Reviews
Reviews and Books
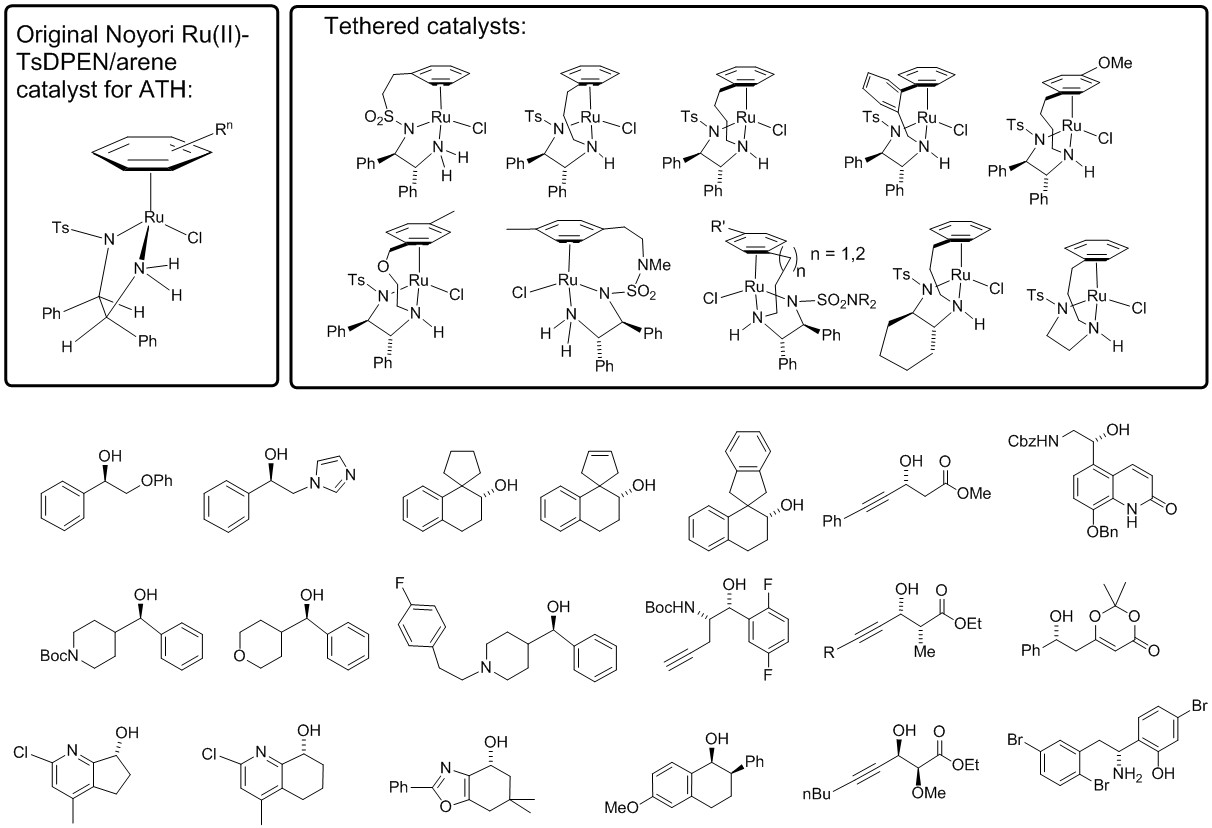
36) Book Chapter 'Tethered Ruthenium Catalysts in Asymmetric Transfer Hydrogenation', Chapter 7 in Asymmetric Hydrogenation and Transfer Hydrogenation, 2021, Wiley-VCH, Editors: Phannarath Phansavath, Virginie Ratovelomanana-Vidal. ISBN: ISBN: 978-3-527-34610-3.
35) 'A diversity of recently reported methodology for asymmetric imine reduction', Jonathan Barrios-Rivera, Yingjian Xu, Martin Wills and Vijyesh Vyas, Organic Chemistry Frontiers (Org. Chem. Front.), 2020, 7, 3312-3342, accepted 2nd September 2020, first published 3rd September 2020.
34) 'Applications of N′-monofunctionalised TsDPEN derivatives in asymmetric catalysis' Jonathan Barrios-Rivera, Yingjian Xu and Martin Wills, Org. Biomol. Chem. 2019,17, 1301-1321, first published on 04 Jan 2019.
