Democratic Republic of Congo (DRC)/ République démocratique du Congo (RDC)
Will Cryptic Infections Impact the Elimination Goal?
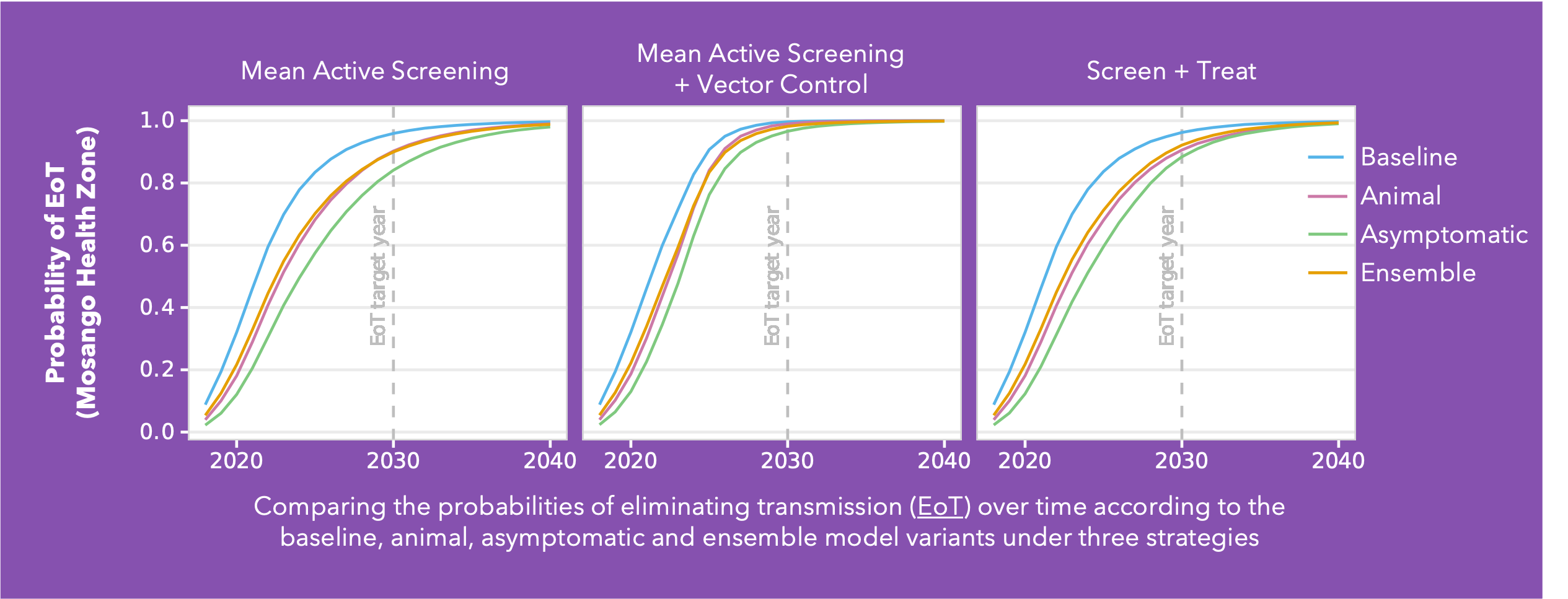
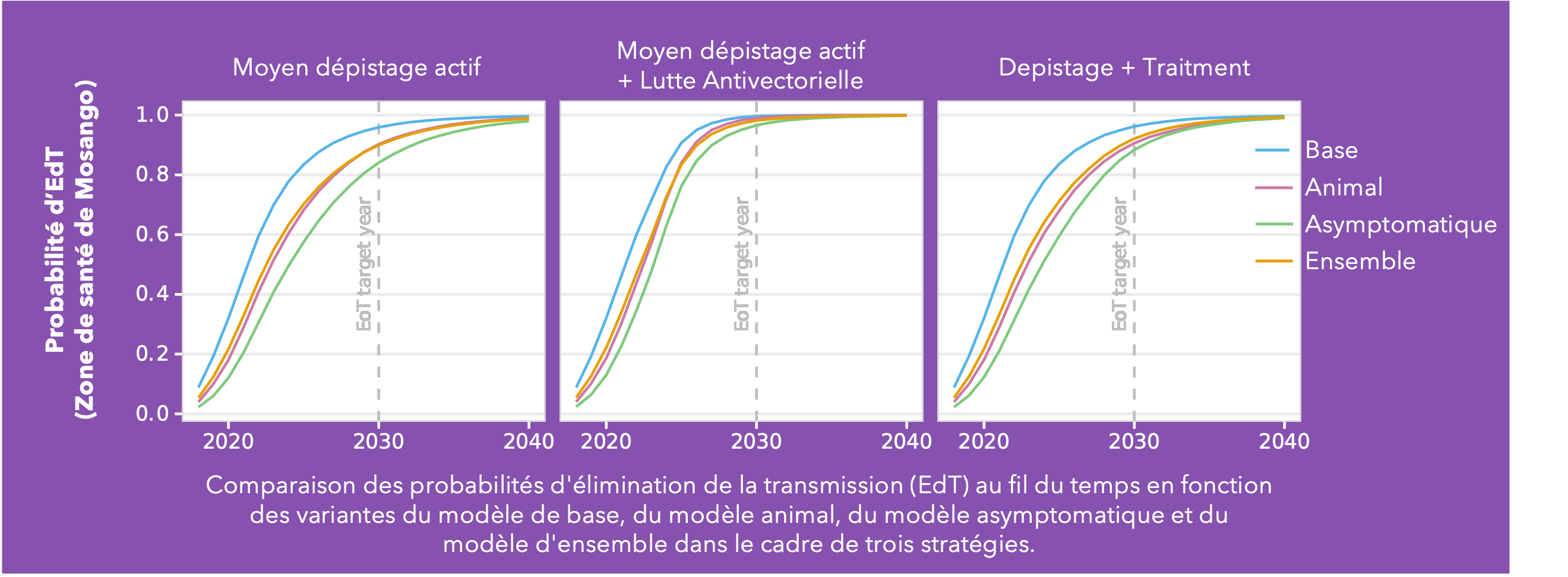
Conducted across five health zones in the DRC and spanning a broad range of prevalences, Ron’s analysis delves into the much-debated topic of cryptic infections and their potential impact on eliminating transmission (EoT). Building on the baseline Warwick gHAT model (blue line in the figure) two additional model variants were added: the animal transmission model – allowing for the potential role of animals as carriers and transmitters of infection (red line in figure) - and the asymptomatic transmission model – allowing for the presence of asymptomatic human infections, their detectability in blood tests, and their capacity to self-cure without intervention (green line in figure). By gathering statistical evidence for these model variants in each health zone, it is possible to infer which has the greatest statistical support. This ensemble of model variants (orange line in figure) was used to assess the probability of EoT across the health zones under specific strategies.
As anticipated, the baseline model is the most optimistic regarding the probability of achieving EoT. Furthermore, simulations under a Screen + Treat strategy highlight the potential benefit in reducing transmission by removing the parasitological confirmation step before treatment, particularly in the asymptomatic model variant. The results also highlight the advantages of vector control measures specifically in the presence of cryptic infections. However, whilst vector control is a valuable tool, implementing such a resource-intensive intervention necessitates careful prioritisation.
This analysis indicates we can be cautiously optimistic and that despite emerging evidence of trypanosomes in the skin without detectable blood parasites, their contribution to transmission dynamics appears limited in the DRC. Nevertheless, we acknowledge the potential for asymptomatic transmission, albeit with a relatively minor impact on our elimination targets. As we refine our understanding of gHAT transmission dynamics, these insights will serve as invaluable guides for developing targeted interventions.
