World
 Although much of the research within the Zeeman Institute has focused on the pandemic dynamics in the UK, the methods have global application.
Although much of the research within the Zeeman Institute has focused on the pandemic dynamics in the UK, the methods have global application.
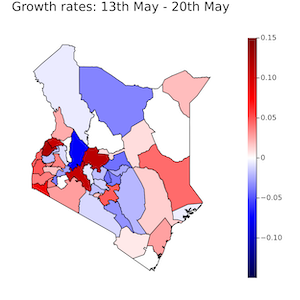
Most notably, there is a strong link between the Zeeman Institute and researchers at the KEMRI-Wellcome Trust Centre in Kenya. In collaboration with statisticians, modellers and bioinformaticians at the Kemri-Wellcome Trust Research Programme (KWTRP) in Kenya, we have been amongst the first to apply sophisticated inference and model projection to national data from tropical sub-Saharan Africa. As the availability of COVID-19 data, and the public health situation, in Kenya has evolved we have continuously updated our modelling techniques.
Our current focus is on projecting different scenarios of COVID-19 vaccine roll-outs in Kenya lead by John OjalLink opens in a new window. Additionally, current work in progress for Kenya includes spatially highly resolved stochastic simulations (lead by Andrea Parisi; see below) and explicit strain competition models (lead by Rabia Aziza).
Below is a selection of modelling work that was aimed at nowcasting/forecasting the ongoing pandemic emergency in Kenya and around the world.
| Sam Moore, Edward M. Hill, Louise Dyson, Michael J. Tildesley, Matt J. Keeling (2021) The impacts of increased global vaccine sharing on the COVID-19 pandemic; a retrospective modelling study medRxiv DOI: 10.1101/2022.01.26.22269877 |
We use an age-structured model of SARS-CoV-2 dynamics, matched to national data from 152 countries, to investigate the global impact of different vaccine sharing protocols during 2021. We assume a direct relationship between the emergence of variants with increased transmissibility and the cumulative amount of global infection, such that lower global prevalence leads to a lower reproductive number within each country. We compare five vaccine sharing scenarios, from the current situation, through sharing once a particular within-country threshold is reached (e.g. all over 40s have received 2 doses), to full sharing where all countries achieve equal age-dependent vaccine deployment.
Compared to the observed distribution of vaccine uptake, we estimate full vaccine sharing would have generated a 1.5% (PI -0.1 - 4.5%) reduction in infections and a 11.3% (PI 0.6 - 23.2%) reduction in mortality globally by January 2022. The greatest benefit of vaccine sharing would have been experienced by low and middle income countries, who see an average 5.2% (PI 2.5% - 10.4%) infection reduction and 26.8% (PI 24.1% - 31.3%) mortality reduction. Many high income countries, that have had high vaccine uptake (most notably Canada, Chile, UK and USA), suffer increased infections and mortality under most of the sharing protocols investigated, assuming no other counter measures had been taken. However, if reductions in vaccine supply in these countries had been offset by prolonged use of non-pharmaceutical intervention measures, we predict far greater reductions in global infection and mortality of 64.5% (PI 62.6% - 65.4%) and 62.8% (PI 44.0% - 76.3%), respectively.
| Samuel P.C. Brand, John Ojal, Rabia Aziza, Vincent Were, Emelda A. Okiro, Ivy K. Kombe, Caroline Mburu, Morris Ogero, Ambrose Agweyu, Edwine Barasa et al. (2021) "COVID-19 Transmission Dynamics Underlying Epidemic Waves in Kenya" Science DOI: 10.1126/science.abk0414 |
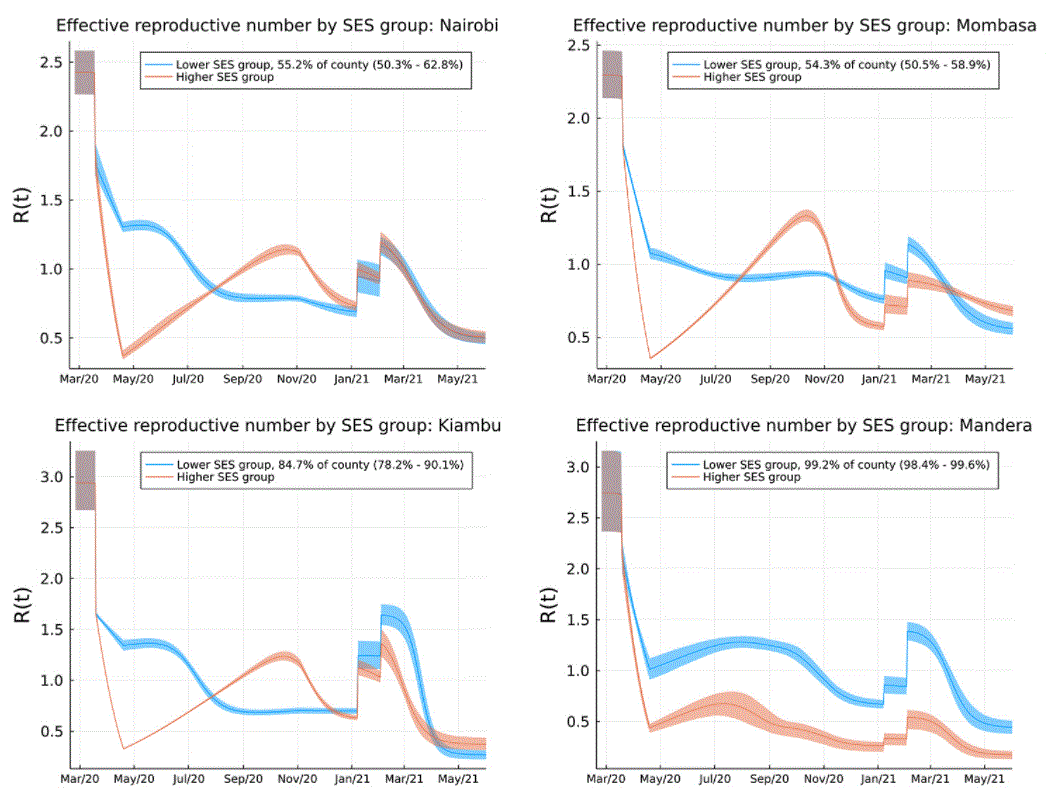 It has been recognised around the globe that the socio-economic status (SES) of individuals is a critical determining factor in their risk of SARS-CoV-2 infection, and developing severe COVID-19 disease. In this study, we integrate data from Google mobility data, longitudinal serological surveys and the laboratory PCR data into a mathematical model aimed at investigating the evidence for differential SES risk in Kenya.
It has been recognised around the globe that the socio-economic status (SES) of individuals is a critical determining factor in their risk of SARS-CoV-2 infection, and developing severe COVID-19 disease. In this study, we integrate data from Google mobility data, longitudinal serological surveys and the laboratory PCR data into a mathematical model aimed at investigating the evidence for differential SES risk in Kenya.
We found evidence that in urbanised regions of Kenya, characterised by high-density informal settlement, the risk of infection during the first wave in Kenya was extremely high among people in lower SES groups with very low ascertainment of infection by PCR testing. Subsequent waves in Kenya are explained by the delayed infection of people among higher SES groups who could maintain higher levels of social distancing for longer, and the introduction of highly transmissible variants of concern: Alpha, Beta and, more recently, Delta variants.
| John Ojal, Samuel P.C. Brand, Vincent Were et al. (2021) "Revealing the extent of the first wave of the COVID-19 pandemic in Kenya based on serological and PCR-test data" Wellcome Open Res 6:127 https://doi.org/10.12688/wellcomeopenres.16748.1 |
| Brand, S. P., Aziza, R., Kombe, I. K., Agoti, C. N., Hilton, J., Rock, K. S., Parisi, A., Nokes, D.J., Keeling, M., and Barasa, E. (2020). "Forecasting the scale of the COVID-19 epidemic in Kenya" (medRxiv) https://doi.org/10.1101/2020.04.09.20059865 |

