Monica Kumar
PhD
Project: "Functional amyloids; investigating the dynamics of aggregation and release of Somatostatin" (Oct 2019 - Sept 2022)
Supervised by Dr Paul Wilson (Chemistry)
As of December 2019, searching for the term ‘amyloid fibrils’ on the widely regarded journal database, Web of Science, yields a considerable 16,900 hits. Notably, over 5000 of the aforementioned hits are from journal articles published in the last five years, highlighting the current interest in studying these highly ordered, insoluble protein aggregates. Given the well-documented association of amyloid fibrils with several neurodegenerative conditions including Alzheimer’s and Parkinson’s disease, the extent of interest in amyloid structures is unsurprising.
It must be noted that although some amyloid fibrils are considered pathogenic with regards to neurodegenerative diseases, other proteins have been found to form ‘functional amyloids’, where the fibrillation process is integral to the protein performing its desired function. Structurally, the defining properties of pathogenic and functional amyloids are shared; typical fibrils are between 7 – 12 nm in diameter and are highly stable, well-ordered and insoluble materials with dominant cross b-sheet secondary structure. Further research into amyloid fibrillation mechanisms have found that most proteins possess the inherent propensity to misfold and aggregate into fibrillar morphologies when subject to denaturing conditions such as extreme pH or temperature. Hence, a stronger understanding of what determines if an amyloid has pathogenic or functional associations is required.
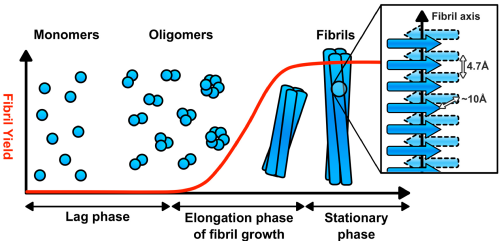
The advent of contemporary microscopic techniques in the 1950s has been instrumental in investigating amyloid fibril morphology and thus advancing our understanding of the structures. Alongside microscopic methods, several spectroscopic techniques have also been key in conducting kinetic fibrillation studies. These techniques include birefringence studies using Congo red dye, kinetic studies of fibrillation using Thioflavin T (ThT) fluorescence assays, identification of characteristic b-sheet structure using circular dichroism (CD) or Fourier-transform infrared spectroscopy (FTIR) and studying amyloid morphology using transmission electron microscopy (TEM) or more recently, atomic force microscopy (AFM).
MSc Mini Projects
Project 1: "Functional Amyloids: Investigating the Dynamics of Somatostatin Aggregation and Release"
Supervised by Dr Paul Wilson (Chemistry)
It is well-documented that many proteins have the ability to form insoluble, β-sheet rich protein aggregates known as amyloids. Pathogenic amyloids are ubiquitously discussed in scientific literature due to their association with neurodegenerative diseases such as Alzheimer’s and Parkinson’s. This project focused on functional amyloid aggregation, whereby the aggregation process is essential in allowing key physiological processes to occur in-vivo. Functional amyloids are known to aggregate reversibly, much unlike known pathogenic amyloids; this reversibility also contributes to the functionality of the amyloids.
Somatostatin-14 (SST) is neuropeptide hormone responsible for broad inhibitory effects on endocrine secretions in the brain, pancreas and gastrointestinal tract. SST has been reported to self-assemble into a functional amyloid structure both with and without the presence of a glycosaminoglycan (GAG). GAGs such as heparin are sulfonated polymers that have been strongly associated with the acceleration of both functional and pathogenic amyloid formation in-vivo. The negatively charged sulfate groups in heparin are thought to bind to positively charged amino acids residues (lysine and arginine), altering the peptide’s structure and thus accelerating its propensity to aggregate. Despite recognising these associations, our understanding of how the presence of GAGs affects the self-assembly mechanism by which peptides/proteins aggregate is limited.
The main aim of the project was to determine if SST aggregation could be induced ex-vivo and to investigate the effect of NaCl solution and the presence of five synthetic heparin-mimicking polymers on the kinetics and dynamics of the peptide aggregation. Thioflavin T dye, commonly used in biophysical studies of peptide aggregation, was added to the SST samples to monitor any amyloid formation over time using UV-vis fluorescence.