Crump RE, Aliee M , Sutherland SA, Huang C, Crowley EH, Spencer SEF, Keeling MJ, Shampa C, Miaka EM and Rock KS Modelling timelines to elimination of sleeping sickness in the DRC accounting for possible cryptic human and animal transmission Link opens in a new window Parasites & Vectors Paper summaries: English Link opens in a new windowFrenchLink opens in a new window
The Cost of Elimination for the Democratic Republic of the Congo
In collaboration with the DRC sleeping sickness control program and their partners, this study evaluated several potential intervention strategies at the health-zone level, focusing on cost-effectiveness in terms of disease burden (DALYsLink opens in a new window) and enhancing the likelihood of eliminating transmission (EoTLink opens in a new window) by 2030.
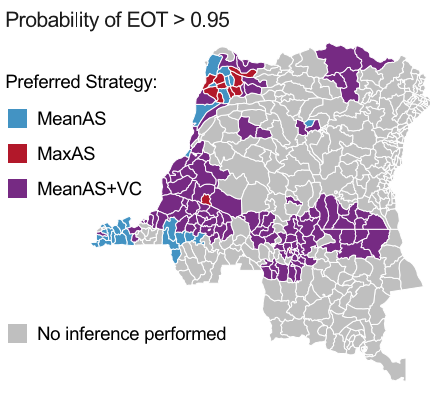
The findings indicate that Kongo Central and Équateur Nord coordinations have a promising outlook under current (Status Quo) strategies with others requiring adjustments to optimise outcomes. A total of 119 health zones are highlighted as requiring a shift in strategies to boost the likelihood of EoT by 2030. Among these, 66 face the additional challenge that even the strategy maximising EoT falls short of a 90% probability by 2030 as indicated by hatched health zones in the map opposite.
