My Research
My current research as an Early Career Fellow built upon the work I carried out as a PhD student in the O'Reilly group in the Department of Chemistry. This work fits into the area of Polymer and Materials chemistry, and has involved collaboration with Chemists, Biologists and Materials Scientists.
in the Department of Chemistry. This work fits into the area of Polymer and Materials chemistry, and has involved collaboration with Chemists, Biologists and Materials Scientists.
Polymer functionalisation and fluorescent labelling
The precise modification of macromolecules is synthetically demanding, and therefore highly efficient reactions (such as 'click' reactions) are required for polymer functionalisation. State-of-the-art polymer synthesis by living or 'controlled' polymerisation mechanisms allows for both spatial control (chain-end versus backbone functionalisation), as well as temporal control (pre-polymerisation or post-polymerisation) over the incorporation of functionality.
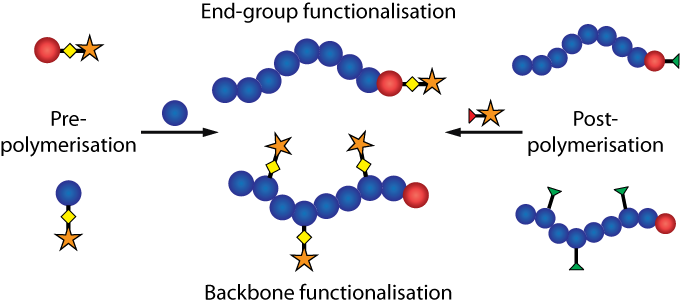
One motivation for developing these efficient chemistries is the desire to incorporate a fluorescent 'label' into macromolecular architectures. This is the result of fluorescently labelled polymers finding a myriad of applications in organic electronic devices, as sensor materials, for studying the physical properties of polymers, and for labelling polymer materials (nanoparticles, hydrogels, membranes etc.) for fluorescence
detection/imaging in biomedical applications.
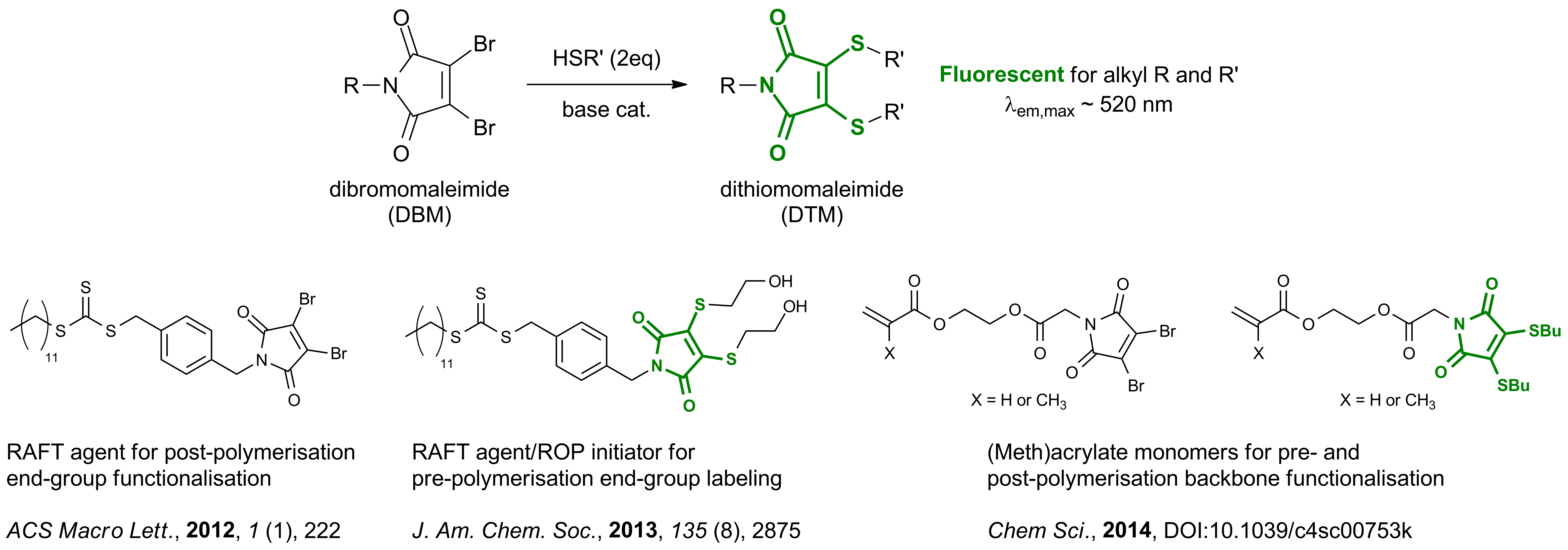
My research has focussed on investigating the properties of a newly discovered fluorophore and using this for polymer fluorescent labelling. The dithiomaleimide (DTM) fluorophore has been incorporated into polymer structures as end-groups or along the backbone. The DTM group is formed by a highly efficient reaction between dibromomaleimide (DBM) and thiols (initially reported by Baker and Caddick
and Caddick at UCL), allowing DTM incorporation by both pre- and post-polymerisation functionalisation.
at UCL), allowing DTM incorporation by both pre- and post-polymerisation functionalisation.
