SMALP mini project
Project title: Novel Biophysical methods for Membrane Protein Research
Project suitable as: Wet Project Dry Project Also submitted to MOAC
Project timing: Slot 1 (22/03 to 11/06/2010) Slot 2 (14/06) maybe
Supervisor (the person who will be doing the day to day supervision of the mini-project):
Name: Dave Roper
Department:Biological Sciences Building, Room: Office B126 Lab C10
E-mail address: david.roper@warwick.ac.ukPhone number: Office B126 Lab C10
Second supervisor (if applicable):
Name:Alison Rodher
Department: Chemistry Building, Room: B607
E-mail address:a.rodger@warwick.ac.uk Phone number:74696
Project outline
A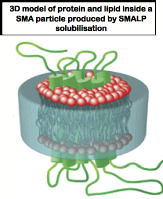 . Background to project:Cell membranes play a vital role in cell functioning, creating a protective layer between the interior of the cell and the external environment and providing a mechanism for the cell to detect what is happening in its surroundings and respond to any changes. Membrane proteins gather information about the cell's environment, and tell the cell what changes need to be made. This interaction between cells and the environment plays a vital role in biology. Membrane proteins perform a wide range of functions, including respiration, signal transduction and molecular transport. In addition, over 50% of commercially available drugs, such as antihistamines, beta blockers and morphine target membrane proteins. This makes them a vital area of study for researchers wanting to gain a better understanding of how these drugs interact with the human body, and thus look at ways to improve their functioning and reduce the possibility of harmful side effects. The study of membrane proteins is complicated by the fact that these proteins are generally difficult to isolate and study. Most purification techniques for membrane proteins require detergent extract providing a totally artificial environment for subsequent biophysical study that can affect or mask vital biological properties.
. Background to project:Cell membranes play a vital role in cell functioning, creating a protective layer between the interior of the cell and the external environment and providing a mechanism for the cell to detect what is happening in its surroundings and respond to any changes. Membrane proteins gather information about the cell's environment, and tell the cell what changes need to be made. This interaction between cells and the environment plays a vital role in biology. Membrane proteins perform a wide range of functions, including respiration, signal transduction and molecular transport. In addition, over 50% of commercially available drugs, such as antihistamines, beta blockers and morphine target membrane proteins. This makes them a vital area of study for researchers wanting to gain a better understanding of how these drugs interact with the human body, and thus look at ways to improve their functioning and reduce the possibility of harmful side effects. The study of membrane proteins is complicated by the fact that these proteins are generally difficult to isolate and study. Most purification techniques for membrane proteins require detergent extract providing a totally artificial environment for subsequent biophysical study that can affect or mask vital biological properties.
Recently a novel methodology using a Styrene Maleic Acid Lipid Particle (SMALPs) polymer that forms a nano-scale “cookie cutter” has been developed by colleagues at U. Birmingham that shows considerable promise for isolating membrane proteins intact, in their correct lipid environment 1.Rapid purification can then be achieved by affinity techniques like Immobilised Metal ion Affinity Chromatography (IMAC) when the target protein is labeled with a suitable poly-histidine tag. Due to the size of the particle formed by the SMALP around the membrane protein, some proteins (dependent on fold) are readily solublised but are not always suitable for IMAC purification as the histidine tag is encapsulated by the SMALP.
O bjectives:1. We now wish to adapt an existing Green Fluorescent Protein (GFP) fusion vector system for membrane proteins 2, 3with a more advanced histidine tag (see below) to enable both SMALP extraction and IMAC purification of all membrane systems. The first stage of this project will involve molecular biology techniques to modify the existing fusion vector with GFP containing a poly-histidine tag in a loop between structural b-strand. We will then go on to test this novel vector with a number of example membrane proteins including the bacterial cell division proteins FtsW 5, 6. Given that GFP fluorescence can be modified to other colours 7, 8, this system could be easily adapted to study multi-membrane protein interactions via in-situand in-vivomicroscopy techniques. In the first instance we are particularly interested in adapting this system to the study of bacterial cell division proteins providing for the first time, highly purified membrane protein samples for biophysical study.
bjectives:1. We now wish to adapt an existing Green Fluorescent Protein (GFP) fusion vector system for membrane proteins 2, 3with a more advanced histidine tag (see below) to enable both SMALP extraction and IMAC purification of all membrane systems. The first stage of this project will involve molecular biology techniques to modify the existing fusion vector with GFP containing a poly-histidine tag in a loop between structural b-strand. We will then go on to test this novel vector with a number of example membrane proteins including the bacterial cell division proteins FtsW 5, 6. Given that GFP fluorescence can be modified to other colours 7, 8, this system could be easily adapted to study multi-membrane protein interactions via in-situand in-vivomicroscopy techniques. In the first instance we are particularly interested in adapting this system to the study of bacterial cell division proteins providing for the first time, highly purified membrane protein samples for biophysical study.
