Mary Halebian
Welcome!
I am currently a final year (3rd) PhD student part of the Medical Research Council (MRC) Doctoral Training Partnership at Warwick Medical School, UK. My supervisors are with Dr. Corinne Smith in the School of Life Science Department of University of Warwick and Prof. Frances Brodsky in UCL
General background
I finished my high school with a final overall grade of 92% from The Grammar School, Nicosia Cyprus and was always a keen enthusiast of science, more specifically Biology.
I studied Biology at the Kingston University London as an undergraduate degree in 2010 to 2013. My interests included areas of intracellular communications, protein biochemistry and bioenergetics. My final year undergraduate dissertation project was on the subject of enzymology; under the supervision of Dr. Robert Hatch. The aim of that project was the separation and analysis of the laccase enzymes from the fungus Pleurotusostreaus. This 6 month project allowed me to improve my laboratory skills especially on biophysical techniques and the confidence to work independently in a research environment and organise my own experiments.
In 2013, I decided to join the MRC-DTP course in the University of Warwick due to the structure of the program with year 1 as a MSc degree in Interdisciplinary Biomedical Research (IBR), following with a 3 year PhD in the area of quantitative biology.
During my MSc degree, I had the chance to broaden my knowledge in different disciplines, including computational modelling and mathematical skills (Rstudio, MatLab, Fiji) as well as laboratory skills (microscopy and biophysical techniques) through the two mini projects in term 3 of the MSc degree. The two (12 week) research mini projects have enhanced my research laboratory skills and extended my knowledge on new techniques related to areas such as structural biology (kinetics and imaging) and cell signalling through GPCRs using yeast/mammalian chimeras and mammalian organisms. The skills I acquired through my undergraduate dissertation and the mathematical, computational and biological skills from my MSc degree, I believe provided a good foundation for pursuing a PhD. As a person who is persistent in the face of challenge, I wanted to get out of your comfort zone and challenge myself with new techniques in different disciplines; hence the MRC- DTP program was ideal for me.
2014 - Present: PhD, Warwick Medical School, UK
PhD Project Title:
"Analysis of clathrin and adaptor interactions"
Supervisors:
1) Dr. Corinne J. Smith (The University of Warwick, UK)
2) Prof. Frances Brodsky (University College London, UCL)
Funded by: Medical Research Council
Techniques: E.coli and insect cell (sf9) protein expression, affinity chromatogrphy purification, Electorn Microscopy (JEOL 2200FS 200KV)-K2 (DED camera) negative stain and cryoEM, manual plung freezing for cryoEM grids, spectrophotometry, flowrymeter, single-particle imaging (Relion and EMAN2), ITC, SPR, Western Blotting and Protein gel electorophoresis (Coomassie Blue), Clathrin purification from pig brains, SMALP purification of receptors from insect cell expressed pellets.
2013 - 2014: MSc, Interdisciplinary Biomedical Research, Warwick Medical School, UK
Mini project 1: "Expression and Purification of L1 protein from the Human Papilloma Virus type 11 and analysis of protein assembly mechanisms using Dynamic Light Scattering"
- Supervisor: Dr. Corinne J. Smith (Molecular and Cell Systems, Life Science) and Joseph Jones (MOAC PhD student)
- Skills acquired: Dynamic Light Scattering, Transmision Electron Microscopy, Protein expression and purification of E.coli.
Mini Project 2: "Investigating different signalling response profiles of triple dimerisation-mutant GLP-1R with wild type GLP-1R in yeart/mamalian chimeras"
- Supervisor: Dr. Graham Ladds (Biomedical Cell Biology)
- Skills acquired: Tissue Culturing, Yeast transformations, yeast GPCR signalling assays, data analysis (Prism 6.0).
Funded by: Medical Research Council
2010 - 2013: BSc (Hons), Biology (First Class), Kingston University London, UK
Final Year Research Dissertation: "The separation and analysis of laccase isozymes from Pleurotus ostreaus"
Overall PhD Research Interest
Background:
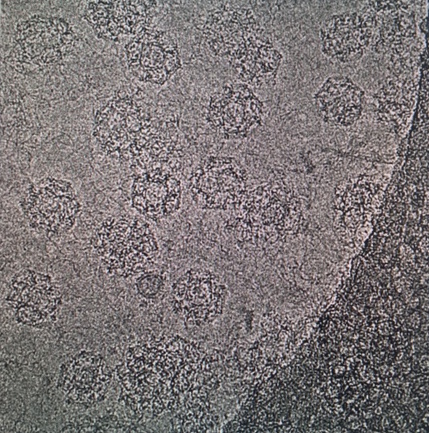
Clathrin-mediated endocytosis is essential for mammalian cells to function and underpins a range of processes which are important for human health and disease. These include nutrient uptake, synaptic vesicle recycling, signaling mechanisms and determination of cell polarity. Defects in clathrin-mediated endocytosis cause pathologies such as autosomal recessive hypercholesterolemia.