Fluorescent polymer nanoparticles
The ability to incorporate the dithiomaleimide (DTM) fluorophore into synthetic polymers allowed us fabricate fluorescently labelled polymer nanoparticles, by the self-assembly of DTM functional amphiphilic polymers. When incorporated into a nanoparticle the DTM fluorophore shows a disinclination to self-quench, leading to concentration independent molar emission and long average fluorescent lifetimes (~20nm).
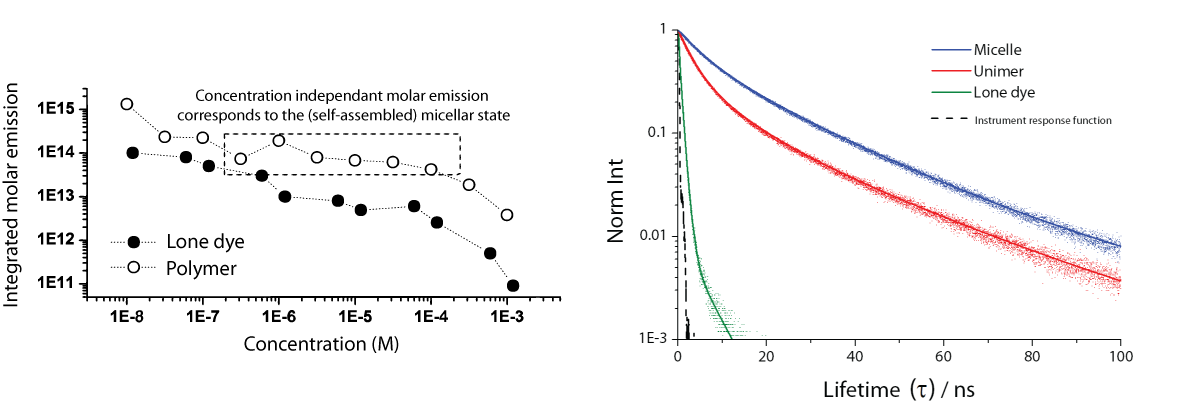
These fluorescent nanoparticles were demonstrated as cellular imaging agents by fluorescence lifetime microscopy (FLIM). One great advantage for this system is that fluorescent lifetime is significantly different in the micelle (self-assembled), polymer unimer (disassembled) and lone dye (degraded) states. This allows differentiation between these macromolecular states within different areas of the tissue by FLIM.

Relevant publications
Hollow polymer nanoparticles
The synthesis of hollow nanoparticles is of interest due to their potential use as nanoreactors, nanocontainers, purification agents, delivery vehicles, and nanofluidic materials. The synthesis of hollow nanoparticles often requires multistep fabrication strategies involving the construction of solid particles and subsequent core removal, or templating methodologies.
Using advanced nanoparticle characterisation techniques (stain-free TEM imaging using graphene oxide substrates) we discovered the spontaneous formation of hollow nanoparticles upon drying poly(acrylic acid)-b-poly(lactide) block copolymer micelles to a surface. Both hollow cylindrical and spherical nanoparticles could be produced using this approach, and particles could be harvested from the surface to provide solutions of these hollow nanoparticles.
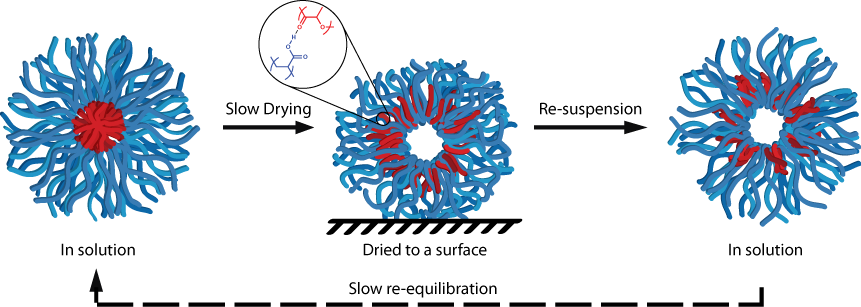
We propose that the mechanism for the spontaneous formation of hollow particles results from hydrogen bonding between the core and corona upon dehydration, leading to the possibility for expanding this approach to many other block copolymer self-assemblies.
Supervisor
Prof. Rachel K. O'Reilly
Department of Chemistry, University of Warwick
Major Collaborators
Dr Jeffery E. Raymond
Department of Chemistry and Laboratory for Synthetic-Biologic Interactions, Texas A&M University
Prof. Christophe Chassenieux and Dr Olivier Colombani
Institut des Molécules et Matériaux, Université du Maine
Prof. Thomas H. Epps, III and Elizabeth G Kelley
Department of Chemical and Biomolecular Engineering, University of Delaware
Prof. Andrew P. Dove and Prof. David M. Haddleton
Department of Chemistry, University of Warwick
