William Woodruff
I completed my first degree in Physics at the University of Warwick in 2011. I particularly enjoyed the research and instrumentation side of my course and decided to pursue a Masters qualification by doing the AS:MIT MSc programme at the University of Warwick. The first half of the year was the taught course which was pretty tough but certainly rewarding. I particularly enjoyed the NMR module and as a result I've chosen to do my research project in electron paramagnetic resonance.
As a physicist who took a dive into chemistry I feel perfectly qualified to tell other soon to graduate physicists that this course is awesome and you'd fit right in (with a midge of elbow grease goes a long way). I'm now studying EPR and as a result I'm back working with physicists and I'm working near my AS:MIT colleagues which is nice. I've found the cross learning between chemistry and physics outstandingly beneficial in understanding the fundamentals of my research as well as helpful across the whole of the course.
AS:MIT Research Project: Dynamic nuclear polarization (DNP) using electron-nuclear double resonance
The idea of DNP is to polarise nuclei using electron polarisation. The advantage of using electron polarisation is that it is much greater than nuclei polarisation (Fig. 1). Thus, when the polarisation is transfered between electrons and nuclei the result is a stronger NMR signal. I am involved with setting up the new instrumentation to measure DNP improvements
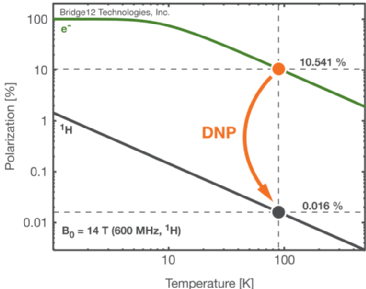
Figure 1. Temperature dependence of the electron and nuclear polarization.
which is currently being upgraded. The technology and DNP as an academic field is progressing quickly as its potential heralds a new paradigm of NMR sensitivity. I am also looking at the stability of radical 4-Hydroxy-TEMPO (Fig. 2) which is commonly used throughout analytical science and I am trying to determine how stable it really is. There isn't much infomation about to what extent TEMPOL is stable in scientific literature but common storage advice dictates it must be kept in the fridge. However, early analysis shows that TEMPOL is even stable at as high as 90°C. Which if confirmed would make cool storage unnecessary. I am using EPR to quantiatively analyse the radical which gives a 3 peak characteristic spectrum (Fig. 3) due to the electron spin 1/2 inteacting with the nitrogen spin 1 nuclei.
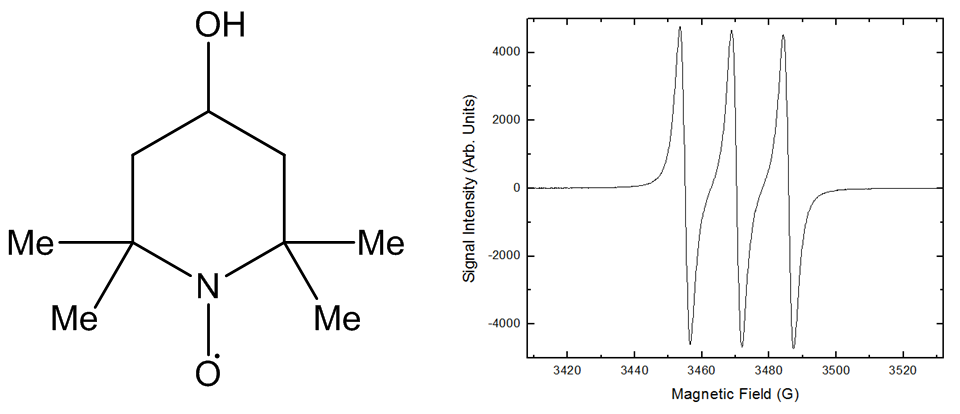 Figure 2 & 3. Chemical structure of TEMPOL with the characteristic spectrum caused by interaction of half spin electron and 1 spin nitrogen nuclei. The electron spin doesn't interact with oxgygen due to it's nuclei being spin 0.
Figure 2 & 3. Chemical structure of TEMPOL with the characteristic spectrum caused by interaction of half spin electron and 1 spin nitrogen nuclei. The electron spin doesn't interact with oxgygen due to it's nuclei being spin 0.

Contact
Email: wewoodruff@gmail.com
Career Aspirations
• Start the tribute band 'Slack Babbath'
• Professional Butterfly Impersonator
• PHD in Recovering from traumatic Masters
