Masanori Mishima
Technical Summary
Animal cells execute cytokinesis by two steps of cleavage of the cell membrane, namely by cleavage furrow ingression by acto-myosin contractile ring and by secondary ingression at the intercellular bridge for abscission of the daughter cells. Microtubule-based structures formed after anaphase onset such as the central spindle and midbody play crucial roles in regulation of these processes especially in spatial and temporal coordination with chromosome segregation.
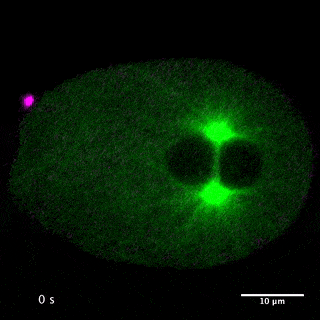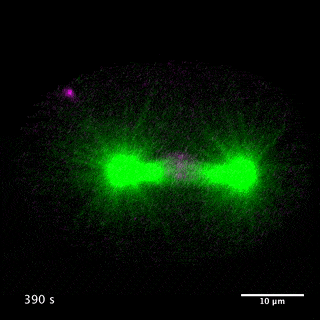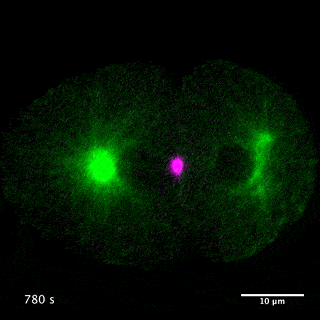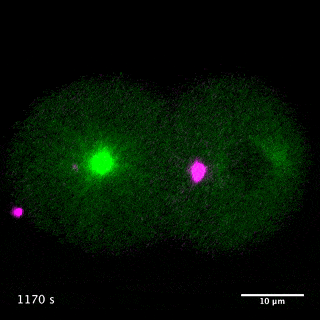 Our laboratory has been studying the mechanisms for the formation and function of these structures using C. elegans and mammalian cultured cells. We have been focusing on centralspindlin and PRC1, two microtubule bundling proteins crucial for the formation of the central spindle and midbody, which also work as key hubs in the protein-protein interaction network for cytokinesis signalling at the centre of these structures. Centralspindlin is the stable heterotetrameric complex of MKLP1 kinesin and CYK4 GAP for Rho-family GTPases. Its accumulation to the spindle midzone is a key spatial cue for contractile ring formation.
Our laboratory has been studying the mechanisms for the formation and function of these structures using C. elegans and mammalian cultured cells. We have been focusing on centralspindlin and PRC1, two microtubule bundling proteins crucial for the formation of the central spindle and midbody, which also work as key hubs in the protein-protein interaction network for cytokinesis signalling at the centre of these structures. Centralspindlin is the stable heterotetrameric complex of MKLP1 kinesin and CYK4 GAP for Rho-family GTPases. Its accumulation to the spindle midzone is a key spatial cue for contractile ring formation.
We have revealed the molecular mechanisms for this specific localisation of centralspindlin in terms of its molecular structure and higher-order assembly as well as of its cellular regulations by phosphorylations and protein-protein interactions. We have also demonstrated the importance of the direct interaction between centralspindlin and PRC1 on the robust formation of the central spindle when it is under mechanical stress. By combination of C. elegans genetics, live imaging and in vitro assays, we are trying to better understand the mechanisms for robust execution of cytokinesis.
While centralspindlin is essential for cytokinesis in all the animal cells examined so far, in collaboration with the group of Dr. Irina Golovleva at Umea, Sweden, we have discovered that a point mutation of MKLP1 can cause a human genetic disease, congenital dyserythropoietic anaemia type III (CDA III). This highlights the importance of robust execution of cytokinesis for human health. To better understand the disease mechanisms of CDA III and obtain more general insights about the consequences of infrequent failure of cytokinesis in vertebrates, we will generate a zebrafish model of this disease.
Selected publications:
- Wontakal, S.N., Britto, M., Zhang, H., Han, Y., Gao, C., Tannenbaum, S., Durham, B.H., Lee, M.T., An, X., Mishima, M. (2022) RACGAP1 variants in a sporadic case of CDA III implicate the dysfunction of centralspindlin as the basis of the disease. Blood, 139:1413-1418. doi: 10.1182/blood.2021012334. PMID: 34818416
- Maruyama, Y., Sugawa, M., Yamaguchi, S., Davies, T., Osaki, T., Kobayashi, T., Yamagishi, M., Takeuchi, S., Mishima, M., Yajima, J. (2021) CYK4 relaxes the bias in the off-axis motion by MKLP1 kinesin-6. Commun Biol., 4:180. doi: 10.1038/s42003-021-01704-2. PMID: 33568771
- Chapa-Y-Lazo, B., Hamanaka, M., Wray, A., Balasubramanian, M.K., Mishima, M. (2020) Polar relaxation by dynein-mediated removal of cortical myosin II. J Cell Biol., 219:e201903080. doi: 10.1083/jcb.201903080. PMID: 32497213
- Capalbo, L., Bassi, Z.I., Geymonat, M., Todesca, S., Copoiu, L., Enright, A.J., Callaini, G., Riparbelli, M.G., Yu, L., Choudhary, J.S., Ferrero, E., Wheatley, S., Douglas, M.E., Mishima, M., D'Avino, P.P. (2019) The midbody interactome reveals unexpected roles for PP1 phosphatases in cytokinesis. Nat Commun., 10:4513. doi: 10.1038/s41467-019-12507-9. PMID: 31586073
- Lee, K.Y., Esmaeili, B., Zealley, B., Mishima, M. (2015) Direct interaction between centralspindlin and PRC1 reinforces mechanical resilience of the central spindle. Nat Commun., 6:7290. doi: 10.1038/ncomms8290. PMID: 26088160

