Bovine TB
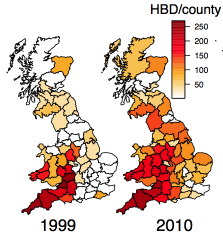 Bovine tuberculosis (bTB) is one of the most complex, persistent and controversial problems facing the UK cattle industry, costing the country an estimated £100 million per year. Cattle movements are a good predictor of disease incidence, although a causal relationship can only be identified in a small percentage of cases. The low sensitivity of the diagnostic test leads to considerable ambiguity in determining transmission routes, with the relative importance of direct cattle-to-cattle transmission and transmission via a local intermediary wildlife or environmental reservoir fiercely debated.
Bovine tuberculosis (bTB) is one of the most complex, persistent and controversial problems facing the UK cattle industry, costing the country an estimated £100 million per year. Cattle movements are a good predictor of disease incidence, although a causal relationship can only be identified in a small percentage of cases. The low sensitivity of the diagnostic test leads to considerable ambiguity in determining transmission routes, with the relative importance of direct cattle-to-cattle transmission and transmission via a local intermediary wildlife or environmental reservoir fiercely debated.
Work in Warwick has examined bTB from a variety of perspectives:
Ongoing Research
Building on our long history of quantitatively analysing the spread of bTB, our lastest project aims to include farmer behaviour within our predictive modelling framework. This will enable us to better understand the movement of cattle across the country (especially risky moves from areas of high infection to regions of low infection) and the likely acceptability of control measures including vaccination.
Key Elements of Past Research
Modelling
SBIDER researchers developed and parameterised a detailed stochastic spatio-temporal model for the spread and control of bTB in Britain. The model is founded on the recorded cattle movements and combined most within and between farm transmission. A stochastic SEI (Susceptible, Exposed (or Latent), Infectious) process, is used to capture the infection dynamics within each farm. Within-farm transmission is modelled as both direct (cattle-to-cattle) and indirect through the local farm environment, which allows persistence of infection. Spread between farms is captured by the movement of approximately 30,000 daily cattle movements that can transport infection, and by a regional infection reservoir that facilitates both short distance spread and persistence. A herd-level testing policy is incorporated that explicitly uses recorded routine and pre-movement test dates, and generates follow-up tests in accordance with government policy.
The model is able to capture the observed spread and rise of bTB cases over a twelve year period since 2000. It is currently being used to test a range of control strategies.
Network Analysis
Given the importance of cattle movements for the spread of bovine tuberculosis, it is somewhat surprising that spoligotypes (an equivalent of strains) exist in distinct spatial patches. However, work in Warwick is showing how these spatial patches are related to structures in the underlying movement network, and also that the localised nature of most movements keeps this patchy structure.
Statistical Analysis
Statistical analysis is focused towards the identifying risk factors for herds testing positive to bTB. Herd size, type of cattle (beef vs dairy), history of infection, spatial proximity to cases and movements from previously infected farms have all been shown to be important risk factors.
Grant (2009) Intractable Policy Failure: The Case of Bovine TB and Badgers
The failure to eliminate bovine TB from the English and Welsh cattle herd represents a long-term intractable policy failure. Cattle-to-cattle transmission of the disease has been under emphasised in the debate compared with transmission from badgers despite a contested evidence base. Archival evidence shows that mythical constructions of the badger have shaped the policy debate. Relevant evidence was incomplete and contested; alternative framings of the policy problem were polarised and difficult to reconcile; and this rendered normal techniques of stakeholder management through cooption and mediation of little assistance.
Carrique-Mas, Medley & Green (2008) Risks for bovine tuberculosis in British cattle farms restocked after the foot and mouth disease epidemic of 2001
The key findings of this work were that restocked herds were significantly more likely to break down at the first test after restocking if they had purchased cattle from previously infected, high risk, herds and also that where farms had had bovine TB prior to foot and mouth disease there was an increased risk of HBD, suggesting a local non-cattle source infecting these farms.
Courtenay, et. al. (2006). Is Mycobacterium bovis in the environment important for the persistence of bovine tuberculosis?
We show that the detectability of viable M. bovis at badger setts and latrines is strongly linked to the frequency of M. bovis excretion by infected badgers, and that putative M. bovis in the environment is prevalent on a large proportion of endemic cattle farms in Britain. These results raise important questions about the role of an environmental reservoir in bTB persistence, and opens the possibility of a non-invasive tool for monitoring transmission potential by badgers.
Publications:
Szmaragd, C., Green, L.E., Medley, G.F., Browne, W.J. (2013) Factors associated with herd restriction and de-restriction with bovine tuberculosis in British cattle herds. Preventive Veterinary Medicine, 111 (1-2), pp. 31-41.
Medley, G.F., Green, L.E. (2013) Enhancing the sensitivity of tests for bovine TB. Veterinary Record, 172 (4), pp. 96-97.
Green, L.E., Carrique-Mas, J.J., Mason, S.A., Medley, G.F. (2012) Patterns of delayed detection and persistence of bovine tuberculosis in confirmed and unconfirmed herd breakdowns in cattle and cattle herds in Great Britain. Preventive Veterinary Medicine, 106 (3-4), pp. 266-274.
