Reseach
Research
With the increase in demand for energy worldwide and a diminishing supply of fossil fuels, we need to find as many types of renewable energy as possible. Currently we still produce over 75 % of our electricity in the UK from fossil fuels, with only 7 % produced by renewable energy. Of this 7 %, the majority is produced by wind and thermal sources, with solar only accounting for 0.1 % of the electricity generation.
If we harnessed only 0.01 % of the energy that hits earths surface we could meet all our energy requirements. However current silicon solar cells cost a great deal of money and energy to produce, so an alternative, at least initially for niche applications, could be organic solar cells. These have the advantages of far lower processing costs, they are far thinner (~200 nm compared to hundreds of micrometres for silicon cells) and can be mounted on flexible substrates. They also have a shorter payback period, ie the length of operating time required to repay the energy used to fabricate the cells, compared to silicon cells.
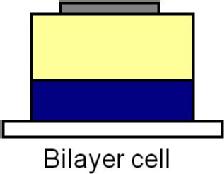
An organic solar cell typically contains a front transparent electrode, a back reflective electrode and a bilayer of organic materials sandwiched between the electrodes, shown in the bilayer cell below. One of the organic materials acts as a donor material and one as an acceptor of electrons (from the donor).
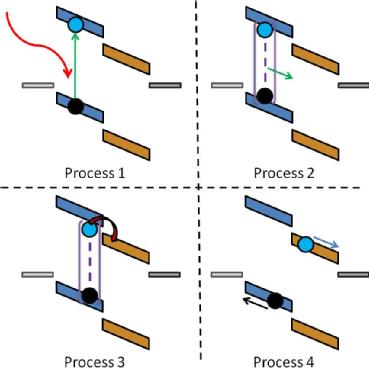
Organic solar cells work in a slightly different manner to the traditional silicon cells. In silicon cells the light is absorbed to excite an electron, leaving behind a hole, with the charges separated easily. In organic materials, after excitation (process 1) the charges are bound together to form a hole-electron pair, known as an exciton. As it is a neutral species the exciton cannot feel the built in field of the cell and so diffuses randomly (process 2) until it reaches a site where it is favourable to separate the charges (process 3). The separated charges can then be transported under the cells built in field to the respective electrodes (process 4).
The video link below shows how we make and test our cells!
How to make a organic solar cell video!
My research
In my work I have shown how the insertion of just 5 nm of thermally evaporated molybdenum oxide into cells, between the ITO and donor material, can improve both the cells performance and the cell stability. This is reported initially for ClAlPc in the first paper listed in my publications section, and then also for other systems with more detail in the second paper. The fifth paper also shows how vanadium oxide can be utilised in the same manner, and in addition has n-type character and is not stoichiometric V2O5 when thermally evaporated.
More content coming soon!
