnacles, 5 dogfish cases, bryozoan on one egg case, Solen marginatus, Petricola pholadiformis | A. nodosum, U. lactuca, F. serratus, F. vesiculosus | NA | Pebbly shingle | Reflective, spilling breakers calm water wide surf zone | Dirt car park and toll, power station in distance | Mild breeze | 21.5 |
| Sandwich bay 2 | C. papyraceae mussels razor clams, flies spirobis sp. Seabirds dogfish eggs epiphytic bryozoan on F. vesiculosus, dogfish eggs, Chlamys distorta elasmobranch egg case-ray/ skate, Donax vittatus, Buccinum undatum, Cerastderma edule, family Archidae chlamys multistiata | S. muticum, F. vesiculosus, F. serratus A. nodosum, U. lactuca, C. crispus, Palmatia sp. (smaller one similar) filamentous red algae | As above | As above | As above | Clear sunny | 21.5 | |
| Ramsgate | limpet, pupae, woodlouse, beach fleas, chartella papyraceae, seabirds, whelk, epiphytic bryozoan on sheet red algae maybe P. palmatia | L. digitata, F serratus, serratus, F. corallina, U. lactuca, F. ceranoides, E. intestinalis, Cladophora rupestris, Ceramium sp., P. palmatia | Thin patchy but regular line | Fine sand with rocks at bottom of beach covered in algae forming small low rock pools | Dissipative spilling breakers, wide surf zone | Seawall, groyne well used, wind farm in distance. Numerous burrows of varying size but minimal if any movement when dug up/ disturbed | Overcast windy light rain | 20.5 |
| Ramsgate 2 | flies beachfleas spider | F. serratus, P. palmate, U. lactuca, filamentous red epiphyte, epiphytic bryozoan, L. digitata | thin patchy but regular line | thick sandy- perfect for sand castles down to 10cm then stones and gravel (possibly artificial) | As above | Sunny | 18 | |
| Margate | Seabirds, crab, mussel, limpet, C. papyraceae | U. lactuca, F. serratus, E. intestinalis, P. palmatia filamentous pinky red algae and filamentus blood red thin algae Calliblepharis ciliata/ Gelidium latifolium, Furcellaria umbricalis | Dense mats of green, red and brown algae | Sand | Dissipative spilling breakers thin surf zone wide beach | Built up main road on sea front sea wall 'dredged' well used, Dense algal cover in breaking waves, lots of plastics and rubbish in strandline, effluent pipe at end of beach. | Rainy overcast | 19.5 |
| Margate 2 | Flies, C. papyraceae | A. nodosum, F. serratus U. lactuca, S. muticum, E. intestinalis, | Thick continuous patchy | Sand | As above | As above | Sunny and cloudless | 18.5 |
| Reculver | Barnacles, mussels, oysters, C. papyraceae, C. edule, Thracia sp., Crepidula fornicata | F. vesiculosus, A. nodosum filamentous pinky red algae and filamentus blood red thin algae C. ciliata/ G. latifolium, F. umbricalis, U. lactuca, F. serratus, F. spiralis, C. crispus | Thin | Pebbly | Dissipative spilling breakers thin surf zone | Seawall, groynes, big boulders, caravan park 20m from beach, oyster farm and water quality station <50m from seafront. | Rainy overcast | 16 |
| Whitstable | L. neritoides, Xantho sp., mussel, oyster, flies, C. edule, C. fornicata, C. papyraceae, Chlamys sp. And 2 whelk sp. (kept) | Corallina sp., A. nodosum, S. muticum, U. lactuca, F. spiralis, F. vesiculosus, G. latifolium, E. intestinalis Ectocarpus siliculosus | Thick and degraded | Pebbly | More dissipative than reflective, plunging breakers, thin surf zone sea very calm | Beach huts main road <100m from sea groynes sea wall skate park car park, sailing club well used, wind farm offshore, oyster factory, heavy rain day before. | Rainy overcast | 20.5 |
| The Swale, Isle of Harty | ladybug, spider, flies, beach fleas, maggots, chlamys sp.carcinus maenas | fucus spiralis, ascophylum nodosum, ulva lactuca, fucus vesiculosus, enteromorpha intestinalis | Wide thin degraded | Stony and concrete | Seawall, jet skiers, shipwreck, ferry pier, car park, ferry no longer in use, unmaintained just concrete. | Overcast | 19.5 | |