Szmaragd, C., Green, L.E., Medley, G.F., Browne, W.J. (2012) Impact of imperfect test sensitivity on determining risk factors: The case of bovine tuberculosis. PLoS ONE, 7 (8), art. no. e43116,
Carslake, D., Grant, W., Green, L.E., Cave, J., Greaves, J., Keeling, M., McEldowney, J., Weldegebriel, H., Medley, G.F. (2011) Endemic cattle diseases: Comparative epidemiology and governance. Philosophical Transactions of the Royal Society B: Biological Sciences, 366 (1573), pp. 1975-1986.
Driscoll, E.E., Hoffman, J.I., Green, L.E., Medley, G.F., Amos, W. (2011) A preliminary study of genetic factors that influence susceptibility to bovine tuberculosis in the british cattle herd. PLoS ONE, 6 (4), art. no. e18806, .
Pontiroli, A., Travis, E.R., Sweeney, F.P., Porter, D., Gaze, W.H., Mason, S., Hibberd, V., Holden, J., Courtenay, O., Wellington, E.M.H. (2011) Pathogen quantitation in complex matrices: A multi-operator comparison of DNA extraction methods with a novel assessment of PCR inhibition. PLoS ONE, 6 (3), art. no. e17916, .
Ramírez-Villaescusa, A.M., Medley, G.F., Mason, S., Green, L.E. (2010) Risk factors for herd breakdown with bovine tuberculosis in 148 cattle herds in the south west of England. Preventive Veterinary Medicine, 95 (3-4), pp. 224-230.
Brooks-Pollock, E., Keeling, M. (2009) Herd size and bovine tuberculosis persistence in cattle farms in Great Britain. Preventive Veterinary Medicine, 92 (4), pp. 360-365.
Ramírez-Villaescusa, A.M., Medley, G.F., Mason, S., Green, L.E. (2009) Herd and individual animal risks associated with bovine tuberculosis skin test positivity in cattle in herds in south west England. Preventive Veterinary Medicine, 92 (3), pp. 188-198.
Grant, W. (2009) Intractable policy failure: The case of bovine TB and badgers. British Journal of Politics and International Relations, 11 (4), pp. 557-573.
Woodbine, K.A., Medley, G.F., Moore, S.J., Ramirez-Villaescusa, A.M., Mason, S., Green, L.E. (2009) A four year longitudinal sero-epidemiological study of bovine herpesvirus type-1 (BHV-1) in adult cattle in 107 unvaccinated herds in south west England. BMC Veterinary Research, 5, art. no. 5, .
Green, L., Medley, G. (2008) Cattle to cattle transmission of bovine tuberculosis: Risk factors and dynamics. Cattle Practice, 16 (2), pp. 116-121.
Courtenay, O., Wellington, E.M.H. (2008) Mycobacterium bovis in the environment: Towards our understanding of its biology. Cattle Practice, 16 (2), pp. 122-126.
Carrique-Mas, J.J., Medley, G.F., Green, L.E. (2008) Risks for bovine tuberculosis in British cattle farms restocked after the foot and mouth disease epidemic of 2001Preventive Veterinary Medicine, 84 (1-2), pp. 85-93.
Reilly, L.A., Courtenay, O. (2007) Husbandry practices, badger sett density and habitat composition as risk factors for transient and persistent bovine tuberculosis on UK cattle farms. Preventive Veterinary Medicine, 80 (2-3), pp. 129-142.
Sweeney, F.P., Courtenay, O., Ul-Hassan, A., Hibberd, V., Reilly, L.A., Wellington, E.M.H. (2006) Immunomagnetic recovery of Mycobacterium bovis from naturally infected environmental samplesLetters in Applied Microbiology, 43 (4), pp. 364-369.
Courtenay, O., Reilly, L.A., Sweeney, F.P., Hibberd, V., Bryan, S., Ul-Hassan, A., Newman, C., Macdonald, D.W., Delahay, R.J., Wilson, G.J., Wellington, E.M.H. (2006) Is Mycobacterium bovis in the environment important for the persistence of bovine tuberculosis? Biology Letters, 2 (3), pp. 460-462.
Green, L. (2005) Modelling disease - A guide for the cattle practitioner. Cattle Practice, 13 (3), pp. 243-248.
Green, L.E., Cornell, S.J. (2005) Investigations of cattle herd breakdowns with bovine tuberculosis in four counties of England and Wales using VETNET data. Preventive Veterinary Medicine, 70 (3-4), pp. 293-311.
Medley, G., Green, L., Ramirez, A.(2003) Bovine TB: Studies on the risk of herd breakdown Veterinary Record, 152 (2), pp. 59-60.
Funded by:
BBSRC, DEFRA
SBIDER people involved:
Orin Courtenay
Matt Keeling
Mike Tildesley
Internal collorabtors:
Liz Wellington
External collaborators:
Ellen Brooks-Pollock (Bristol)
Martin Green (Nottingham)
