SWNT-based Electrochemical Devices
SWNT UMEs
Single-Walled Carbon Nanotube Network Ultramicroelectrodes
Taken from reference 5.
CVD networks of pristine SWNTs grown on silicon oxide have interesting attributes, such as low capacitance and small active area, which make them powerful electrode materials for many applications. Ultramicroelectrodes (UMEs) offer several advantages over macroscopic electrodes, including low background currents and diminished effects of double layer charging. We demonstrate through the use of simple lithographic procedures that it is possible to fabricate SWNT network disk UMEs which offer superior characteristics over conventional metal disk UMEs. The well-defined geometry of the SWNT network electrode is also useful for studying the fundamental electrochemical properties of electron transfer at SWNTs.
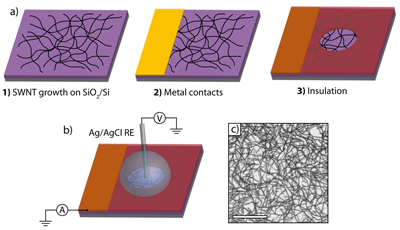
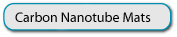
Figure 1: a) Schematic of the lithographic procedure for fabricating SWNT disk UMEs. b) Schematic of the experimental droplet cell setup used for electrochemical measurements. c) FE-SEM image of a typical SWNT network sample, with a density of 4 µmSWNT µm-2. Scale bar is 5 µm.
Cyclic voltammetry experiments demonstrate that for redox species concentrations ≤1 mM the steady-state behavior of the SWNT network electrode is as expected for a metal UME of the same size, however the surface coverage of the active electrode is less than 1%, leading to useful attributes. Given the high mass transport rates to the SWNTs, the reversible nature of the CVs recorded suggests that it is unlikely that electron transfer is defect driven. Interestingly, at concentrations >1 mM for the reduction of Ru(NH3)63+, the CVs became distorted, most likely due to contributions from both SWNT network resistance and electron transfer kinetics.
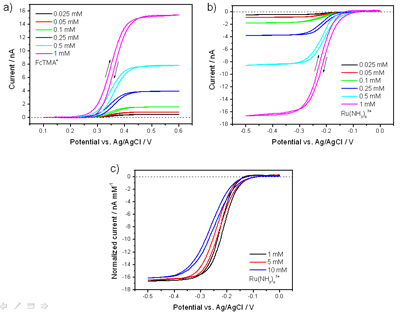
Figure 2: a) FcTMA+ oxidation and b) Ru(NH3)63+ reduction at concentrations of 0.025, 0.05, 0.1, 0.25, 0.5, and 1 mM in 0.1 M NaCl supporting electrolyte at a 100 µm diameter SWNT disk UME; c) Plots of current normalized with respect to redox mediator concentration against potential for Ru(NH3)63+ reduction, for concentrations of 1 mM (black), 5 mM (red) and 10 mM (blue) in 0.1 M NaCl supporting electrolyte. Scan rate in all cases was 4 mV s−1.
Due to the low surface area of the network electrode and the low capacitance of pristine SWNTs, the SWNT network electrodes are shown to have very fast response times, with the charging current decaying significantly faster at a 100 µm diameter SWNT network UME than for a conventional 100 µm Pt UME, sealed in glass. This has critical implications for the use of such electrodes in the measurement of fast kinetic processes especially when employed in conjunction with perturbation techniques such as rapid pulse voltammetry.
Figure 3: Current–time discharge curves at a 100 µm diameter SWNT disk UME (black) and glass sealed 25 µm Pt UME (red), 50 µm Pt UME (green) and a 100 µm diameter Pt UME (pink). Also shown is the response for a 100 µm lithographically defined Pt UME (blue).
Trace Detection
Trace level amperometric detection using high density SWNT network electrodes
Discrimination against currents which arise from background and non-faradaic processes represent a significant challenge in the use of amperometry for trace level analysis. Background and non-faradaic currents typically scale with the area of the electrode and may arise, for example, from capacitative effects associated with charging of the electrode/electrolyte interface (non-faradaic), electrode surface reactions and solvent decomposition (at the limits of the potential window). Two-dimensional networks of pristine SWNTs (with a density above the metallic percolation threshold) which cover only a small fraction of the surface (< 1%) behave as macroscopic electrodes (response dependent on the geometric area of the substrate), but with unprecedented low background currents, dependent on the area of SWNTs. This facilitates cyclic voltammetry at low concentrations ( ~ nM) particularly important given that carbon is a key electrode material for biochemistry.
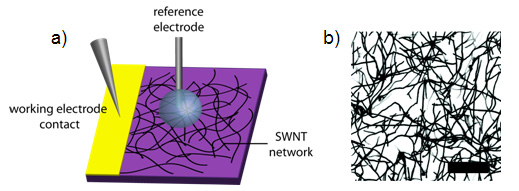
Figure 1: a) Schematic of the setup for electrochemical measurements with SWNT network electrodes. b) FE-SEM image of a SWNT network, density ~ 5 µmSWNT µm-2. Scale bar represents 2 µm. Note that FE-SEM exaggerates the width of SWNTs. Taken from reference 1.