Ahead of the first determined cases of COVID-19 in Kenya (March 2020) we developed a spatial- and age-structure stochastic model of SARS-CoV-2 transmission in Kenya. Epidemiological characteristics of SARS-CoV-2 were drawn from the early literature from the epidemic in Hubei province, China.
We found that, if within-Kenya transmission becomes established, it would be crucial to identify the role of asymptomatically infected individuals. If infections were overwhelmingly caused by symptomatic cases, then there was a reasonable possibility of early containment of the epidemic in Kenya. However, if a substantial proportion of infections originated from infected individuals presenting sub-clinical symptoms then we predicted that the subsequent epidemic could not likely be contained by case isolation alone, with the potential for a large proportion of the population becoming infected.
| Parisi, A., Brand, S.P.C., Hilton, J., Aziza, R., Keeling, M.J. & Nokes, D.J. (2021) "Spatially resolved simulations of the spread of COVID-19 in three European countries", PLoS Comp. Biol. 17(7): e1009090. https://doi.org/10.1371/journal.pcbi.1009090 |

In response to the pandemic, many governments issued measures aimed at containing the spatial spread of the virus and limiting the expected number of deaths. In this study, our goal was to use highly spatially resolved (5x5 km grid) stochastic transmission modelling calibrated to the observed spatial and temporal spread of the virus in Italy, Spain and the United Kingdom. We used a description of the population subdivided in age groups, high resolution population maps, and household structures: this allowed us to take into account the measures imposed by governments and quantify their effectiveness.
| Hilton, J., and Keeling, M. J. (2020). " Estimation of country-level basic reproductive ratios for novel Coronavirus (COVID-19) using synthetic contact matrices" PLoS Comp. Biol. 16(7): e1008031. https://doi.org/10.1371/journal.pcbi.1008031 |
The value of the basic reproductive ratio, R0, changes both according to the epidemiological characteristics of the virus, but also the context in which it spreads. Since susceptibility and transmission of COVID-19 changes with the age of the individual, the age-structure of the population can have a big effect on the value of R0. This work examines the risk posed by COVID-19 in different areas of the world. For some parameter choices (see below), the large proportion of children in Africa effectively buffer transmission leading to a much lower R0; notably under these parameters Italy is an obvious hot-spot while Germany has the lowest transmission in Europe.

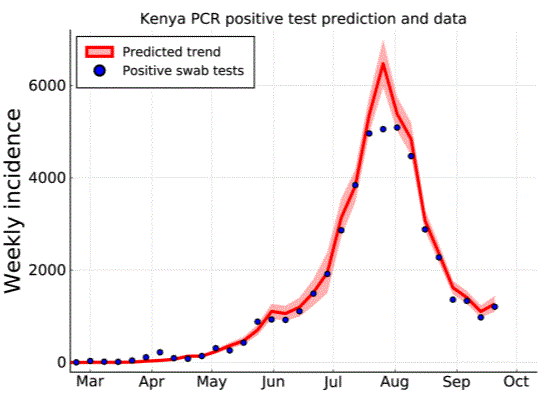 This paper uses analysis of national surveillance PCR test, serological survey and mobility data to develop and fit a simplified transmission model for Kenya aimed at understanding the first wave of COVID-19 in Kenya.
This paper uses analysis of national surveillance PCR test, serological survey and mobility data to develop and fit a simplified transmission model for Kenya aimed at understanding the first wave of COVID-19 in Kenya.