Surface area and porosity analysis (BET)
Gas adsorption is used to measure surface areas and porosity of solids. These characteristics give information on the area of a solid's surface available for processes such as heterogeneous catalysis, the shape and size of pores in a material that may be used for shape selective catalysis, or the volume of gas a solid can contain (for example in CO2 capture).
The technique is suitable for non-porous materials (for surface area measurements) and porous materials ranging from the microporous (such as zeolites and MOFs) with pores < 20 Å, through to macropores of around 1000 Å
How does it work?
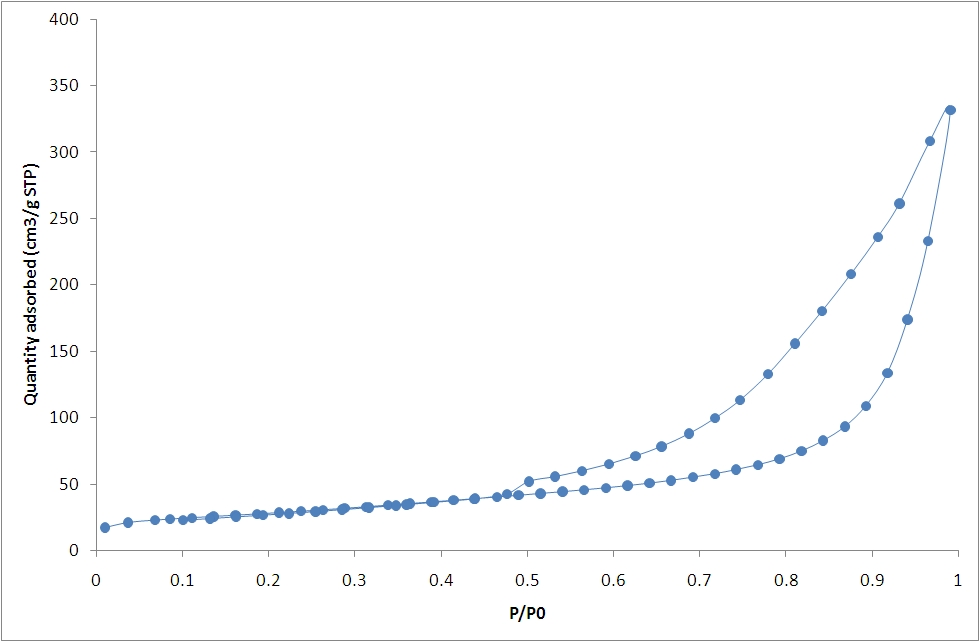
The sample is first degassed to remove surface contamination and adsorbed species using vacuum, heat and/or nitrogen. A probe gas (commonly nitrogen) is introduced at cryogenic temperatures; known aliquots are dosed up to saturation pressure, then a vacuum is applied and the gas removed, which may result in hysteresis behaviour. The relative pressure and quantity of gas adsorbed are measured to give an adsorption isotherm, which varies for different categories of material as shown:
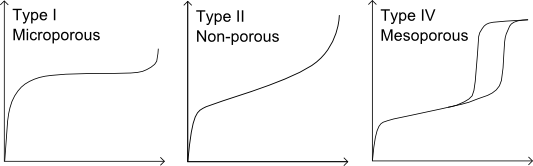
Surface area can then be calculated using the Langmuir or BET (Brunauer, Emmet and Teller) equations. A variety of techniques are available for calculation of pore volumes and size distributions, including BJH (Barrett, Joyner and Halenda), Horvath-Kowazoe and DFT (Density Functional Theory) methods.
Applications
Pharmaceuticals; Ceramics; Adsorbents; Catalysis; Paper; Medical Implants; Aeroscape; Fuel Cells; Geosciences; Filtration; Construction Material; Cement.
Sample handling requirements:
Around 1 g of material in solid or powder form.
Complementary techniques:
Particle size determination, Scanning electron microscopy, Transmission electron microscopy, X-ray diffraction
Warwick Capability:

Contact:
Claire Gerard: / 07385 145064
