My Research
Micelle ProductionUsing known methods self assembled structures were formed from deprotected butyl acrylate and hydrophobic amino acids (phenylalanine and tryptophan). As amino acid are readily avaiable in one chiral form they are a good choice for the starting chirality. |
 |
Separation of Enatiomers
This has been tested on the chiral monomers, polymers and micellar structures. The overall result will hopefully be to sepearate out enatiomers based on a preferred interaction with the 'pure' chirality on the polymer side chain. [1]
Diels Alder
The Diels Alder reaction is a powerful reaction creating carbon-carbon bonds to form cyclohexene rings. Therefore, as an important reaction, much work has been done on trying to improve the endo:exo ratios observed and ee%, which this work tries to improve on. This reaction is being used to demonstrate catalysis within the chiral nanostruatures
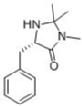
Incorporation of MacMillan CatalystThe MacMillan catalyst is a powerful catalyst for the Diels Alder reaction, being able to produce high ee%. [2] The catalyst is an organic catalyst derived from phenylalanine. This works tries to develop a supported MacMillan catalyst.
|
 |
Copper Catalyst
Cu 2+ has been used to catlyse the diels alder reaction. Ferringa et al. [3] demonstrated that by placing the copper in a chiral environment this would have an impact on the chirality of the resulting product. We hope that by placing the copper in the chiral environment of the micelles, we will also be able to affect the chirality of the product; importanly it will also be a system which can be removed and reused.
[1] Wang, J.; Zhu, X.; Cheng, Z.; Zhang, Z.; Zhu, J. J. Polym. Sci., Part A: Polym. Chem. 2007, 45, 3788
[2] Ahrendt, K. A.; Borths, C. J.; MacMillan, D. W. C. J. Am. Chem. Soc. 2000, 122, 4243
[3] Boersma, A. J.; Klijn, J. E.; Feringa, B. L.; Roelfes, G. J. Am. Chem. Soc. 2008, 130, 11783.
