Jonathan Millar
Technical Summary
 My laboratory has had a long-standing interest in the fundamental mechanisms that regulate the cell division cycle. Initially using the fission yeast, Schizosaccharomyces pombe, as a model organism we have made fundamental discoveries on how cells trigger entry into mitosis (Millar et al., 1991; Millar and Russell, 1992), how this transition is regulated by environmental stress (Millar et al., 1995, Lopez-Avilés et al., 2005) and how cells ensure chromosomes are only replicated once per cell cycle (Wuarin et al., 2002).
My laboratory has had a long-standing interest in the fundamental mechanisms that regulate the cell division cycle. Initially using the fission yeast, Schizosaccharomyces pombe, as a model organism we have made fundamental discoveries on how cells trigger entry into mitosis (Millar et al., 1991; Millar and Russell, 1992), how this transition is regulated by environmental stress (Millar et al., 1995, Lopez-Avilés et al., 2005) and how cells ensure chromosomes are only replicated once per cell cycle (Wuarin et al., 2002).
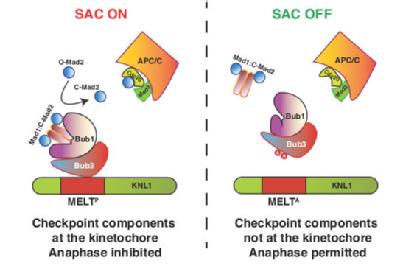
 We have also uncovered novel factors required for the retrieval and bi-orientation of sister chromatids (Sanchez-Perez et al., 2005, Franco et al., 2007) and identified an evolutionarily conserved mechanism that switches off the spindle assembly checkpoint (Vanoosthuyse et al., 2009; Meadows et al., 2011). More recently, we have uncovered how the Bub1, Bub3, BubR1/Mad3, Mad1 and Mad2 spindle assembly checkpoint proteins bind kinetochores and shown that this is conserved in human cells (Shepperd et al., 2012; Silio et al., 2015; Mora-Santos et al., 2016) and identified the RZZ complex, which is absent in yeast, as an independent receptor for the Mad1 and Mad2 proteins in human cells (Silio et al., 2015).
We have also uncovered novel factors required for the retrieval and bi-orientation of sister chromatids (Sanchez-Perez et al., 2005, Franco et al., 2007) and identified an evolutionarily conserved mechanism that switches off the spindle assembly checkpoint (Vanoosthuyse et al., 2009; Meadows et al., 2011). More recently, we have uncovered how the Bub1, Bub3, BubR1/Mad3, Mad1 and Mad2 spindle assembly checkpoint proteins bind kinetochores and shown that this is conserved in human cells (Shepperd et al., 2012; Silio et al., 2015; Mora-Santos et al., 2016) and identified the RZZ complex, which is absent in yeast, as an independent receptor for the Mad1 and Mad2 proteins in human cells (Silio et al., 2015).
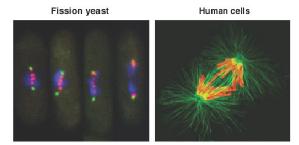
The laboratory uses a combination of genome engineering, molecular genetics, biochemistry, phospho-proteomics and high speed multi-wavelength live cell imaging in both fission yeast and human tissue culture cells (see figure) to explore the mechanistic basis of spindle checkpoint signaling.
Selected publications:
Currie CE, Mora-Santos M, Smith CA, McAinsh AD and Millar JBA. (2018) Bub1 is not essential for the checkpoint response to unattached kinetochores in diploid human cells. Current Biology 28, 929-930.
Meadows JC, Messin LJ, Kamnev A, Lancaster TC, Balasubramanian MK, Cross RA and Millar JB. (2018) Opposing kinesin complexes queue at plus tips to ensure microtubule catastrophe at cell ends. EMBO Reports 2018 doi: 10.15252/embr.201846196.
Meadows, J.C., Lancaster, T.C., Buttrick, G.J., Sochaj, A.M., Mora Santos, M., Messin, L., Mora Santos, M.d.M., Hardwick, K.G. and Millar, JB. (2017) Identification of a Sgo2-dependent but Mad2-independent pathway controlling anaphase onset in fission yeast.Cell Reports 18, 1422-1433.
Mora-Santos MD, Hervas-Aguilar A, Sewart K, Lancaster TC, Meadows JC, Millar JB. (2016) Bub3-Bub1 binding to Spc7 (KNL1) toggles the spindle checkpoint switch by licensing interaction of Bub1 with Mad1-Mad2 Current Biology 10, 2642-2650
Silió, V., McAinsh, A.D, Millar, JB. (2015) KNL1-Bubs and RZZ Provide Two Separable Pathways for Checkpoint Activation at Human Kinetochores. Developmental Cell 35, 600-13.

