My Research
PhD Research
My main area of interest is in organic chemistry towards the synthesis of supramolecular structures called rotaxanes. Rotaxanes are unique interlocked complexes consisting of an axle threaded through a macrocycle, it is prevented from slipping off the axle by using large end groups to act as bulky stoppers. Pseudorotaxanes have no such stopper groups so the macrocycle is only kept on by the non-covalent interactions with the axle. We synthesise the molecules by exploiting the weak intermolecular interactions that exist between the two components. These include a wide variety of interactions from hydrogen bonding to Van Der Waals forces. In the image below there are pictorial representations of a pseudorotaxane and a rotaxane. The number before the name denotes the number of components that are mechanically interlocked.
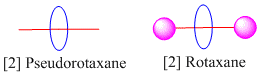
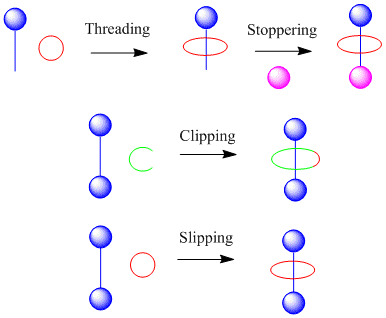
There are three general methods towards forming rotaxanes via the self assembly approach; theses include threading followed by stoppering, slipping and clipping.
- Threading is where the macrocycle is slipped onto the axle and stoppers are attached to trap it into place.
- The clipping approach utilizes a preformed axle and a macrocycle precursor is 'clipped' around it.
- In the slipping approach the axle and macrocyle are preformed and high temperatures are used to distort the macrocycle in such a way that it can pass over one of the stoppers. Once cooled the macrocycle is trapped onto the thread.
Main Supervisor:
Dr Guy Clarkson
Department of Chemistry
University of Warwick
g dot clarkson at warwick dot ac dot uk