2. The fusion proteins prepared in objective 1 will form the basis of a biophysical study of FtsW with membrane mimicking systems made up of different lipids in objective 2. In particular we will probe whether the FtsW binds to the lipid membrane in a preferred orientation using linear dichroism spectroscopy, explore its kinetics of insertion when added to membrane systems using circular and linear dichroism and fluorescence spectroscopy, see whether we can view its localization on membrane surfaces using confocal microscopy. If this phase of the project proceeds well then we will look to assemble FtsZ on the inside of liposomes and probe its interaction with other proteins involved in the formation of the Z-ring.
B. Skills to be learned
General molecular biology skills including PCR, Cloning, mutagenesis, protein expression and membrane protein isolation using SMALPs; fluorescence, circular dichroism, linear dichroism, dynamic light scattering, confocal microscopy
C. Systems biology context
This project is largely about developing tools for studying biological systems. Objective 2 involves testing the tool developed in objective 1 on a very simple system. If successful this will then be used in the wider study of the bacterial cell division system.
C. Resources required@£450 contribution to consumables.
D. References
These fell of the required 1 page and may be found at
1.Knowles, T. J., Finka, R., Smith, C., Lin, Y. P., Dafforn, T., and Overduin, M. (2009) Membrane proteins solubilized intact in lipid containing nanoparticles bounded by styrene maleic acid copolymer, J Am Chem Soc131, 7484-7485. 2.Drew, D., Lerch, M., Kunji, E., Slotboom, D. J., and de Gier, J. W. (2006) Optimization of membrane protein overexpression and purification using GFP fusions, Nat Methods3, 303-313. 3.Drew, D., Slotboom, D. J., Friso, G., Reda, T., Genevaux, P., Rapp, M., Meindl-Beinker, N. M., Lambert, W., Lerch, M., Daley, D. O., Van Wijk, K. J., Hirst, J., Kunji, E., and De Gier, J. W. (2005) A scalable, GFP-based pipeline for membrane protein overexpression screening and purification, Protein Sci14, 2011-2017. 4.Kobayashi, T., Morone, N., Kashiyama, T., Oyamada, H., Kurebayashi, N., and Murayama, T. (2008) Engineering a novel multifunctional green fluorescent protein tag for a wide variety of protein research, PLoS One3, e3822. 5.Errington, J., Daniel, R. A., and Scheffers, D. J. (2003) Cytokinesis in bacteria, Microbiol Mol Biol Rev67, 52-65, table of contents. 6.Margolin, W. (2001) Spatial regulation of cytokinesis in bacteria, Curr Opin Microbiol4, 647-652. 7.Rizzuto, R., Brini, M., De Giorgi, F., Rossi, R., Heim, R., Tsien, R. Y., and Pozzan, T. (1996) Double labelling of subcellular structures with organelle-targeted GFP mutants in vivo, Curr Biol6, 183-188. 8 Service, R. F. (2008) Nobel Prize in chemistry. Three scientists bask in prize's fluorescent glow, Science322, 361. 9.Rodger, A.; Rajendra, J.; Marrington, R.; Ardhammar, M.; Nordén, B.; Hirst, J. D.; Gilbert, A. T. B.; Dafforn, T. R.; Halsall, D. J.; Woolhead, C. A.; Robinson, C.; Pinheiro, T. J.; Kazlauskaite, J.; Seymour, M.; Perez, N.; Hannon, M. J., Flow oriented linear dichroism to probe protein orientation in membrane environments. Phys. Chem. Chem. Phys. 2002,4, 4051-4057. 10.Rodger, A.; Marrington, R.; Geeves, M. A.; Hicks, M.; de Alwis, L.; Halsall, D. J.; Dafforn, T. R., Looking at long molecules in solution: what happens when they are subjected to Couette flow? Physical Chemistry Chemical Physics 2006,8, (27), 3161-3171. 11.Rajendra, J.; Damianoglou, A.; Hicks, M.; Booth, P.; Rodger, P. M.; Rodger, A., Quantitation of protein orientation in flow-oriented unilamellar liposomes by linear dichroism. Chemical Physics 2006,326, (1), 210-220. 12.Hicks, M. R.; Damianoglou, A.; Rodger, A.; Dafforn, T. R., Folding and membrane insertion of the pore-forming peptide gramicidin occur as a concerted process. Journal of molecular biology 2008,383, (2), 358-66.13. Hicks, M. R.; Dafforn, T.; Damianoglou, A.; Wormell, P.; Rodger, A.; Hoffmann, S. V., Synchrotron radiation linear dichroism spectroscopy of the antibiotic peptide gramicidin in lipid membranes. The Analyst 2009,Accepted - awaiting publication.