Clathrin forms multiple interactions with a diverse set of adaptor proteins during its assembly, to form a coat of protein around the newly developing endocytic vesicle. This coat engages with cargo on the cell surface and facilitates inclusion of the cargo into the vesicle. Once the vesicle has budded from the plasma membrane, the clathrin coat is dissembled, allowing the endocytic machinery to be recycled, and the vesicle goes on to deliver cargo to its destination within the cell. Different clathrin adaptor proteins have been found to be associated with cancer biology, such as HIP1 and auxilin.
Conferences, Workshops and Institutional visits
CONFERENCES & WORKSHOPS (Year 1):
- Visiting LMB in Cambridge for microscopy time on their Titon Krios EM (cryo-EM) 300kV: 7th-9th September 2015.
- Warwick Workshop on Molecular Recognition, University of Warwick: 10th-11th September, 2015: Abstract and Poster
- M5 networking conference: Structural Biology: 17th September, 2015, University of Leicester: Abstract and Poster
- 2014 ASCEPT Annual Scientific Meeting(Therapeutic agents and drug targets: GPCRs and beyond): 7-11 December 2014, Melbourne Convention Centre.
Poster: "Disruption of glucagon-like peptide 1 receptor (GLP-1R) dimerisation results in liraglutide sensitivity" (Mini Project 2 of MSc Interdisciplinary Biomedical Research)- presented by Kerry Barkan.
- MOAC DTC Annual Conference: 28th - 30th July 2014, Stratford Upon Avon, UK
Oral Presention: "Actin requirement in clathrin-mediated endocytosis in mitotic cells"
- Biomedical Cell Biology (BCB), WMS Postgraduate Symposium: 21st November 2014, University of Warwick, UK: Abstract and Poster
- British Biophysical Society Conference: 9-11th July 2014, University of Warwick,UK
- "Warwick Clathrin Club Meeting", University of Warwick, UK, Scarman House: 28th May 2015. (Organiser: Mary Halebian). Guest visit: Prof. Frances Brodsky and her group from UCL and Dr. Elizabeth Smythe and her group from The University of Sheffield. Oral Presentation
- "Warwick MRC Doctoral Training Partnership Symposium", Warwick Medical School, Biomedical Cell Biology Division UK, Scarman House: 12th June, 2015. Poster
- "Warwick Quanititative Biomedicine Symposium", Warwick Medical School, Biomedical Cell Biology Division UK, Scarman House: 29th May, 2015.
- EBSA: 10th European Biophysics Congress, Dresden, Germany: 18th-22nd July, 2015. Abstract and Poster
CONFERENCES & WORKSHOPS (Year 2):
- Re-visit LMB in Cambridge for microscopy time on their Titon Krios EM (cryo-EM) 300KV: 4th-6th November 2015.
- CryoMicroscopyGroup Meeting, University of Birmingham: 18th November, 2015
- Biomedical Cell Biology (BCB) Symposium of Warwick Medical School: 3rd-4th December, University of Warwick. Poster
- UK Membrane Trafficking Conference (LMCB, UCL): 21st December 2015, Brunei Gallery, SOAS, London, UK
- SMALP meeting 2016, Univeristy of Birmingham: 28th January 2016.
- Diamond Centre, Harwell, Oxford for microscopy time on there Titon Krios (cryo-EM) 300KV: 10th February 2016-12th February 2016. (via application)
- Molecular Mechanisms seminar (structural biology): SLS University of Warwick: 25th February 2016. Presented a talk: "Structural analysis of clathrin-Beta-arrestin 1 interaction"
- SLS Symposium, Warwick University,UK: 18th-19th April, 2016
- 2nd Annual Clathrin Club Meeting 2016: Friday, 3rd June 2016: Organiser: Mary Halebian. Groups attending: Steve Royle (Warwick), Corinne Smith (Warwick), Elyzabeth Smyth (Sheffield), Frances Brodsky (UCL).
