Electric Fields
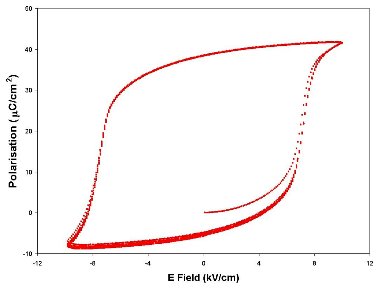
The classic demonstration that a sample is ferroelectric is to measure a spontaneous polarisation that is switchable by the application of an electric field – namely a P-E hysteresis loop as shown below.
 |
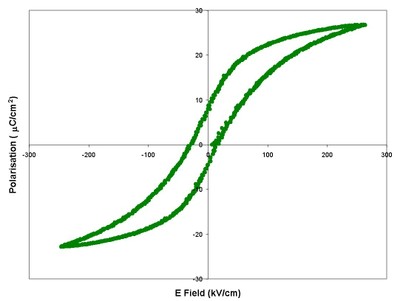 |
| Polarisation versus Electric field (P-E) measurement of a poled soft PZT composition taken at 1 Hz, five consecutive loops overlaid. | Polarisation versus Electric field (P-E) measurement of a 1 μF ceramic multilayer capacitor taken at 1 Hz. The sample is a composite multiferroic with a sandwich of piezoelectric dielectric with magnetostrictive nickel electrodes. |
In collaboration with the NPL Functional Materials Group, the XMaS team has developed an in situ system where P-E loops and X-ray diffraction data are collected simultaneously:
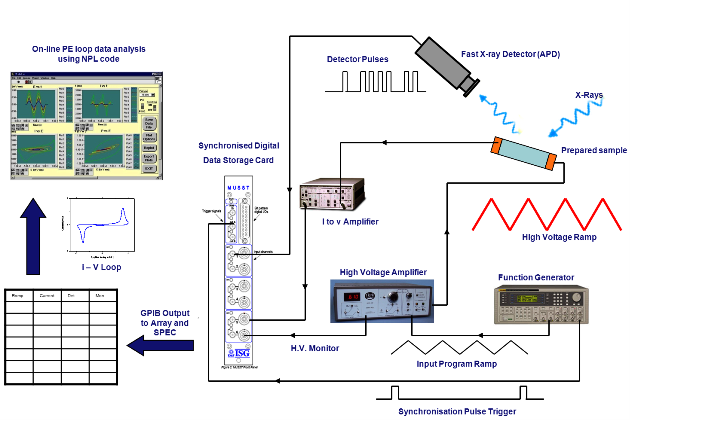
Schematic of the simultaneous acquisition of electrical response and x-ray diffraction during the sample excitation with a triangular electric potential.
The electrical response and the diffraction data can be measured simultaneously by means of a fast acquisition card (MUSST) under the application of either a constant or an oscillating field. The MUSST (Multipurpose Unit for Synchronisation Sequencing and Triggering) card, developed by the ESRF, can collect data in each separate channel at rates up to 20 MHz.
This development aims to help users to carry out studies of ferroelectrics and multiferroics in complex conditions such as magnetic and electric fields. Currently XMaS offers an electric field sample environment with potentials of ± 2 kV with temperatures down to 2 K in a 4 T magnetic field. A separate sample environment provides ± 10 kV down to 10 K in a 1 T field. Samples must be coated with e.g. gold electrodes and electrically isolated by a sapphire support. The high voltage (HV) wires that are connected to the cryostat are soldered onto the gold.
For more details on the P-E measurement system, please contact the team or Mark Stewart at NPL.
 |
More information can be found on the Multiferroics wiki development by NPL and XMaS.
Custom Sample Environments at XMaS
If any custom sample environments are required, please contact a member of beamline staff as soon as possible and we will try to accommodate you. Previous examples have included equipment for performing in situ studies of surfaces in the electrochemical environment. Electrochemical cells have been developed to allow control of the solution and sample temperature over the region of -5 to 80°C and to permit studies of electrochemistry in a magnetic field. Preliminary experiments on the surface reconstructions of Au(111) and Au(001) in electrolyte have been performed.
