Introduction to Endofullerenes
Molecular Surgery:
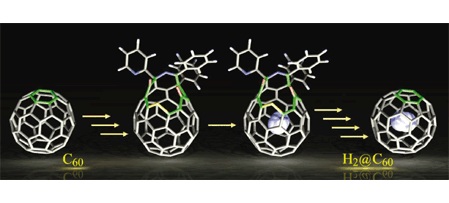
The molecular surgery technique involves a series of carefully controlled chemical reactions to open up the fullerene cage and then insert the guest molecule into the fullerene cage through high temperature and pressure condition. It then follows up with another series of reaction to reseal the orifice while the molecule is inside. This is the technique used to synthesize all variant of dihydrogen endofullerenes. [1, 2]
Spin Isomerism of H2:
Dihydrogen trapped molecules such as H2 and H2O exhibit nuclear spin isomerism which restricts the dynamics of certain spin species of dihydrogen molecules to certain rotational states. The two possible spin-species of dihydrogen molecules are the singlet (para) and triplet (ortho) states . The singlet states have an anti-symmetric spin wavefunction, with a total spin Iz of 0, while the triplet states have a symmetric spin wavefunction, with a total spin Iz of 1. Hence, the para species are NMR silent and only the ortho species can give out NMR signals. The total wavefunction of the molecule is the product of the spin wavefunction and the rotational wavefunction:
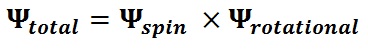
Due to the Pauli Exclusion Principle, the total wave-function of the hydrogen molecule has to be anti-symmetric. Hence, the para states can only have symmetric rotational wavefunctions, with even rotational quantum numbers (j = 0, 2, 4 …). Conversely, the ortho states can only have antisymmetric rotational wavefunctions, with odd rotational quantum numbers (j = 1, 3, 5 …).
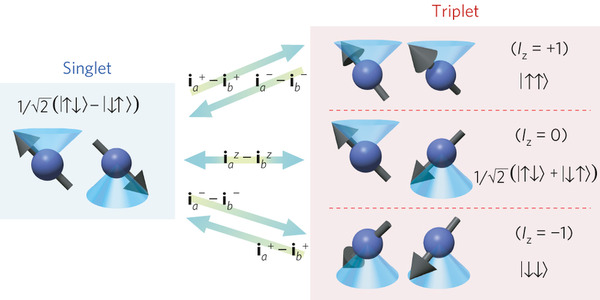
Figure 1. The above illustrates the two possible spin states of H2 molecule, which are the singlet (para) and triplet (ortho) states. The singlet states have an anti-symmetric spin-wavefunction (Iz=0), while the triplet states have a symmetric spin-wavefunction, with a total spin (Iz=1). The degeneracies of the singlet and triplet states are one and three respectively as there are three ways to form a symmetric nuclear spin wave-function, where as there is only one way to form an anti-symmetric nuclear spin wave-function. [3]
References:
1. Rubin Y (1999) Ring opening reactions of fullerenes: Designed approaches to endohedral metal complexes. Top Curr Chem 199:67–91.
2. K. Komatsu, M. Murata, and Y. Murata. (2005) Science, 307:238-240.
3. T. Sugimoto and K. Fukutani (2011) Nature Vol. 7, 307-309.