Insights into to the structure and photochemistry of metal-oxo clusters
Two articles investigating the structure and photochemistry of metal-oxo clusters have been recently published by the Pike group. Metal-oxo clusters are well defined molecular analogues of semiconducting metal oxide materials, and provide an opportunity for precise study.
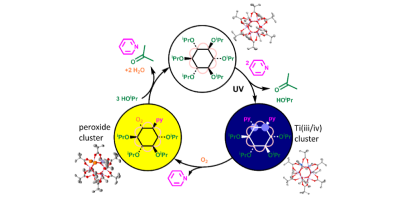
In an article in Chemical ScienceLink opens in a new window earth-abundant titanium-oxo clusters undergo a photoreaction under UV light resulting in a deeply coloured mixed-valence cluster. The study shows that this photoreaction does not occur via free radicals, instead using cooperativity between the multiple titanium centres in the cluster to promote two-electron bond breaking/making steps, paving a way to using these systems for controlled and selective photocatalysis.
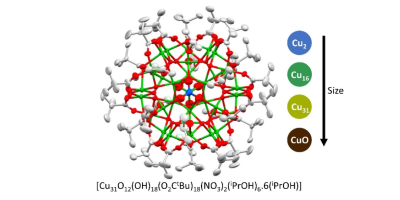
In an article in Chemical CommunicationsLink opens in a new window the structures of new copper-oxo clusters are discovered, including the largest Cu-oxo cluster with only O-donor ligands, which has 31 copper centres and a core diameter of 1.5 nm. This cluster makes a link between small Cu complexes and CuO nanoparticles. The study finds that there is a size colour correlation, with larger clusters absorbing light deeper into the visible region and suggesting potential for useful photochemistry under sunlight.