- WMS PG Symposium: 7th June, Warwick Medical School, UK. Present Poster: "Structural analysis of clathrin-Beta-arrestin 1 interaction".
- Chimera workshop for Master (MSc) students: Demonstrator:Mary Halebian: 2-4pm on Monday,20th June 2016.
- Diamond Centre, Harwell, Oxford for microscopy time on there Titon Krios (cryo-EM) 300KV: 8th-12th July 2016. (via application
- MRC DTP Conference: 4-5th July at National Forest Youth Hostel, Swadlincote, Derbyshire. Present a poster and talk: " Structural analysis of clathrin-Beta-arrestin 1 interaction"
- Overcoming Cellular Barriers: Implications for Industrial Biotechnology: BioProNet, Birmingham Hilton Hotel, 6th-7th July 2016.
- ADVANCED DATA COLLECTION FOR HIGH RESOLUTION CRYOEM: HOW TO MAKE THE MOST OF YOUR NATIONAL FACILITY VISIT workshop, eBIC, Harwell, Diamond Light Source, 6th Septmber 2016.
- Annual Diamond Synchrotron Radiation User Meeting : 7th-8th September, 2016, at Harwell, Diamond Light Source, UK
- CCP3EM-Relion Workshop: University of Leeds: September 19th, 2016
CONFERENCES & WORKSHOPS (Year 3):
- MRC- DTP meeting: present POSTER, 27th September, 2016
- Seminar at University of Birmingham, 1-2pm on 14th November 2016 of on "Visualization of arrestin recruitment by a G-protein-coupled receptor" by AK Shukla.
- Molecular Mechanisms seminar (structural biology): SLS University of Warwick: 14th December 2016.
- UK Membrane Trafficking Conference (LMCB, UCL): 19th December 2016, Brunei Gallery, SOAS, London, UK
- CCPEM Symposium 2017: 24-26th April at Diamond Light Source, Harwell.
- 3rd Clathrin Club Meeting: 22nd May, presentation.
- WMS PG Symposium: 6th June 2017, 10 min presentation of my PhD project
- MRC DTP IBR Symposium: 4th July 2017, 15 min presentation of my PhD project
PUBLICATIONS, AWARDS and Memberships
PUBLICATIONS:
Abstract published in Eur Biophys J (2015) 44(Suppl 1): 43. Title: Single molecule imaging of clathrin-adaptor interactions focusing on a GPCR protein, β- arrestin. Abstract presented in the EBSA: 10th European Biophysics Congress in Dresden, Germany, July 2015.
HALEBIAN, M., MORRIS, K. & SMITH, C. 2017. Structure and Assembly of Clathrin Cages. In: HARRIS, R. & WRIGHT, J. (eds.) Subcellular Biochemistry. Switzerland: Springer. Link:http://link.springer.com/chapter/10.1007%2F978-3-319-46503-6_20
SMITH, S. M., BAKER, M., HALEBIAN, M. & SMITH, C. J. 2017. Weak Molecular Interactions Implicated in Clathrin-Mediated Endocytosis. In: WARWICK, U. O. (ed.). Frontiers (in press)
Kyle Morris, Joe Jones, Mary Halebian, Shenping Wu, Michael Baker, Jean-Paul Armache, Richard Sessions, Alex Cameron, Yifan Cheng, Corinne Smith, "Near-atomic resolution of assembled clathrin revealed by cryo-EM of multiple cage geometries" (to be published, 2017)
AWARDs:
- Medical Research Council, MRC, UK award for 4- years Interdisciplinary Biomedical Research MSc+PhD (2014-2017).
- EBSA Bursary Award (400 EUR) for the "EBSA: 10th European Biophysics Congress, Dresden, Germany: 18th-22nd July, 2015" conference registration and accomodation.
MEMBERSHIPs:
- Society of Biology
- Nature Cell Biology
- Royal Microscopy Society
- European Microscopy Society
- Course representative for BCB MRC DTP cohort 2013 for 3 years (2014-2017) - SSLC, Warwick Medical School.
- Supervising Undergraduate Final year student for her lab dissertation project in the Smith's Group (May-June 2016) and master student (MIBTP) (January-March 2017 and July-September 2017) (tbc).
- URSS student - Mentoring and lab supervison (June-August 2016)
Contact details: M.Halebian@warwick.ac.uk
Links: MRC Doctoral Training Partnership (cohort 2013), LinkedIn
Twitter: @maryhaleb

Warwick Medical School
Gibbet Hill Road
Coventry
CV4 7AL