Project 2: "Application of Solution and Solid-State NMR Techniques to Characterise the Protein Structure, Dynamics and Interactions of the beta-hairpin Docking Domain of the AerJ Polyketide Synthase"
Supervised by Professor Józef R. Lewandowski (Chemistry)
Polyketide synthases (PKS) are a family of “molecular machines”, with multi-domain, megaenzyme structures that are capable of biosynthesising polyketide products. Many of these natural products possess antimicrobial, anticancer and immunosuppressive properties, making them invaluable in the field of medicine. Although some key information is known about the biosynthesis pathways of the polyketides, a more comprehensive understanding of the structure of the complex PKS system could allow for enzyme engineering to generate novel chemical compounds. Extracted from Microcystis aeruginosa, the beta-hairpin docking domain (bhDD) from the AerJ PKS is the protein of interest in this report. bhDD is known for its responsibility in facilitating protein-protein interactions within the PKS synthesis pathway and understanding more about how the domain functions is of significance to structural biologists.

2D 1H–15N HSQC and a series of 3D triple resonance solution NMR experiments were employed to fully assign the protein backbone of bhDD and TALOS+ software was used to predict the secondary structure of the protein, including the presence of two alpha-helices and two beta-strands. Inverse Cross Polarisation (INVCP) Magic Angle Spinning (MAS) Solid-State (SS) NMR of bhDD was also conducted, utilising sedimentation of the protein using ultracentrifugation of the sample under the MAS conditions. Future perspectives on how to further characterise bhDD using both solution and SS NMR are also discussed.
Previous Education
MSc Molecular and Analytical Science (Distinction) - University of Warwick (Sep 2018 - Sep 2019)
Including taught modules in Chromatography, NMR and Electrochemistry and two, 10-week research projects.
MChem Chemistry (First Class Honours) - University of Warwick (Sep 2014 - Jun 2018)
Previous Research Projects
2017/18 - MChem Research Project: "Surface Enhanced Raman Spectroscopy (SERS) for the Detection of Formaldehyde in Aqueous Solution'
Supervised by Dr Nikola Chmel - Warwick Biophysical Chemistry
Pharmaceutical excipients are key, inert components of many formulations which contribute significantly to medicinal safety and efficacy. Formaldehyde is a common reactive impurity that forms over time within many commonly used drug excipients. This is a significant problem for the pharmaceuticals industry as the presence of impurities like formaldehyde degrade active drug ingredients, thus reducing the drug quality and shelf life. Raman Spectroscopy is a technique that can provide a non-destructive, unique vibrational fingerprint of analyte molecules. However, hindered by a weak signal intensity, it can be amplified by a modified technique, Surface Enhanced Raman Spectroscopy (SERS), a core technique in this report. The aim of this project was to explore the use of functionalised silver and gold nanoparticles to quantitatively detect formaldehyde in aqueous solution using SERS.
2017 - MRC IBR Summer Project: "Determination of Kif15-Tpx2 Binding Interfaces Using Cross-Linking Mass Spectrometry"
Supervised by Professor Andrew McAinsh - Warwick Medical School (CMCB)
An array of proteins are involved in the initiation, mediation and completion of cell division. Kinesins, are a family of motor proteins that are heavily involved in the mitotic process. This project specifically focuses on Kif15, a motor protein responsible for the maintenance of the bipolar spindle. Previous investigations have shown that microtubule-associated protein, TPX2, is responsible for localising Kif15 to the spindle MTs, allowing it to maintain the mitotic spindle. Additionally, studies have found that interactions between Kif15 and TPX2 can inhibit Kif15's motility along MTs, hindering its role within bipolar spindle assembly.
The focus of this project was to ascertain the binding interfaces between Kif15 and TPX2 using Cross-Linking Mass Spectrometry, to gain a clearer understanding of the mechanism by which TPX2 inhibits Kif15's motility along MTs. Kif15 has the potential to become a future therapeutic agent against cancer if the mechanism of its motility inhibition is understood further.
2015 - URSS Summer Project: "Investigating the Photochemistry of Methyl Anthranilate"
Supervised by Professor Vas Stavros - Warwick Physical Chemistry
Using femtosecond gas-phase pump-probe spectroscopy, the ultrafast dynamics of methyl anthranilate were studied through the irradiation the sample with UV radiation. Whilst in an electronically excited state, time-resolved velocity map imaging and photoelectron spectroscopy methods were used to determine potential relaxation pathways. Determining the specific relaxation pathway allows us to understand how the molecule would behave as a potential sunscreen.
Interests
-
Avid runner - part of a local running club and have completed Amsterdam Marathon 2017, St Albans Half-Marathon 2017, Spring Wolf Run 10k 2016 and Coventry Half-Marathon 2015 and 2016
- Enjoys reading - particularly dystopian fiction novels