Emily Corlett
I am currently in the third year of my PhD as part of the Molcular Analytical Sciences CDT.
PhD
The titile of my PhD is Integrating NMR Crystallography into an Experimental and Informatics-Based
Solid-State Characterisation of Pharmaceuticals and Agrochemicals. It builds on the work I completed during my first MSc project and is supervised by Prof. Steven Brown in the Solid State NMR Group, co-supervised by Prof. Richard Walton (Chemistry) and Dr. Helen Blade (AstraZeneca).
Salt and cocrystal formation are used in the pharmaceutical and agrochemical industries to change the physical properties of compounds
Fumaric acid is a pharmaceutically aceptable counterion for salt formation which forms a range of solid structures with different isomers of lutidine:
- salts (ionic molecules)
- cocrystals (neutral molecules)
- cocrystal of salt (mixture of neutral and ionic molecules)
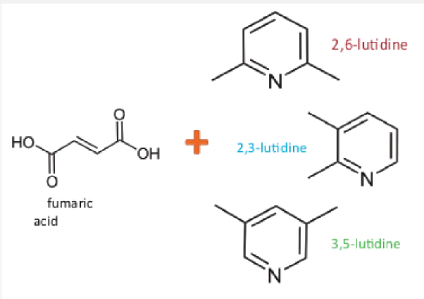
The aim is to use an NMR crystallographic approach to characterise the intermolecular interactions within each system and understand why different forms are adopted, as well as investigating their effects on the systems properties and the complementarity of NMR and XRD
MSc Projects
NMR crystallography for solid-state characterisation of a cocrystal
Cocrystalization can be used to alter the chemical properties of an active ingredient to improve its performance. As structure determines both function and stabilty, it is important to make sure it is fully understood particularly for pharmaceuticals and agrochemicals. Density functional theory was used to calculate theoretical chemical shifts from the X-ray crystal structure of a cocrystal of two fungicides, pyramthanil and dithianon. These calculated shifts were then compared with 1D and 2D solid-state NMR experiments to validate the structure and give an indication of the interactions involved in maintaining it.
Pöppler, A. C., Corlett, E. K., Pearce, H., Seymour, M. P., Reid, M., Montgomery, M. G., & Brown, S. P. (2017) Acta Cryst. C, 73, 149-156.
Developing mass spectrometry to investigate protein carbonylation in plants
Protein carbonylation is a widely accepted biomarker of oxidative stress and thought to play a key role in the drought response of plants, including maize. Modification sites are difficult to identify however, due to the inherently low abundance of modifications throughout the entire proteome. The aim of the project was to develop tagging and enrichment strategies to allow easier identification and quantification of modification sites by MS/MS.
Academic Background
BSc Physics - Durham University (2010 - 2013)
MSc Biophysical Sciences - Durham University (2013 - 2014)
Previous Projects
MSc project - I developed a protocol for determining the proteolytic stability to chymotrypsin of novel peptide mimetics. The digestion was monitored using HPLC to seperate the original species from the digestion products. The protocol was then extended for application to a wider range of mimetics, including peptoids.

e.corlett@warwick.ac.uk
