Aparna Ratheesh
Macrophages (red) migrating into the extended germband of a stage 12 Drosophila embryo (green).
Technical Summary
Cell migration through complex tissue barriers is a key driving force behind essential life processes such as embryonic development and immune homeostasis as well as pathological conditions such as inflammation and cancer metastasis. Within a living organism, cells do not migrate in isolation and hence understanding the mechanical and biochemical interactions between the migrating cells and the surrounding environment is crucial in understanding cell migration in vivo. Yet because of the technical challenges involved in visualizing these process live in vivo, not much is known about how these interactions facilitate migration in a complex tissue.
My lab uses the migration of Drosophila melanogaster macrophages during embryonic development to answer these fundamental questions. Drosophila embryos provide an excellent model system where we can combine fast genetic manipulations with quantitative live imaging, biophysical tools and mathematical modeling. Currently the lab is focusing on two key aspects:
(1) How do macrophages sense the stiffness of the surrounding tissues and alter their force generating machinery to facilitate migration?
(2) How does the macrophage generated forces affect tissue mechanics, cell fate and positioning of the surrounding tissues?
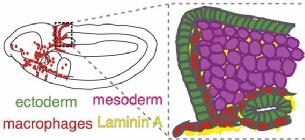
Selected publications
Ratheesh, A.et al. Drosophila TNF modulates tissue tension in the embryo to facilitate macrophage invasive migration. Developmental Cell. 2018 May 7;45(3).
Ratheesh, A. et al. Drosophila immune cell migration and adhesion during embryonic development and larval immune responses. Curr Opin Cell Biol. 2015 Oct;36:71-9.
Ratheesh, A. and Yap, A.S. A bigger picture: classical cadherins and the dynamic actin cytoskeleton. Nat Rev Mol Cell Biol. 2012 Oct; 13(10): 673-9.
Ratheesh, A. et al. Centralspindlin and α-catenin regulate Rho signaling at the epithelial zonula adherens. Nat Cell Biol. 2012 14, 818–828.
Ratheesh, A. and Yap, A.S. Doing cell biology in embryos: regulated membrane traffic and its implications for cadherin biology. F1000 Biol Rep. 2010 Apr 27; 2.