SECM of Networks
Assessment of the Electrochemical Behavior of 2D Networks of SWNTs
SECM has been employed in the feedback mode to assess the electrochemical behavior of 2D networks of SWNTs. Even though the network comprises both metallic and semiconducting SWNTs, at high density (well above the percolation threshold for metallic SWNTs) and with approximately millimolar concentrations of redox species, the network behaves as a thin metallic film, irrespective of the formal potential of the redox couple (Figure 2). This is particularly striking since the fractional surface coverage of SWNTs is only ~1% and SECM delivers high mass transport rates to the network.
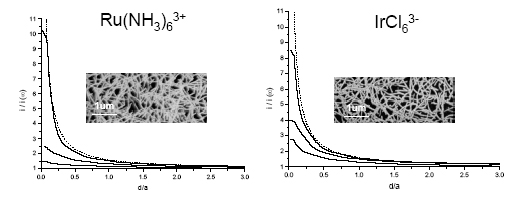
Figure 2: Experimental approach curves recorded on two high density SWNT samples with a 25 µm diameter Pt UME (RG = 10). Tip biased at (left) Etip = -0.5 V and (right) Etip = 0.90 V. In each case the concentrations of mediator were (bottom to top) 20, 10, 1 mM. Taken from reference 2.
The SECM feedback response diminishes at higher concentrations of the redox species. However, wet gate measurements show that at the solution potentials of interest the conductivity is sufficiently high that lateral conductivity is not expected to be limiting (Figure 3). This suggests that reaction kinetics may be a limiting factor, especially since the low surface coverage of the SWNT network results in large fluxes to the SWNTs, characterized by a low density of electronic states.
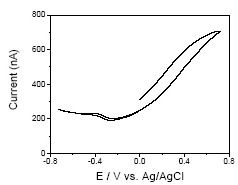
Figure 3: Conductivity voltammetry for a SWNT network electrode (area of exposed SWNTs: 350 µm x 60 µm), density 6.5 ± 0.2 µm µm-2 in a solution containing 1 mM Ru(NH3)63+ in 0.1 M KNO3 supporting electrolyte, at a scan speed of 25 mV s-1. Taken from reference 2.
Metal Templating
Metal templating of nanoparticles and nanowires on SWNT networks
SWNT networks consitute ideal templates for the electrochemical deposition of nanowires and nanoparticles. Initial research focused on the templated deposition of Pt and Ag. Figure 4 shows a FE-SEM image of Pt deposition on a high density SWNT network. Close to the Au contact Pt nanowires can be seen, moving out away from the contact electrode growth tends towards nanoparticle formation with the density decreasing and size of the nanoparticles decreasing with distance. To obtain information on the mechanism of metal growth a device such as the one shown in Figure 4 is constructed. In this case the current signal recorded can be attributed solely to the metal nucleation characteristics.

Figure 4: FE-SEM image of Pt deposition on a high density SWNT network. Taken from reference 3.
m-/s- SWNTs
Simultaneous electrochemical and electrical measurements in solution
We have been interested in examining the electrochemical characteristics of networks of pristine SWNTs on insulating surfaces. The network is electrically contacted with lithographically defined Au contacts and then insulated using a photoresist. An area of the network is exposed by selectively removing the photoresist (Figure 5), thus the electrochemical response is due solely to the SWNTs. By making simultaneous electrochemical and electrical measurements in solution we were able to show that in certain potential ranges (solution and species dependant) the electroactive area of the network is effectively reduced, indicating electrochemistry at semiconducting SWNTs is no longer playing a significant role and the conductivity of the network is dependent upon the applied potential. This is not surprising given that the network contains approximately 2/3 semi-conducting SWNTs and 1/3 metallic SWNTs.
However, this effect is density dependent. For networks which are above the metallic percolation threshold, i.e. there are continuous metallic pathways, the network acts like a thin metallic film (even though the surface coverage of SWNTs on the insulating substrate is only 1 %!) and the electrochemical behaviour is independent of the formal potential of the redox couple. For networks which are above the percolation threshold but do not contain continuous metallic pathways, the response appears to show a dependance on formal redox potential.


Figure 5: a) Schematic and b) FE-SEM image of a SWNT network electrode. c) (top) Cyclic voltammograms at 10 mV s-1 for the oxidation and reduction respectively of 5 mM Fe(phen)32+ and Ru(NH3)63+ in 0.2 M KNO3 for the Au band and SWNT array electrode, and (bottom) Conductivity of the SWNT array electrode as a function of the electrode potential, in the above solution.4
The behaviour shown in Figure 5(c) is due to electrostatic doping of the semi-conducting SWNTs at the more positive applied potentials, which increases the conductance of the network and therefore the magnitude of the electrochemical response.
References
- P. Bertoncello, J. P. Edgeworth, J. V. Macpherson and P. R. Unwin, J. Am. Chem. Soc. 2007, 129, 10982.
- N. R. Wilson, M. Guille, I. Dumitrescu, V. R. Fernandez, N. C. Rudd, C. G. Williams, P. R. Unwin, and J. V. Macpherson, Anal. Chem. 2006, 78, 7006.
- T. M. Day, N. R. Wilson, P. R. Unwin, J. V. Macpherson, J. Am. Chem. Soc. 2005, 127, 10639.
- T. M. Day, N. R. Wilson, J. V. Macpherson, J. Am. Chem. Soc.. 2004, 126, 16724.
- I. Dumitrescu, N. R. Wilson, P. R. Unwin, J. V. Macpherson, Anal. Chem.. 2008, 80(10), 3598.