33) 'The Development of Phosphine-Free "Tethered" Ruthenium(II) Catalysts for the Asymmetric Reduction of Ketones and Imines', Hans G. Nedden, Antonio Zanotti-Gerosa and Martin Wills. Jointly authored with Johnson Matthey, Cambridge. First published: 15 August 2016. The Chemical Record, Volume 16, Issue 6
December 2016, Pages 2619–2639. Available in open-access form.
32) 'Imino Transfer Hydrogenation Reactions' ReviewTopics in Current Chemistry, April 2016, 374:14
First online: 14 March 2016. http://link.springer.com/article/10.1007/s41061-016-0013-7?wt_mc=alerts.TOCjournals
A review of methods for imine reduction, including asymmetric reductions.
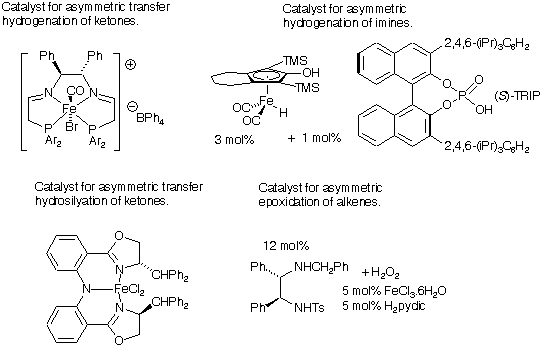
31) ‘Asymmetric catalysis using iron complexes – ‘Ruthenium Lite’?’ M. Darwish and M. Wills, Catal. Sci. Technol. 2012, 2 (2), 243 - 255; http://xlink.rsc.org/?doi=C1CY00390A. WRAP.
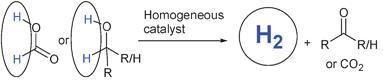
30) ‘Hydrogen generation from formic acid and alcohols using homogeneous catalysts’ Tarn C. Johnson, David J. Morris and Martin Wills, Chem. Soc. Rev., 2010, 39, 81 - 88 (B904495G) (at http://xlink.rsc.org/?doi=B904495G ).
29) ‘Asymmetric Catalysis Using Air: Clean Kinetic Resolution of Secondary Alcohols’, Martin Wills, Angew. Chem. Int. Edn. 2008, 47, 4264-4267.
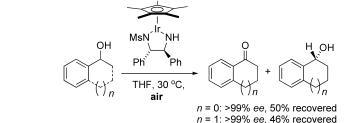
28) ‘Asymmetric catalysts for pressure and transfer hydrogenation of ketones.’, David J. Morris and Martin Wills, Chem Oggi, 2007, 25, Catalysis Supplement, 11-13.
27) ‘Imino Reductions by Transfer Hydrogenation’, Chapter 11 of ‘Modern Reduction Methods’ ed. P. Andersson and I. Munslow, 2007, Wiley-VCH, Weinheim, pp 271-296.
26) Weighing is optional: Highly active and practical catalysts for asymmetric ketone transfer hydrogenation. David J Morris and Martin Wills, Pharmachem. September 2006, pp 8-11.
25) ‘The synthesis and application of BrXuphos: A novel monodentate phosphorus ligand for the asymmetric hydrogenation of ketones’, Martin Wills, Yingjian, Xu, Gordon Docherty and Gary Woodward, in Stanley M. Roberts and John Whittall’s, “Catalysts for Fine Chemical Synthesis Volume 5: Regio- and Stereo- Controlled Oxidations and Reductions, Wiley 2006. pp116-121.
24) Chapter 12.2 The synthesis and application of ESPHOS: A new diphosphorus ligand for the hydroformylation of vinyl acetate. pages 230-237 of 'Catalysts for Fine Chemical Synthesis 3: Metal catalysed Carbon-Carbon bond-Forming Reactions', ed. S. M. Roberts, J. Xiao, J. Whittall and T. E. Pickett, Wiley 2004. ISBN: 0-470-86199-1.
23) J. Hannedouche and M. Wills, “New methodology for the asymmetric reduction of ketones”, contribution to Current Opinion in Drug Discovery and Development, 2002, 5 (6), 881-891.
22) “Enantioselective catalysis using phosphorus-donor ligands containing two or three P–N or P–O bonds “, J. Ansell and M. Wills, Chem. Soc. Rev., 2002, 31, 259-268.
21) “cis-Aminoindanol”, a contribution to the Electronic Reagents for Organic Synthesis series, M. Wills and R. Eaves, Wiley. 2002.