| Leysdown on sea | C. edule, C. fornicata, S. marginatus, barnacles, spider, beetle, flies, C. papyraceae whelk sp. >6cm long | F. vesiculosus, U. lactuca, A. nodosum, F. spiralis | Thin patchy continuous | Fine sand and broken shell- texture of oats | Dissipative, wide surf zone wide beach plunging breakers | Seawall, MASSIVE caravan park and seaside resort, jet skiers, car parks above and on beach, well used, horse riders- poo, kite surfers, groynes, wind farm. In sand amphipods less energetic- behaviour change curl up and play dead don't jump so much lighter in colour. | Overcast and windy | 18.5 |
| Minster | Maggots, spider, flies mussels, weird bug (caught), barnacles, S. marginatus, Littorina sp. Whelk sp. >6cm long oysters, C. edule | A. nodosum, F. spiralis, U. lactuca, E. intestinalis | Patchy thin continuous sparse and dry not degraded | Pebbly | Reflective plunging waves thin surf zone | Seawall, groynes, busy shipping lane offshore, well used for angling and dog walking, walked a good mile along strandline digging every couple of metres only found amphipods under one piece of strandline in last 8 minutes. | Sunny and breezy | 18.5 |
| Allhallows | Mussels, Scallops Littorina sp. Seabirds, Flies, Oyster | Seagrass, F. ceranoides. F. vesiculosus, A. nodosum, M. stellatus, E. intestinalis, U. lactuca | continuous patchy damp | Shingle and sand | Dissipative, spilling thin surf calm, sheltered beach | Groynes, seawall, leisure park | Overcast and warm |
Notes
[1] Rebekah Simpson achieved a 1st class BSc (Hons) Marine Biology and Oceanography in 2011 before going on to complete her MRes in Marine Biology in October 2012
References
Andersson, A. (1962), 'On a collection of amphipoda of the family talitridae from the Canary Islands', Arkiv. Zoology, 15, 211-18
Atkinson, D. (2011), 'Predicting biological rates based on boundaries: Metabolic scaling theory', University of Plymouth Seminar Series, 4 February 2011
Backlund, H. O. (1945), 'Wrack fauna of Sweden and Finland ecology and chorology', Opuscula Entomologica Supplementum, 5, 1-236
Bate, C. S. (1862), 'Catalogue of the specimens of amphipod crustaceans', in The Collection of the British Museum, London, p. 399
Bate, C. S. and O. J. Westwood (1863), A History of the British Sessile-eyed Crustacea, London
Behbehani, M. L, and R. A. Croker (1982), 'Ecology of beach wrack in northern New England with special reference to Orchestia platensis', Estuarine, Coastal and Shelf Science, 15, 611-20
Bock, K. D. (1967), 'Experimente zur Okologie von Orchestia platensis', Zeitschrift Morphologie und Okologie der Tiere, 58, 405-28
Bousfield, E. L. (1958), 'Distributional ecology of the terrestrial Talitridae (Crustacea: Amphipoda) of Canada', Proceedings of the Tenth International Congress of Entomology, 1, 883-98
Bousfield, E. L. (1973), Shallow-water Gammaridean Amphipoda of New England, Ithaca: Cornell University Press
Bousfield, E. L. (1982), 'The amphipod superfamily talitroidea in the north-eastern Pacific region. Family talitridae: Systematics and distributional ecology', Biological Oceanography, 11, 26-30
Carlton, J. T. (1989), 'Man's role in changing the face of the ocean: biological invasions and implications for conservation of near-shore environments', Conservation Biology, 3, 265-73
Carlton, J. T. (2011), 'Invasions of the Sea: the Science, History and Policy of Adding Species to Marine Communities', Marine Invasive Seminar at the Marine Biological Association of the United Kingdom, 7 February 2011
Carlton, J. T. and J. B. Geller (1993), 'Ecological Roulette: The Global Transport of Nonindigenous Marine Organisms', Science, 261, 76-82
Chilton, C. (1921), 'Fauna of the Chilka Lake', Memoirs of the Indian Museum, 5, 519-58
Clark, S. (2011), Personal Communication
Cohen, A. N., J. T. Carlton and M. C. Fountain (1995), 'Introduction, dispersal and potential impacts of the green crab Carcinus maenas in San Fransisco Bay, California', Marine Biology, 122, 225-37
Curry, A., R. F. Grayson and T. D. Milligan (1972), 'New British records of the semi-terrestrial amphipod Orchestia cavimana', Freshwater Biology, 2 (1), 55-56
Dahl, E. (1946), 'The Amphipoda of the Sound', Part I, Terrestrial Amphipoda. Acta University Lund, N.F. Adv waters, 2(42), 1-53
