Genomics-driven exploitation of aphid-killing bacteria as a source of novel biopesticides
Principal Supervisor: Dr Mojgan Rabiey
Secondary Supervisor(s): Dr Matthew Jenner (Warwick, Chemistry), Dr Lijiang Song (Warwick, Chemistry), Prof Robert Jackson (Birmingham)
University of Registration: University of Warwick
BBSRC Research Themes: Sustainable Agriculture and Food (Microbial Food Safety, Plant and Crop Science )
Apply now!
Deadline: 23 May, 2024
Project Outline
Aphids are major insect pests of agriculture and horticulture, causing damage to many economically important crop plants through direct feeding and/or as efficient vectors of numerous plant viruses. At present, aphid control measures are limited and rely heavily on insecticides such as carbamates, pyrethroids, neonicotinoids, tetramic acids, and chordotonal organ modulators such as flonicamid/pymetrozine. The target for many of these chemicals is the insect central nervous system, however, insects can rapidly evolve resistance to insecticides thus rendering these chemicals ineffective. There is therefore an urgent need to develop alternative and more sustainable means of aphid control.
Recent studies have shown that environmental microbes have varying abilities to kill insects. On this basis, screening of >140 different plant-associated bacteria through aphid ingestion assays identified multiple bacteria that could kill aphids, with nine strains of different genera showing potent aphid-killing ability. One bacterium in particular (PpR24), a member of the Pseudomonas fluorescens species complex, was observed to grow inside aphids, and is capable of killing insecticide-resistant aphid clones. Furthermore, when sprayed onto plants, PpR24 can reduce aphid infestation. RNA-sequencing and genetic manipulation of PpR24 has demonstrated that it uses a range of toxins to kill the aphid. Together, these findings indicate great promise for the use of this bacterium, and others, as biocontrol agents to control aphid infestation of crop plants. Building on this concept, a recent genome analysis of one of the other nine strains (P. fluorescens PfR37) identified a biosynthetic gene cluster (BGC) that may produce a pyrrolizidine alkaloid-like molecule. This class of molecules are usually produced by plants as a defence mechanism against insect herbivores but has recently been discovered to be produced by bacteria.
This project aims to apply a genomics-driven chemical ecology approach to understand the molecular details of the aphid-microbe interaction. Initially, a broad screen of environmental bacteria will be conducted to identify novel aphid-killing strains. These will be prioritised on the basis of aphid-killing ability and subjected to complete genome sequencing to identify the biosynthetic potential of each bacterium. Prioritised strains will be cultured in the laboratory to induce specialised metabolite production and screened using liquid chromatography-high resolution mass spectrometry (LC-HRMS) to identify metabolites of interest and linked to BGCs via gene knock-out experiments. Metabolites will be purified using preparative-high performance liquid chromatography (HPLC) and chemical structures will be determined using NMR spectroscopy. A combination of crude extracts and purified metabolites will be tested for their toxicity towards aphids using an established assay, with the ultimate aim of identifying novel aphid-killing agents.
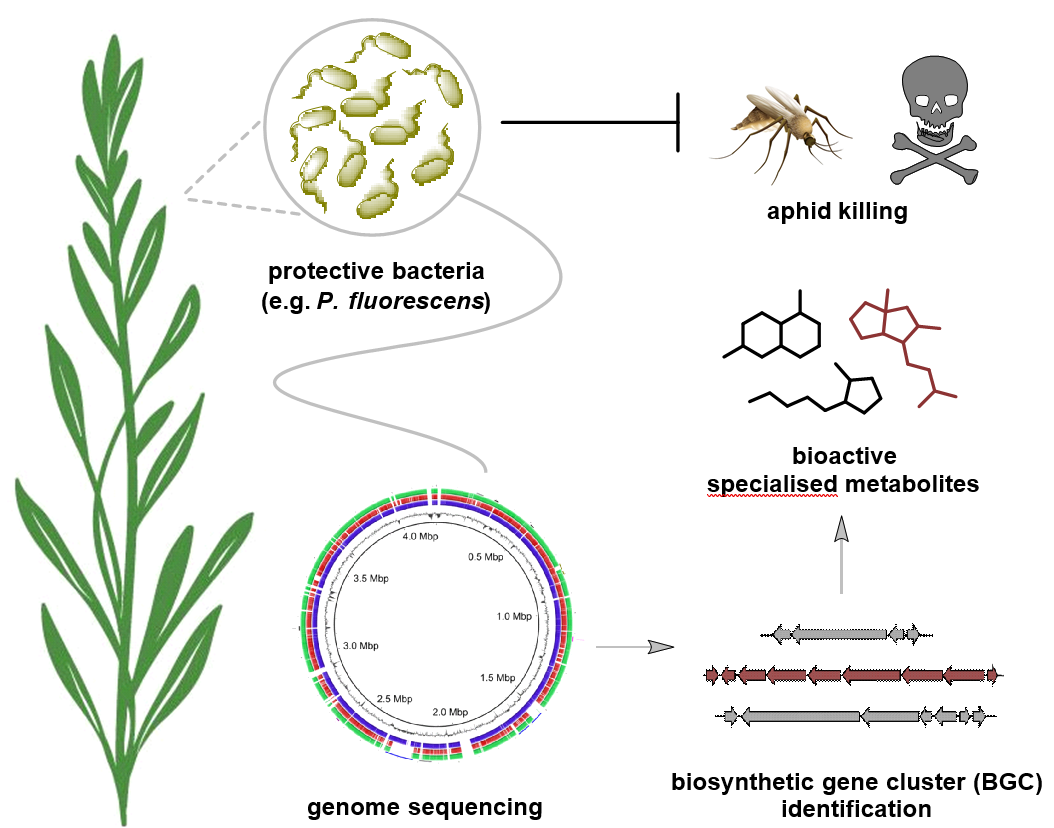
Fig. 1 | Environmental bacteria protect plants from aphid infestation through production toxic specialised metabolites. Schematic highlighting how plant-protective bacteria are able to produce aphid-killing specialised metabolites through expression of biosynthetic gene clusters housed within their genomes.
This project will be hosted in the laboratories of Dr. Mojgan Rabiey (Warwick, SLS) and Dr. Matthew Jenner (Warwick, Chemistry) at the University of Warwick, and conducted in collaboration with Prof. Robert Jackson (University of Birmingham).
Techniques
Genomics and Bioinformatics
Microbiology and Cell Culture
Molecular Cloning and Genetic Engineering
Metabolite Screening (LC-HRMS)
Metabolite Purification (HPLC)
Chemical Structure Elucidation (NMR)