How can we store electricity?
How can we store electricity?
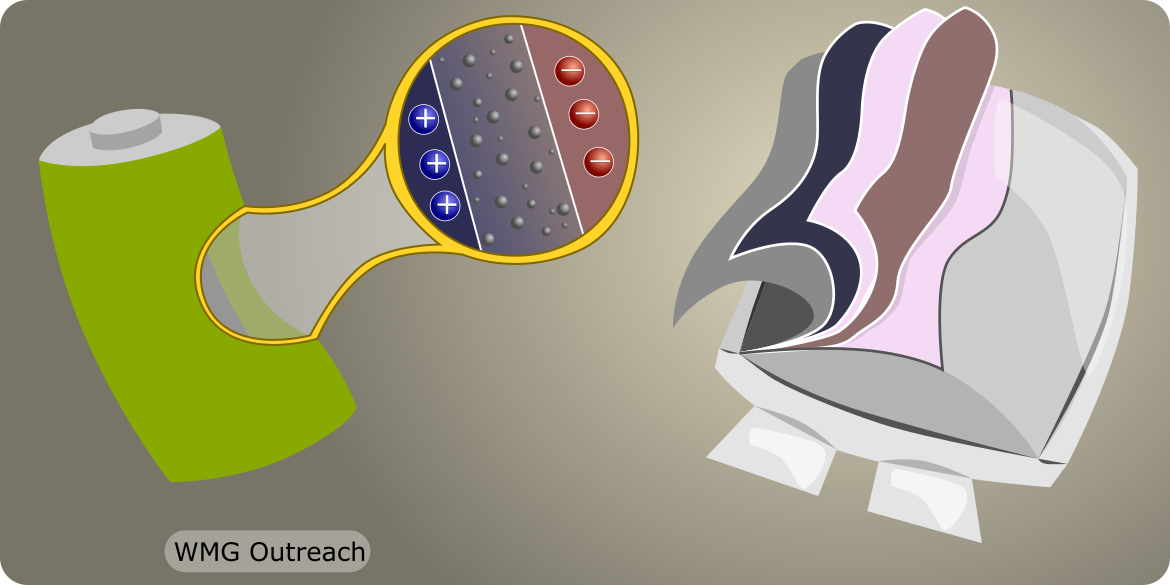
How can these batteries - either pouches or cylinders of metal - store up so much energy?
 Introduction
Introduction
Great, so now we know how we get the power to push the vehicle along. But as we described the electrons going through the motor we mentioned that they gave a bit of energy to the motor, didn’t we? Where does all this energy come from? We know that the electrons’ charge can’t change and the same number of electrons has to end up on each side of the circuit to keep things balanced. So what actually happens to provide the energy in this circuit?
Batteries
Batteries can store energy. Think of a way that you could use some energy to set something up and then release the energy again.
- Set up a domino run – you give the energy to make the blocks stand up. Knocking them over releases the energy you gave and the blocks fall back down.
- Place a ball on a table – you give the energy to raise the ball off the floor. Rolling it over the edge releases that energy and the ball falls.
- Stretch an elastic band – you provide the energy to stretch the material out and letting go will ping it back to shape and release that energy.
|
In each case we can give energy to the system and then release it again. That’s all a battery is! This is why is so dangerous to use damaged batteries because if they do crack open all the energy inside them will be released at once. But if we could safely look inside a battery, what would it look like? Would a pile of electrons just fall out? |
 |
 Focus
Focus
Building a battery
You can actually build a battery yourself using items that a lot of people have at home, believe it or not. It's actually a really common science fayre project to build a 'Potato Clock' which turns the potato itself into a battery! WMG tend to do it with lemons instead. All you need is a piece of copper (like a 1p or 2p coin) and a material coated in zinc - some galvanised screws will do this so raid the shed/garage - after asking for help) and when you push these metals into the lemon and then connect them up into a circuit there will be some electricity flowing!
These lemon batteries do work but they have very little power! Only just enough to light a tiny LED.
So do lemons store up loads of electricity?
No.
The potato battery, lemon battery, or any other type of battery made with household items like this only works because of the bits of metal you shove into the fruit/vegetable. It's actually those metals doing all of the work! Electrons move from the zinc metal, through the lemon, into the copper. That's where the electricity in the lemon battery comes from - the interaction of the copper and the zinc metals.
Energy storage materials
The battery is filled with very special chemical materials. They can exist in a couple of different forms, each with a different amount of energy. When we charge a battery we move the atoms and molecules around in the battery and push them from the low energy form into the high energy. This is like stretching up the elastic band. When we release the elastic band – or provide a circuit for the electrons to flow around from one end of the battery to the other – the energy can be released and the materials return to the lower energy form.
Battery capacity
If we want our car to travel further we need to be able to provide the energy to the motor for longer. We need to be able to have more electrons rushing through the circuit carrying their little bit of energy with them.
This means we either need to have a material in the battery that can store more energy per electron – so the high energy form is even more energetic than the low energy form (like stretching the elastic band further and further), or we need to have more of the material itself.
Unfortunately more material makes the battery heavier so the ideal situation is to have a really big difference between the high energy state and the low energy state. Think back to the elastic band though… what happens if it stretches too far? Snap.
Video of battery failure
When batteries snap they really do go for it. All the energy inside the battery can be released at once with explosive results! We have to be very careful about how hard we push the batteries during use to stay safe.
 Video
Video
You don’t just have to take my word for it – let’s hear what some of WMG’s battery experts think about electric vehicles.
 Conclusion
Conclusion
Batteries are made up of layers of electrodes with electrolyte in between. When we charge a battery we move the atoms and molecules around into an ordered, high energy arrangement. When we discharge the battery - that is to say when we use it - we move electrons around a circuit and balance that charge by moving ions inside the battery. Battery technology will help us to electrify transport and that will help us to meet our Climate Change targets and be Carbon Neutral by 2050.

 Introduction
Introduction  Focus
Focus  Video
Video  Conclusions
Conclusions  What's next?
What's next?  Authors
Authors 