Den Hartog, C. (1963), 'The amphipods of the Deltaic region of the river Rhine, Meuse and Scheldt in relation to the hydrography of the area', Journal of Sea Research, 2, 40-67
Dick, J. T. A., I. Montgomery and R. W. Elwood (1993), 'Replacement of the indigenous amphipod Gammarus duebeni celticus by the introduced G. pulex: differential cannibalism and mutual predation', Journal of Animal Ecology, 62, 79-88
Dick, J. T. A., N. Nelson and J. D. D. Bishop (1997), 'Introduction experiments with Gammarus spp. (Crustacea: Amphipoda) in the Isle of Man (British Isles), 1949- 1995', Journal of Zoology, London, 242, 209-2,16
Dick, J. T. A. and D. Platvoet (2000), 'Invading predatory crustacean Dikerogammarus villosus eliminates both native and exotic species', Proceedings of the Royal Society B, 267, 977-9,83
Drake, J. M and D. M. Lodge (2007), 'Hull fouling is a risk factor for intercontinental species exchange in aquatic ecosystems', Aquatic Invasions, 2 (2), 121-1,31
Environment Agency (2011), 'Floating down the river: The River Thames-its geology, geography and vital statistics from source to sea', available at www.the-river-thames.co.uk/thames.htm, accessed 7 March 2011
Friend, J. A. and A. M. M. Richardson (1986), 'Biology of terrestrial amphipods', Annual Review of Entomology, 31, 25-48
Hayward, P., T. Nelson-Smith and C. Shields (1996), Sea Shore of Britain and Europe, London: Harper Collins
Hou, Z. and S. Li (2003), 'Terrestrial talitrid amphipods from China and Vietnam', Journal of Natural History, 37 (20) 2442-48
Humberto, A. and B. Garcés (1988), 'Dessication tolerance of Platorchetsia platensis (Kröyer, 1845) Amphipoda: Talitridae)', Revista de Biologia Tropical (San Jose), 36 (1), 63-66
Humberto, A., B. Garcés and A. Marsh (1991), 'Studies on the distribution and ecology of Platorchestia platensis (Kröyer, 1845) at Lake Wyman, Boca Raton, Florida', Quarterly Journal of the Florida Academy of Sciences, 54 (1), 1-10
Invasive Species Advisory Committee (ISAC) (2006), 'Invasive Species Definition Clarification and Guidance White Paper', National Invasive Species Council, available at www.doi.gov/NISC/global/ISAC/ISAC_documents/ISAC%20Definititions%20White%20Paper%20%20-%20FINAL%20VERSION.pdf, accessed 12 January 2011
Iwasa, M. (1939), 'Japanese Talitridae', Zoology, 6 (4), 255-69
Jo, Y. W. (1988), 'Talitridae (Crustacea-Amphipoda) of the Korean coasts', Beaufortia, 38 (7), 153-79
Karlbrink, F. (1969), 'Distribution and dispersal of Talitridae (Amphipoda) in southern Sweden', Oikos, 20, 327-34
Kunkel, B.W. (1910), 'The amphipods of Bermuda', Transactions of the Connecticut Academy of Arts and Sciences, 16, 1-115
Lacey, N. (2009), 'Description of the Polish population of Platorchestia platensis (Kröyer), with a comparison of P. monodi and P. japonica', unpublished BSc (Hons) thesis
Little, C. (1983), The colonisation of land: origins and adaptations of terrestrial animals, Cambridge: Cambridge University Press
Lockwood, J. L. and M. L. McKinney (2001), Biotic Homogenization, New York: Klumer Academic/Plenum Publishers
MacNeil, C., J. T. A. Dick, F. R. Gell, R. Selman, P. Lenartowitcz and N. H. B. Hynes (2009), 'A long-term study (1949- 2005) of experimental introductions to an Island: freshwater amphipods (Crustacea) in the Isle of Man (British Isles)', Diversity and Distributions, 15 (2), 232-41
Moore, P. G. and C. H. Francis (1985), 'On the water relations and osmoregulation of the beach-hopper Orchestia gammarellus (Pallas) (Crustacea: Amphipoda)', Journal of Experimental Marine Biology and Ecology, 94 (1-3) 131-50
Morino, H. (1978), 'Studies on the Talitridae (Amphipoda, Crustacea) in Japan. Part III, Life history and breeding activity of Orchestia platensis (Kröyer)', Publications of the Seto Marine Biological Laboratory, 24, 245-67
Morino, H. and R. Ortal (1995), 'Two Platorchestia species (Amphipoda, Talitridae) from Israel', Crustaceana, 68 (7), 824-83
Morritt, D. and J. I. Spicer (1998), 'The physiological ecology of talitrid amphipods: an update', Canadian Journal of Zoology, 76, 1965-82