Based on existing strategies (Status Quo), 118 out of 165 health zones are on track to achieve EoT by 2030. This endeavour comes with a projected cost of $67.5M and an accumulation of an estimated 175K DALYsLink opens in a new window or 7979 deaths over a 16-year time horizon. Introducing new interventions aimed at reaching EoT with greater than 90% probability, we anticipate that 16 more health zones could reach EoT requiring $118M of additional investment. This move could prevent 114K DALYsLink opens in a new window or 5403 deaths. Despite this being a relatively small investment, it's crucial to highlight that nearly 15% of this ($18.3M) would be required in the first year of switching strategies and would require a huge operational undertaking.
Explore all the results of the Whole DRC cost-effectiveness analysis in our graphical user interface.
Antillon M, Huang C, Sutherland SA, Crump RE, Brown PE, Bessell PR, Crowley EH, Snijders R, Hope A, Tirados I, Dunkley S, Verlé P, Lebuki J, Shampa C, Miaka EM, Tediosi F, Rock KS (2026) Cost-effectiveness of end-game strategies against sleeping sickness across the Democratic Republic of CongoLink opens in a new window MedRXiV (not yet peer-reviewed) Paper summaries: EnglishLink opens in a new window FrenchLink opens in a new window
From health zones to health areas in the DRC
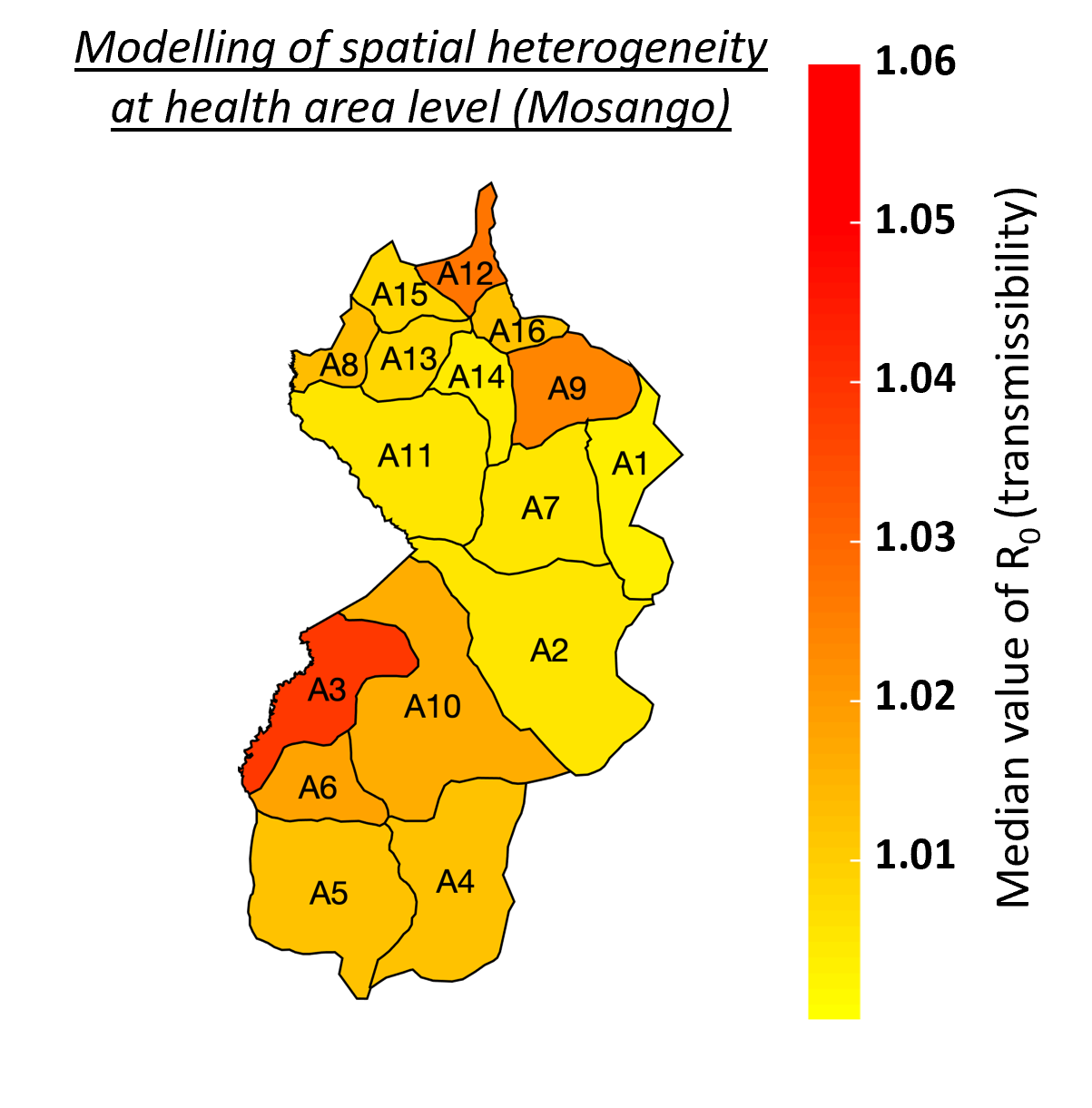
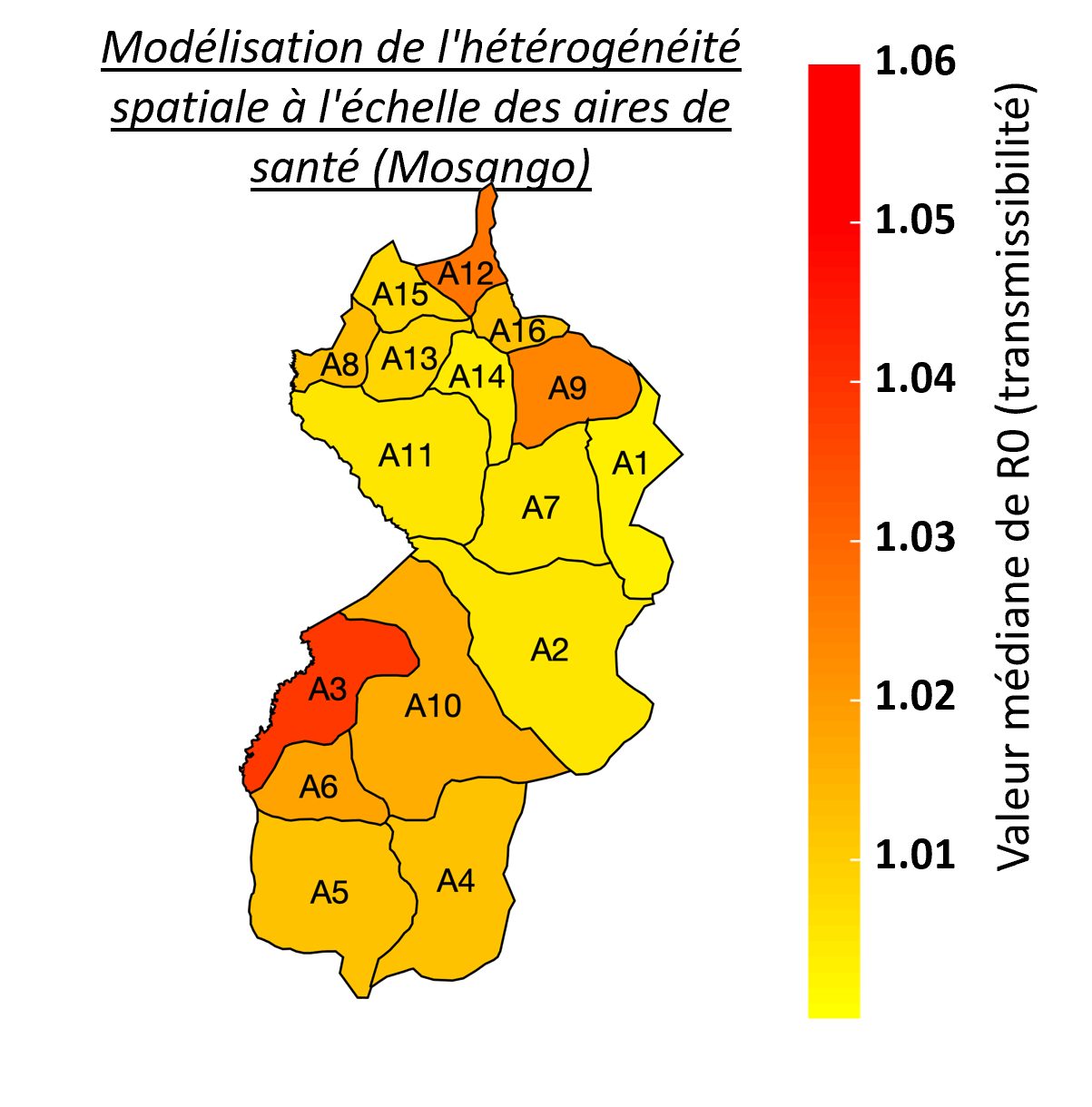
Despite the intensification of gHAT control activities leading to a decline in case numbers in the last twenty years, new challenges have now emerged that include locating infection hotspots and where and how to focus future elimination efforts. As the HAT MEPP team continue to support on the ground activities, and as cases continue towards zero, so the model needs to capture the diseases’ heterogeneity at smaller spatial scales. For the DRC, a country with the highest gHAT burden, data at the smaller health area level is limited, often noisy and with small population sizes (health areas contain approximately 10,000 people versus 150,000 per health zone), which make this modelling task particularly challenging. Focusing on the health zone Mosango (DRC), our model was fitted to data from its 16 health areas and an aggregation of the results was shown to correspond well with previous health zone level fits (right; blue represents aggregated health area fits, red represent the health zone fit, black line represents real case data). Adaptation of the model also enables it to better capture chance events in small populations or at very low infection levels and to reflect the model's uncertainty providing better estimates of when EoT for gHAT may occur. The advantages of smaller spatial scale modelling is reflected in the detail available in locations where cases occur and can indicate in which health area most transmission of gHAT is happening (left: red represents higher transmissibility (R0) value). Health area modelling will enable the simulation of localised intervention strategies which were not possible at larger spatial scales. Although roll-out to other analysable health areas is now necessary, using smaller spatial scales to reflect the real-world situation in the models will improve recommendations of how and where to focus efforts and interventions to achieve elimination of gHAT transmission.
