Electrochemical sensing directly in bodily fluids – a move towards point of care and in vivo analysis
In vivo sensing has become a fundamental topic in modern medicine due to the ability to directly interrogate biological processes in living organisms. However, few sensors meet the necessary criteria of an in vivo sensor, which must achieve good sensitivity, dynamic range, reversibility, biocompatibility and resistance to fouling. Currently, the focus of most in vivo sensors remains on monitoring normal biological function, whereas there is also need to study pharmaceutical compounds, such as acetaminophen (APAP), for therapeutic monitoring and understanding toxicological effects. Herein, we proposed the use of boron-doped diamond (BDD), as a material for in vivo sensing of APAP. The effects of sp2 carbon on the ability to probe biological matrices were also investigated using electrodes containing varying sp2 content focusing on specificity and detection limits.
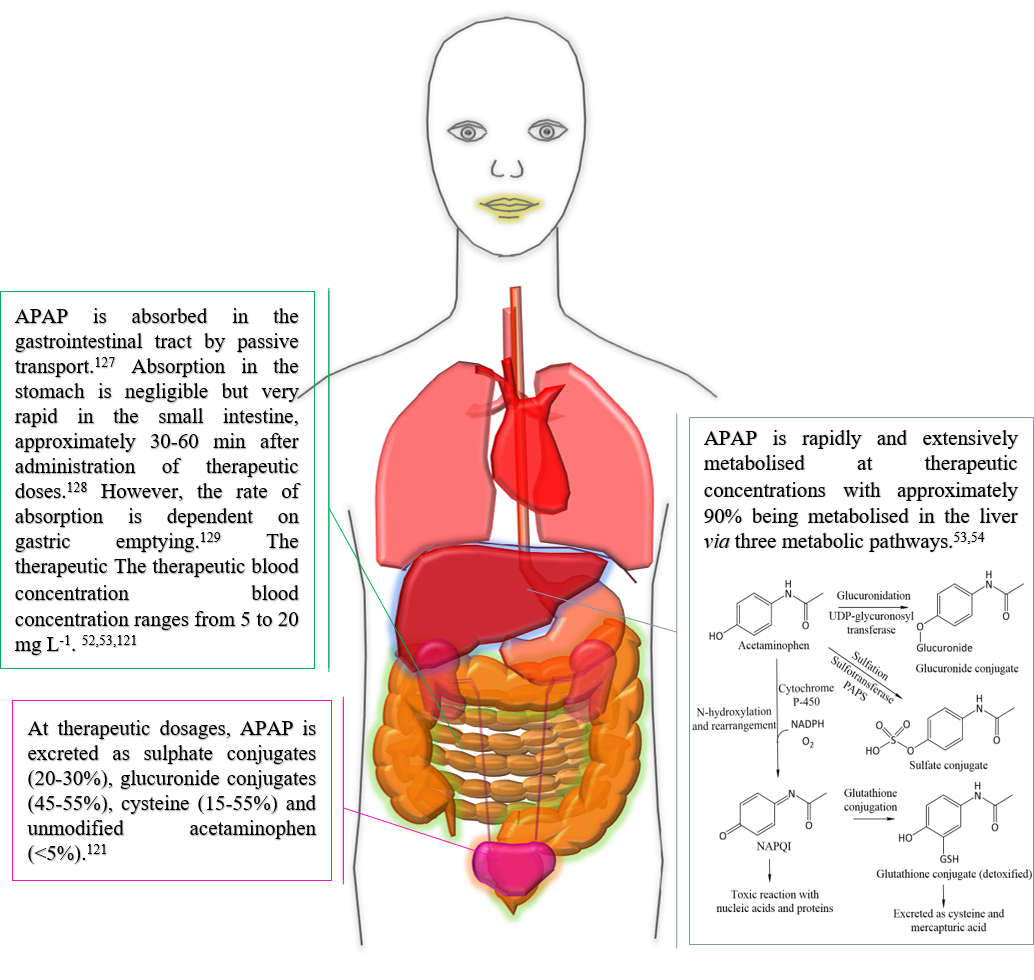
Fig. 1. Pharmacokinetic pathway of APAP.
Proof-of-concept was ascertained through in vitro work by analysing APAP in the presence of 4% bovine serum albumin and synthetic urine, using cyclic voltammetry (CV) and differential pulse voltammetry (DPV).
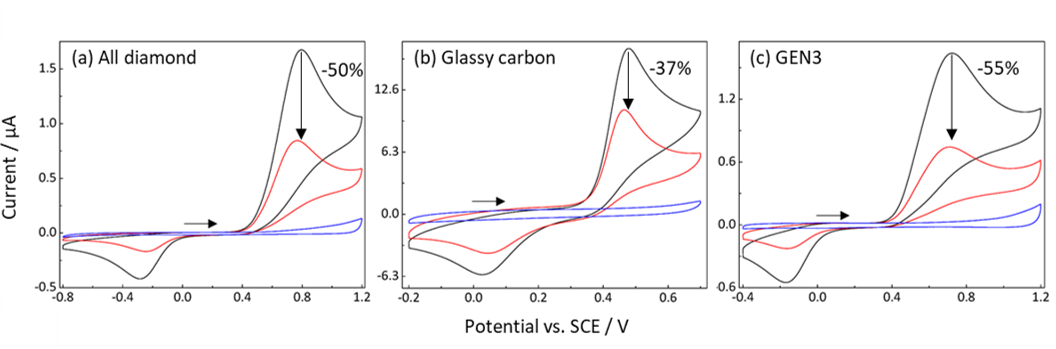
Fig. 2. Cyclic Voltammograms (CVs) of 1 mM (black line), 0.5 mM (red line), and blank (blue line) paracetamol (APAP) in 4% bovine serum albumin (BSA) obtained using (a) all-diamond, (b) glassy carbon and (c) GEN3 electrodes. There is a clear shift in the anodic peak potential when all-diamond (+0.79 V), GC (+0.48 V) and GEN3 (+0.72 V) electrodes were used.
