I did my first degree in at the Manchester Metropolitan University. At least the first degree I actually finished... My 2nd degree is from the University of Tampere, Finland, where I studied at the Faculty of Business Administration and Economics. I Started there at 2003 but did not actually finish until 2009, just before beginning the AS:MIT course.
Title of my Project
Electroanalytical Investigation of the Properties of Single Crystal Boron Doped Diamond (SCBDD)
Abstract of my Project
Diamonds are known of their special properties. They are physically robust and chemically inert, have a high thermal conductivity and can act as an insulators. Adding an impurity, such as Boron, into the diamond lattice causes an extra valency into the lattice because Boron has only three valence electrons. These valencies can facilitate the movement of electrons through the diamond lattice and make diamond an electric conductor.
Adding Boron into the lattice has its drawbacks. Because Boron is slightly smaller atom than Carbon each Boron distorts the lattice slightly thus weakening the bonds throughout the crystal lattice. Also since one covalent bond is effectively missing it is easy to see that higher the Boron concentration in the lattice, better the electric conductivity of the diamond (due to holes) but worse the other diamond properties. It is therefore important to determine the concentration of Boron that gives an optimal (metallic) conductivity to the material but still retains all of the physical and chemical properties of diamond. This can lead to new improved applications in electroanalytical sensing.
1. Capillary measurements
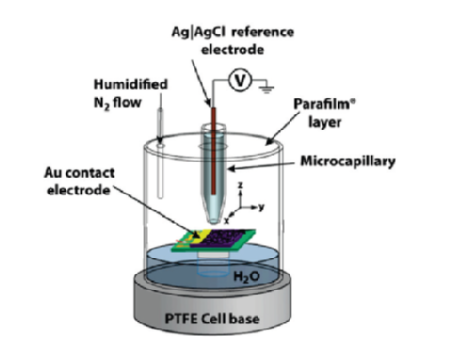
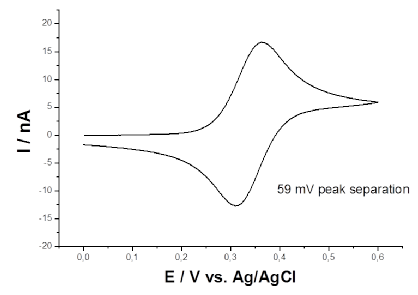
Capillary measurements are a fast way to probe the electrochemical behavior of a conducting material. A small (50 to 70 um in diameter) capillary filled with electrolyte is brought to near contact with the sample surface so that the meniscus at the capillary tip makes a contact with the sample. A schematic presentation of the system is shown in Image 1 [1]. The circuit is completed via a gold contact on the sample enabling the characterization of the conducting properties of the material. A cyclic voltammogram (Image 2) can be recorded with such a setting to investigate the electrochemical properties of the sample.
2. Intermittent Contact-Scanning Electrochemical Microscopy (IC-SECM):
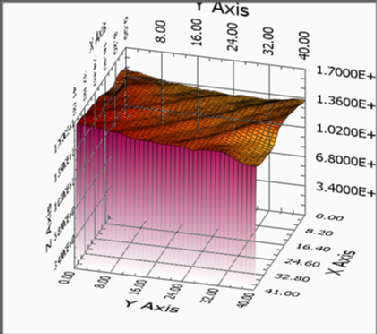
IC-SECM allows the electric conductivity and the surface topography to be determined simultaneously and independently [2]. At least in theory the surface of the SCBDD scould be completely flat without any features and electrochemically homogenous. As the capillary measurements are confined to the area of the capillary used (50-70 um) and do not give information of the topography IC-SECM is used to gain further information of the Boron doped diamond samples. Images 3 and 4 display the topography and electrical conductivity of gold bands recorded with 10 micrometer UME.

References
- J. Phys. Chem. C 2010, 114, 13241–13248
- Anal. Chem. 2010, 82, 6334–6337