Davis CN, Crump RE, Sutherland SA, Spencer SEF, Corbella A, Chansy S, Lebuki J, Miaka EM, Rock KS (2024) Comparison of stochastic and deterministic models forgambiensesleeping sickness at different spatial scales: A health area analysis in the DRC.Link opens in a new window Link opens in a new windowPLoS Computational Biology Paper summaries: EnglishLink opens in a new window FrenchLink opens in a new window
Modelling the risk of programme interruptions in the DRC
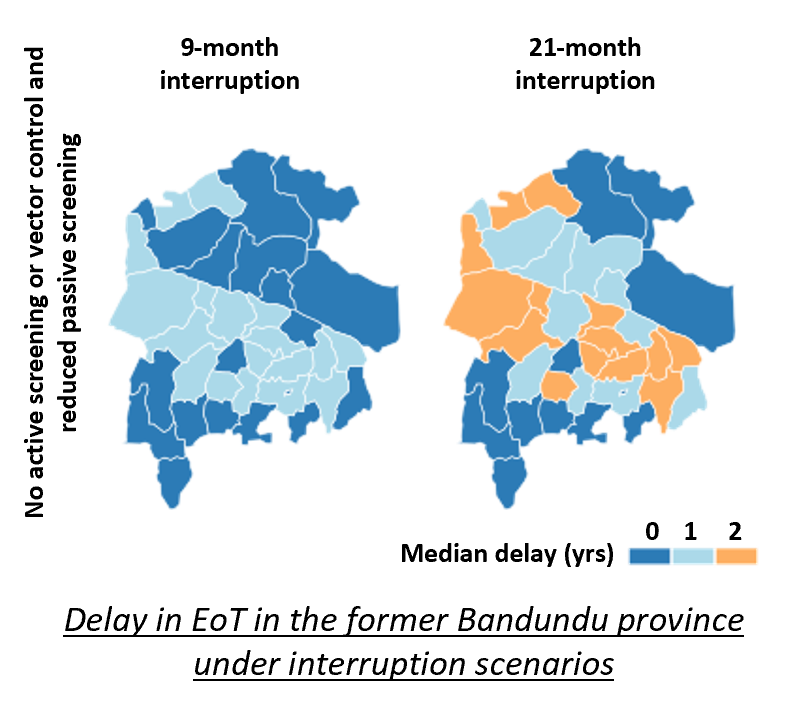
Motivated by the widespread disruption to health systems caused by the COVID-19 pandemic, the team's recent research has sought to understand the impact that unplanned disruptions to the gHAT control programme could have on disease burden and time to achieve the elimination of transmission (EoT). Analysis of interruption scenarios was performed on 38 health zones from the former Bandundu province of the DRC. Dates and durations of interruptions were loosely based on the COVID-19 pandemic and we considered how interruptions to different programme components would impact transmission dynamics, disease burden and the probability and year of EoT.
This important piece of research demonstrates that disruptions to gHAT control programmes can impact the level of disease burden and the probability and year of EoT. However, they are believed to be fairly resilient to short-term shocks if activities are resumed post-interruption with EoT not predicted to be delayed by more than the length of interruption plus one additional year under the most extreme interruption scenario (see figure above). This is likely to be a consequence of the slow-progressing nature of the gHAT infection
Identifying regions of DRC requiring intensified strategies
Mathematical modelling has been used to study the overall impacts of combinations of the currently available intervention methods on transmission across the DRC and highlight regions requiring intensified interventions to achieve elimination of transmission (EOT) by the WHO’s 2030 goal. The Warwick gHAT model - previously developed and fitted to longitudinal human case data in DRC (see above) – was used to predict the expected numbers of active cases, passive cases and new infections.
Data and analysis toolLink opens in a new window: Projected case detections, new infections and estimated year of elimination of transmission of gHAT for DRC health zones, resulting from different intervention strategies, are available to view online via a data and analysis tool (GUI), providing online access to the vast amount of data and results of this research project in a user-friendly way.
