Research
The Pike group is part of the Synthesis and Catalysis and Materials and Polymers Research Themes at the University of Warwick.

Pike group research interests lie at the interface between molecules and materials. In particular, we are designing new metal-oxo cluster molecules. These molecules can be considered tiny versions of well-known semiconducting materials e.g. ZnO or TiO2, however at such small sizes the structures are well defined and we can probe reactivity and mechansism using molecular characterisation techniques (e.g. NMR spectroscopy). These new cluster species have potential uses in (photo)catalysis and as building blocks or precursors to functional materials.

We are particularly interested in understanding the atomic structure of these molecules (by techniques such as X-ray crystallography), and locating potential active sites for well-defined catalytic processes.

In partnership with collaborators, we study the electronic structure of these systems. The combination of metal and oxygen orbitals begins to form ‘bands’ of molecular orbitals as the clusters become larger in size and act less like molecules and more like materials.
We specialise in utilising reactive organometallic, metal amide and metal alkoxide reagents to build our clusters. The group use our own glovebox and Schlenk lines in the lab to ensure air free conditions for these reactions.
Highlighted publications: Hydrolysis of organometallic and metal–amide precursors: synthesis routes to oxo-bridged heterometallic complexes, metal-oxo clusters and metal oxide nanoparticles
By utilising a range of spectroscopic techniques we can understand photoredox reactivity that occurs when we irradiate cluster with light. This can give insight into processes that can occur at semiconductor surfaces and will lead to new photocatalysts.

Highlighted publications: Photo-redox reactivity of titanium-oxo clusters: mechanistic insight into a two-electron intramolecular process, and structural characterisation of mixed-valent Ti(III)/Ti(IV) products
Well-defined multi-metallic clusters can also act as ideal single-source precursors for functional materials, for example, cluster molecules containing Bi and V can be heated to prepare BiVO4 thin films useful as photoanodes for photoelectrochemical water splitting.

Highlighted publications: Single‐Source Bismuth (Transition Metal) Polyoxovanadate Precursors for the Scalable Synthesis of Doped BiVO4 Photoanodes
A simple one-step synthetic route to access a range of metal-doped polyoxovanadate clusters
The use of mixed-metal single source precursors for the synthesis of complex metal oxides
Seb's previous research projects include:
- Understanding the synthesis, redox reactivity and catalytic potential of nanoparticles
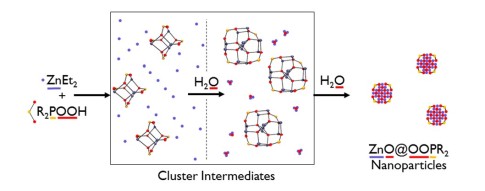
Highlighted publications: Simple Phosphinate Ligands Access Zinc Clusters Identified in the Synthesis of Zinc Oxide Nanoparticles.
Reversible Redox Cycling of Well-Defined, Ultrasmall Cu/Cu2O Nanoparticles.
- Organometallic chemistry within the solid state

Highlighted publications: Stoichiometric and Catalytic Solid-Gas Reactivity of Rhodium Bis-Phosphine Complexes.
Solid-State Synthesis and Characterization of σ-Alkane Complexes
Synthesis and Characterization of a Rhodium(I) σ-Alkane Complex in the Solid State.

Tom making copper rainbows in the lab!
Crystal structure of a complex containing Bi, V and Co metals, useful as a single source precursor to access Co-doped BiVO4 for photoelectrochemical water oxidation.

Colour change of a titanium-oxo cluster upon exposure to UV light.
1 nm sized Ti-oxo cluster