Nichols, F. H., J. K. Thompson and L. E. Schemel (1990), 'Remarkable invasion of San Francisco Bay (California, USA) by the Asian Clam Potamocorbula amurensis (II). Displacement of a former community', Marine Ecology Progress Series, 66, 95-101
Normant, M., M. Feike, A. Szaniawska and G. Graf (2007), 'Adaptation of Gammarus tigrinus (Sexton, 1939) to new environments – some metabolic investigations', Thermochimica Acta, 458, 101-11
Persson, L. E. (2001), 'Dispersal of Platorchestia platensis (Kroyer) (Amphipoda: Talitridae) along Swedish coasts: A slow but successful process', Estuarine, Coastal and Shelf Sciences, 52, 201-10
Pinkster, S., M. Scheepmaker, D. Platvoet and N. Broodbakker (1992), 'Drastic changes in the amphipod fauna (Crustacea) of Dutch inland waters during the last 25 years', Bijdragen tot de Dierkunde, 61, 193-204
Reid, D. M. (1947), 'Talitridae (Crustacea, Amphipoda)', Synopses British Fauna, 7, 1-25
Ruffo, S. (1949), 'Amphipodes (II). Résultats du voyages de la Belgica en 1897–99', Rapports Scientifiques, Zoologie, Anvers: Commission administrative du Patrimoine de l'Institut royal des Sciences naturelles de Belgique, 1-58
Sala, O. E., F.S. Chapin III, J. J. Armesto, E. Berlow, J. Bloomfield, R. Dirzo, E. Huber-Sanwald, L. F. Huenneke, R. B. Jackson, A. Kinzig, R. Leemans, D. M. Lodge, H. A. Mooney, M. M. Oesterheld, N. L. Poff, M. T. Sykes, B. H. Walker, M. Walker and D. H. Wall (2000), 'Global Biodiversity Scenarios of the Year 2100', Science, 287, 1770-74
Scapini, F., F. Campacci and M. Audoglio (1999), 'Variation among natural populations of Talitrus saltator (Amphipoda): morphometric analysis', Crustaceana, 72 (7), 659-62
Serejo, C. S., and J. K. Lowry (2008), 'The Coastal Talitridae (Amphipoda: Talitroidea) of Southern and Western Australia, with comments on Platorchestia platensis (Kröyer, 1845)', Records of the Australian Museum, 60, 189-94
Simpson, R. (2011), 'The invasive biology of the talitrid amphipod, Platorchestia platensis in northwest Europe', The Plymouth Student Scientist, 4 (2), 278-92
Spicer J. I. (1996), Personal Communication
Spicer, J. I. (1998), Personal Communication
Spicer, J. I. and U. Janas (2006), 'The beachflea Platorchestia platensis (Kröyer, 1845): a new addition to the Polish fauna (with a key to Baltic talitrid amphipods)', Oceanologia, 48 (2), 321-29
Spicer, J. I. (2010), Personal Communication
Stebbing, T. R. R. (1906), 'Amphipoda I Gammaridea, Das Tierreich', Lief, 21, 806
Stephensen, K. (1935), 'Indo-Pacific terrestrial talitridae', Museum Occasional Papers, 10, 1-20
Stock, J. H. and C. K. Biernbaum (1994), 'Terrestrial amphipoda (Talitridae) from Ascension and Saint Helena (South Central Atlantic)', Journal of Natural History, 28, 795-811
Teigsmark, G. (1981), 'Orchestia platensis (Talitridae), an amphipod new to the Norwegian fauna', Sarsia, 66 (2), 165
Wildish, D. J. (1969), 'A new sub-species of Orchestia leach (Amphipoda: Talitridae) from Britain', Crustaceana, 16 (3), 288-90
Wildish, D. J. and R. J. Lincoln (1979), 'Occurrence of Orchestia platensis (Kröyer, 1845) (Amphipoda, Talitridae) in Britain', Crustaceana, 36, 199-200
Wildish, D. J. (1982), 'Talitoidea (Crustacea, Amphipoda) and the driftwood ecological niche', Canadian Journal of Zoology. 60, 3071-74
Wildish, D. J. (1987), 'Estuarine species of Orchestia (Crustacea: Amphipoda: Talitroidea) from Britain', Journal of Marine Biological Association of the United Kingdom, 67, 571-83
To cite this paper please use the following details: Simpson, R. (2012), 'The Current Status of Platorchestia Platensis in the United Kingdom', Reinvention: a Journal of Undergraduate Research, British Conference of Undergraduate Research 2012 Special Issue, www.warwick.ac.uk/go/reinventionjournal/issues/bcur2012specialissue/simpson. Date accessed [insert date]. If you cite this article or use it in any teaching or other related activities please let us know by e-mailing us at Reinventionjournal@warwick.ac.uk.
Poster from BCUR
Click on image to download full PDF version
Please note: Figure 4 removed until permission to reproduce received.