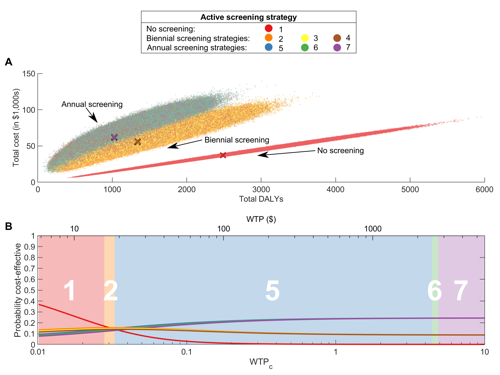
Cost and cost-effectiveness evaluations for 5 health zones
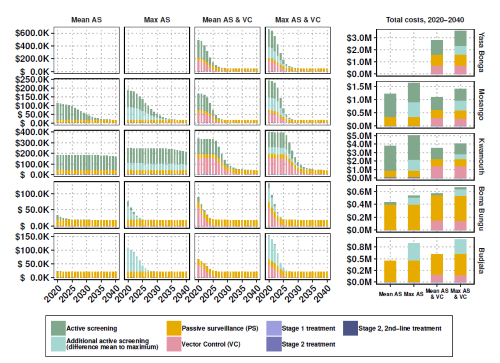
The Warwick gHAT model has been further expanded to provide the cost and cost-effectiveness of the four intervention strategies (see opposite) in terms of net benefit (cost of interventions vs cost of ill-health/disability/death), which is added to the model. Costs include diagnosis, confirmation, and staging via lumbar puncture, well as the cost of the drug itself and the administration. The research offers a number of different options highlighting the most efficient intervention strategy to achieve elimination by 2030 and the optimal strategy to lower costs.
Data and analysis toolLink opens in a new window: Projected costs of different intervention strategies and cost-effectiveness for 5 health zones in DRC are available to view online via a data and analysis tool (GUI), providing online access to the vast amount of data and results of this research project in a user-friendly way.
Antillon M, Huang C, Crump RE, Brown P, Snijders R, Miaka EM, Keeling MJ, Rock KS, Tediosi F (2022). Cost-effectiveness of sleeping sickness elimination campaigns in five settings of the Democratic Republic of Congo.Link opens in a new window Nature Communications Paper summaries: EnglishLink opens in a new window, FrenchLink opens in a new window
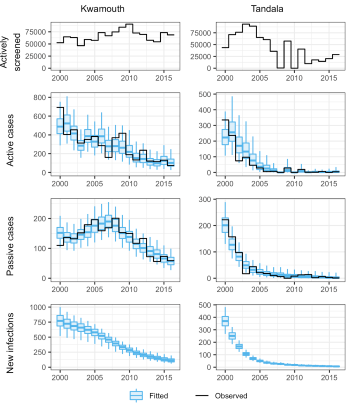
Matching the gHAT model to real case data
State-of-the art statistical fitting methodology has been used to automate calibration of a model of gambiense Human African Trypanosomiasis (gHAT) to longitudinal data (2000–2016) across endemic health zones of the Democratic Republic of Congo (DRC). Through modelling with this data we aim to quantify key underlying factors which contribute to the observational and transmission variation across the country.
The image opposite shows how well the model fits to the timeseries of reported cases, and where there has been a change in the passive case detection rate during the data collection period.
Data and analysis toolLink opens in a new window: Retrospective (2000-2016) DRC gHAT case data by health zone is available to view online via a data and analysis tool (GUI), providing online access to the vast amount of data and results of this research project in a user-friendly way.
Optimising screening with low and falling case numbers
With limited numbers of active screening teams and resources, it is important to optimise their activities with the aim of driving towards elimination. With gHAT case numbers decreasing, we investigate how to optimise active screening visits to individual villages in DRC, such that the costs of the screening programmes can be minimised, while continuing to avert disability-adjusted life years. A stochastic gHAT infection model has been implemented across a range of active screening strategies and the net monetary benefit (NMB - cost of interventions vs cost of ill-health/disability/death) of each calculated.
