Kinetochores
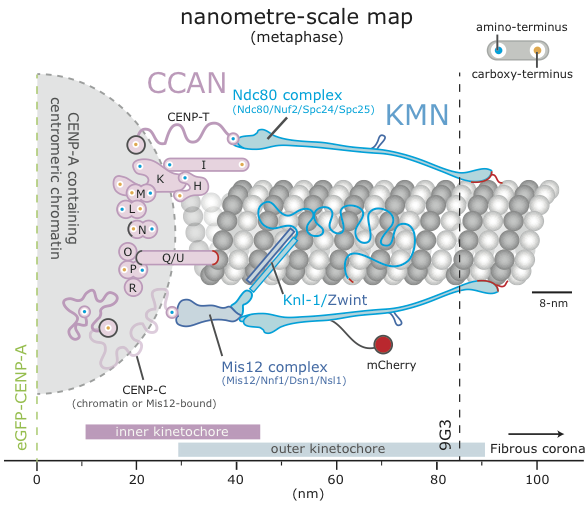
Kinetochores are another example of how evolution can produce something magnificent and efficient – accurate cell division would not be possible without both the cooperation and counteraction of the large number of proteins that form the kinetochore. Indeed, cancer along with many degenerative diseases is caused by a cell’s inability to divide accurately, and therefore it is an important current area of research. One such current model of the kinetochore is shown to the left (figure adapted from McAinsh 20141), representing only a handful of kinetochore proteins.
Kinetochores and Microtubules
As shown above, kinetochores bind to the end of microtubules, which can both grow and shrink in length. Kinetochores travel with these microtubule ends, and are responsible for the regulation of these growth and shrinkage states – this is important, as this means kinetochores are capable of generating forces to allow movement of DNA about the cell. Furthermore, it has been seen in marsupial cells that kinetochore length (defined along the axis of the microtubule) changes between these states, therefore kinetochore structure is important in this regulation.
Where My Interest Lies
It is widely believed that chromatin between sister kinetochores contains some form of ‘tension sensor’, which allows the kinetochore to regulate microtubule dynamics. However, I believe that their regulation abilities are intrinsic to the kinetochore (based on observations in our most recent publication2), and therefore conformational deformations in kinetochore-shape may well be caused by some sensor within the kinetochore, or vice versa.
My Day-to-Day Activities
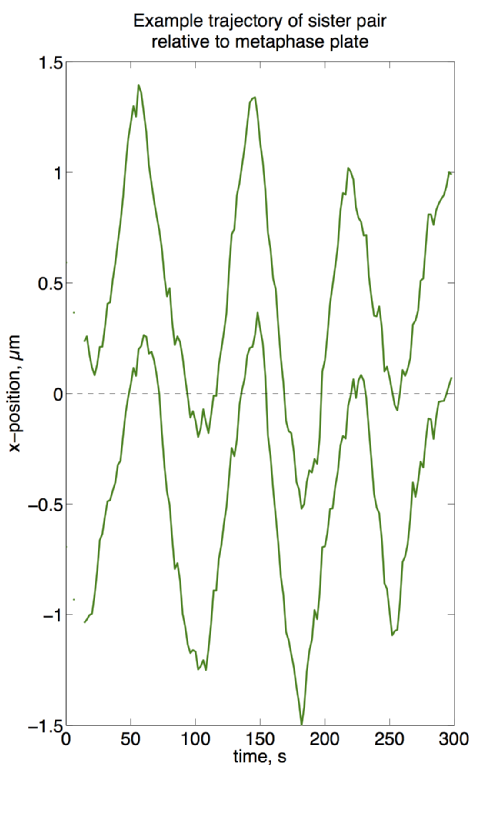
I investigate kinetochore location by tagging fluorescent proteins to kinetochore proteins in live cells, then observe them under a confocal spinning disc microscope (see top right, a human (HeLa) cell in mitosis – scale bar represents 2µm). Pairs of kinetochores are observed to exhibit back-and-forth motion3–7 (see bottom right), and it is this motion that demonstrates a kinetochore’s ability to direct chromosomes around the cell.
My Ultimate Goal
I have tagged two differently-coloured fluorescent proteins at either end of the kinetochore, and intend to investigate kinetochore length changes during this directed chromosome motion – I have learned some interesting preliminary behaviour thus far, and hope to demonstrate my findings in poster-form in Philadelphia, PA at the ASCB Meeting 2014 this December (on my birthday!).
References
- McAinsh 2014
- Vazquez-Novelle et al. 2014
- Skibbens et al. 1993
- Jaqaman et al. 2010
- Dumont et al. 2012
- Vladimirou et al. 2013
- Burroughs and McAinsh (personal communication)
Movie of green fluorescent protein (eGFP) linked to kinetochore protein CENP-A during mitosis.
Example trajectory of a pair of kinetochores, as measured from eGFP signal over 5 minutes every 2s.