20) “Recent Developments in Asymmetric Transfer Hydrogenation and Hydroformylation Reactions”, M. Wills, Chem. Oggi, 2001, 19, 20-23.
19) “Chemistry and clinical biology of the bryostatins”, R. Mutter and M. Wills, Bioorgan. Med. Chem., 2000, 8, 1841-1860.
18) M.Wills and H. Tye, “Asymmetric Catalytic Processes, 1997”, J. Chem. Soc., Perkin Trans 1, 1999, 1109-1132.
17) M. J. Palmer and M. Wills, “Asymmetric Transfer Hydrogenation Reactions", Tetrahedron: Asymmetry, 1999, 10, 2045-2061.
16) G. Buono, O. Chiodi and M. Wills "Applications of Non-Organometallic Phosphorus Reagents in Enantioselective Catalysis”, Synlett, 1999, 377-388.
15) M. Wills “Asymmetric Catalytic Processes, March-December 1996”, J. Chem. Soc., Perkin Trans 1, 1998, 3101-3120.
14) S. Ebrahim and M. Wills, "Asymmetric Catalysis using Polymeric a-Amino Acid Catalysts", Review for Tetrahedron: Asymmetry, 1997, 8, 3163.
13) "Hydrogenation of compounds containing C=C, C=O and C=N bonds", a review of the field for the "Chemistry of the Functional Groups" series edited by S. Patai, 1996, Supplement A3: The Chemistry of Double-Bonded Functional Groups Part 1, Chapter 15, pp 781-842.
12) "Main Group Elements in Synthesis, January 1994-June 1995", review for "Contemporary Organic Synthesis", volume 3, 1996, pp 201-228.
11) "The Synthesis of Organoboron and Organogermanium Compounds"; a contribution to Comprehensive Organic Functional Group Transformations. Volume 2, 1995, pp 513-548.
10) "Organoiron Chemistry" Chapter 3 in "Practical Approach Series - Transition Metals in Organic Synthesis", ed. S. E. Thomas (series ed. L. M. Harwood and C. J. Moody), Oxford University Press, 1997, Chapter 4, pp 99-132.
9) M. Wills, "Recent Developments in Asymmetric Synthesis", a review of the work carried out in my research group in connection with the Meldola Medal and Prize. Chemical Society Reviews", 1995, 24, pp177-185. Contribution invited by The Royal Society of Chemistry.
8) "Organic Synthesis", an OUP Chemistry Primer, co-authored with C. L. Willis, Oxford University Press, 1995. Translated into German and Japanese.
7) "Main Group Elements in Synthesis, July 1992-December 1993", review for "Contemporary Organic Synthesis", 1994, 1, 339-366.
6) "The Asymmetric Reduction of Ketones", M. Wills and J. R. Studley, Chemistry and Industry, 1994, 552.
5) "Main Group Elements in Synthesis, 1991-July 1992", review for General and Synthetic Methods (RSC Specialist Periodical Report), volume 16, ed. G. Pattenden, pub. Royal Society of Chemistry, 1994, 320-402.
4) "Polycarbocyclic Compounds with Separate Ring Systems, and Spiro Compounds", Chapter 9 of volume IIB, the Second Supplement to Rodd's Chemistry of Carbon Compounds, ed. M. Sainsbury, Elsevier, 1993, Amsterdam, 181-220.
3) "Main Group Elements in Synthesis, 1990", review for General and Synthetic Methods (RSC Specialist Periodical Report), volume 15, ed. G. Pattenden, pub. Royal Society of Chemistry, 1993, 201-261.
2) "Main Group Elements in Synthesis, 1989", review for General and Synthetic Methods (RSC Specialist Periodical Report), volume 14, ed. G. Pattenden, pub. Royal Society of Chemistry, 1992, 275-330.
1) "Main Group Elements in Synthesis, 1988", review for General and Synthetic Methods (RSC Specialist Periodical Report), volume 13, ed. G. Pattenden, pub. Royal Society of Chemistry, 1991, p285-343.
Online Conferences
“Recent Developments in the area of Asymmetric Transfer Hydrogenation”, contribution to the 2nd Electronic Conference on Synthetic Organic Chemistry (ECSOC-2), 1-30 September 1998 (invited paper). M. Wills, M. J. Palmer, A. R. C. Smith, J. A. Kenny and T. Walsgrove.