Peer reviewed paper: Davis, C, Rock, KS, Antillón, M, Miaka, EM, Keeling, MJ (2021). Cost-effectiveness modelling to optimise active screening strategy for gambiense human African trypanosomiasis in endemic areas of the Democratic Republic of CongoLink opens in a new window. BMC Medicine. Paper summaries: EnglishLink opens in a new window, FrenchLink opens in a new window
COVID-19 interruption impact of gHAT
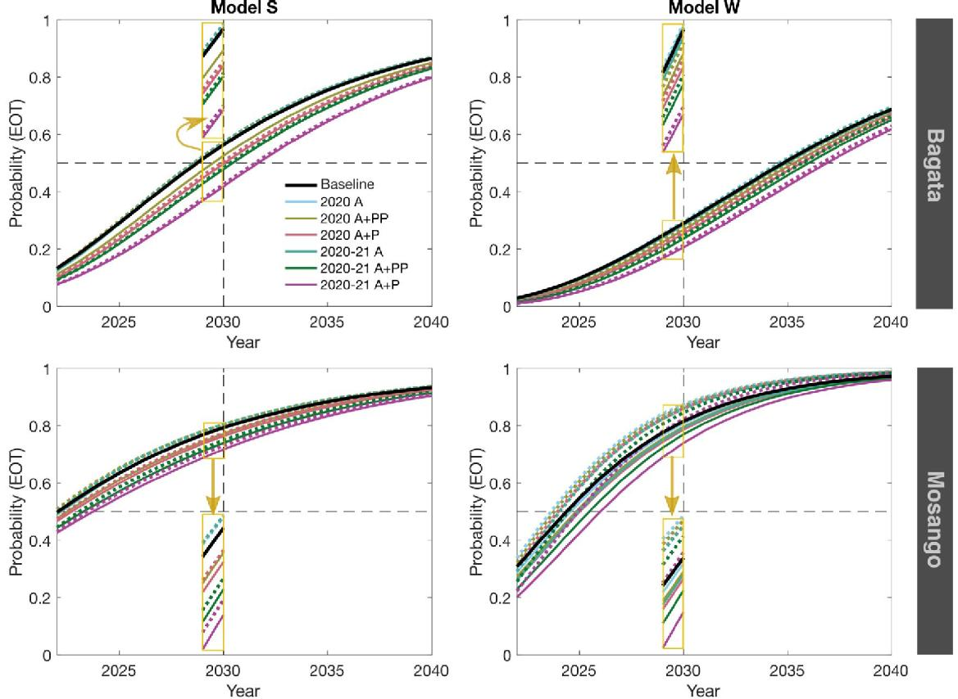
The Warwick and Swiss TPH teams independantly fitted their models to data to two health zones of the Democratic Republic of Congo (DRC) - Bagata and Mosango, both moderate-risk - and use them to assess what the impact of interruptions of medical interventions (active screening and passive surveillance) could have in these regions.
We predicted that delays in achieving elimination of transmission (EOT) would be similar to or less than the length of interruption. For Mosango, EOT may still be achieved by 2030, however, in Bagata the elimination goal is unlikely without intensifying interventions, even without interruptions.
The results suggest that retaining functioning passive surveillance, even partially, can help to avoid significant delays in EOT and to prevent substantial increases in mortality. Mitigation through increasing coverage of active screening following interruption could also improve the probability of meeting the 2030 EOT goal.
Peer reviewed paper: Aliee, M, Castaño, MS, Davis, CN, Patel, S, Miaka, EM, Spencer, SEF, Keeling, MJ, Chitnis, N, Rock, KS (2021). Predicting the impact of COVID-19 interruptions on transmission of gambiense human African trypanosomiasis in two health zones of the Democratic Republic of CongoLink opens in a new window. Transactions of the Royal Society of Tropical Medicine and Hygiene. Paper summaries: EnglishLink opens in a new window, FrenchLink opens in a new window
Meeting Report: NTD Modelling Consortium. The potential impact of programmes interruptions due to COVID-19 on 7 neglected tropical diseases: a modelling-based analysis