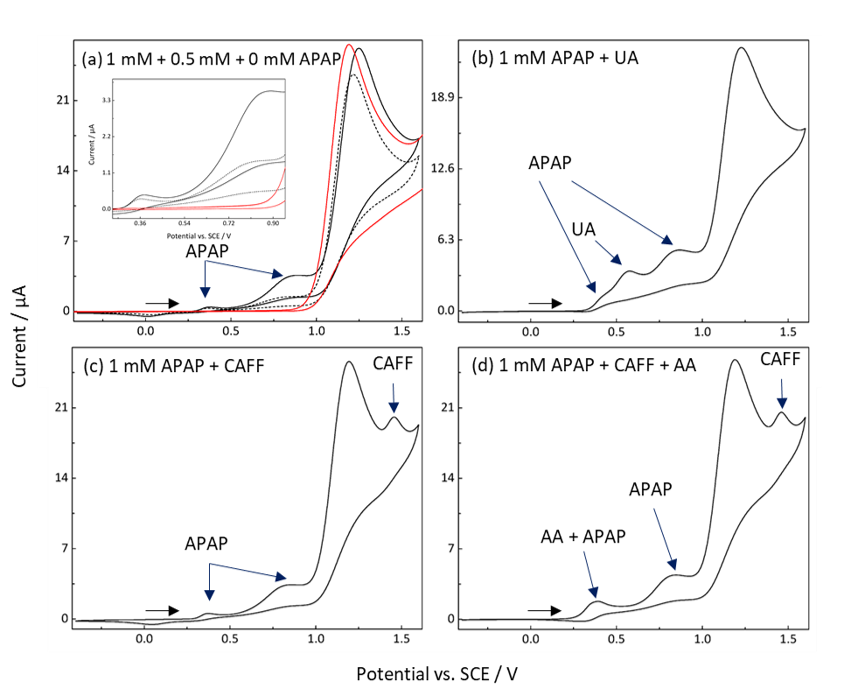
Fig. 3. Simultaneous identification of APAP and other compounds of interest in synthetic urine (pH = 6.9). (a) Identification of APAP in synthetic urine was completed by obtaining CVs of 1 mM (black solid line), 0.5 mM (black dashed line) and blank (red) APAP.
Electrodes containing greater sp2 content exhibited increased fouling in the presence of albumin attributed to the adsorption of hydrophilic domains onto the electrode surface. However, sp2 content was not the sole contributor to electrodes performance, but also the manner of electrode fabrication, treatment and material growth. Despite this, all electrodes demonstrated sufficient limits of detection (LOD) and quantification (LOQ) for monitoring therapeutic APAP concentrations in blood.
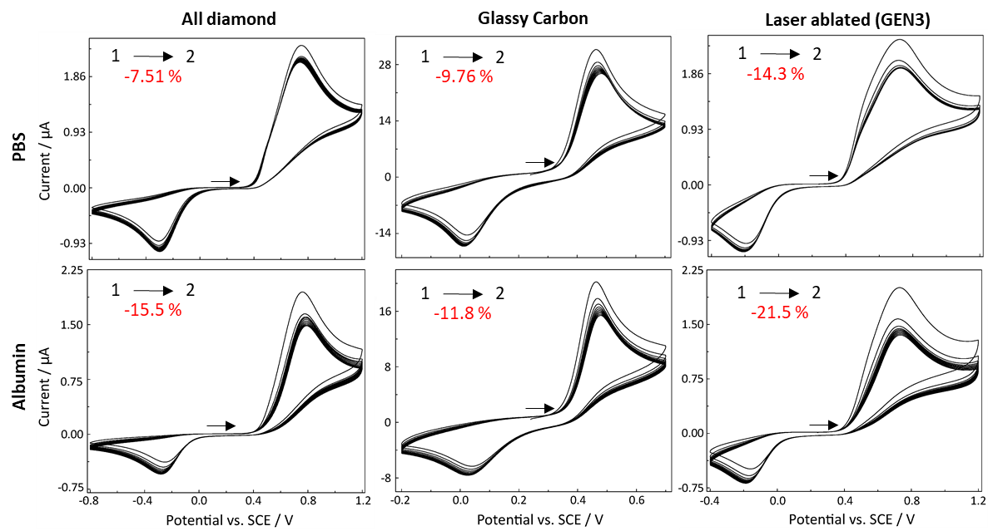
Fig. 4. CVs obtained of 1mM APAP in PBS (pH = 7.4) and 4% BSA using all diamond, GC and GEN3 electrodes for investigating electrode fouling. Ten full scans were obtained at a scan rate of 0.1 V s-1 and electrode fouling measurements were calculated from the decrease in anodic peak current between the first and second scan.
This work paved the way for future in vivo research using BDD which, if successful, could revolutionise personalised medicine by ensuring that the correct drug is provided at the right time and dose.
Fig. 5. Typical three-electrode setup.