News
A Golden Window Electrode for Organic Solar Cells
The Hatton group together with Professor Tim Jones report the development of a rapid, solvent free method for the fabrication of large area gold films with a thickness of only 8 billionths of a metre, in Advanced Functional Materials: http://dx.doi.org/10.1002/adfm.201002021. These films combine high optical transparency and electrical conductivity with a remarkably low surface roughness and are exceptionally robust towards ultra-sonic agitation in a range of common solvents. By incorporating a random array of circular apertures into the films using microsphere lithography the team also show how the optical properties can be optimized for application as the transparent electrode in organic solar cells. These ultra-thin films are potentially widely applicable for a variety of applications, where stable, chemically well-defined, ultra-smooth transparent electrodes are required such as in the emerging fields of nanoelectronics and nanophotonics.


Bon Lab features on the cover of Polymer Chemistry
 |
A paper by PhD student Nicholas Ballard and associate professor Stefan Bon, previously selected by the Royal Society of Chemistry as a hot paper, features on the April 2011 cover of Polymer Chemistry. The paper entitled Hybrid biological spores wrapped in a mesh composed of interpenetrating polymer nanoparticles as "patchy" Pickering stabilizers describes a new method for the decoration of the intricate morphology of spore particles with polymer nanoparticles and investigates their behaviour at liquid–liquid interfaces. It is found that a large difference in the interfacial activity between spherical microspheres and the anisotropic particles synthesized exists, which is explained in terms of particle wettability. read the paper ; more on the BonLab |
Graphene oxide’s solubility disappears in the wash
|
Drs Rourke and Wilson’s team made their discovery when treating the graphene oxide with sodium hydroxide (NaOH) in an attempt to increase the usefulness of the oxygen containing functional groups believed to be bound to the graphene. Unfortunately it seemed to make things worse rather than better. Indeed at high enough concentrations of NaOH Dr Rourke was left with a black suspension. The Warwick led researchers recalled that it had been shown that oxidation debris adheres to carbon nanotubes but the weak nature of the connection of this oxidation debris to the carbon nanotubes meant that a wash with a base can simply remove the oxidative debris. Experiments showed that in that particular case oxidative debris was found to make up almost a quarter of the mass of the “oxidized carbon nanotubes”. The researchers felt a similar process maybe happening in the Graphene Oxide they were studying. The results may also help explain the inordinately high levels of oxygen people were claiming to find in graphene oxide. Chemists were already struggling to identify enough plausible carbon to oxygen bonds to accommodate the amounts of oxygen believed to form part of graphene oxide.
The remaining liquid was also dried to give a white powder that the Warwick researchers showed contained the “oxidative debris” or OD; the OD was shown to be made up exclusively of small, low molecular weight compounds (i.e. less than 100 atoms). The graphene oxide recovered from washing process formed about 64% of the mass of the “graphene oxide” at the start of the process. The recovered OD or oxidative debris formed at least 30% of the weight of the mass of the original “graphene oxide”. Drs Rourke and Wilson’s team believe this shows that much of the oxygen that was believed to be closely bonded to the carbon in the graphene oxide was actually not bonded at all but simply lying on top of the graphene sheets, loosely connected to them as “oxidative debris”. This oxidative debris contained a large quantity of oxygen that simply came out in the wash when the graphene oxide was treated with sodium hydroxide. This creates a significant proble Drs Rourke and Wilson say “Our results suggest that models for the structure of graphene oxide need revisiting. These results have important implications for the synthesis and application of chemically modified graphene particularly where direct covalent functionalization of the graphene lattice is required.” The paper entitled: The Real Graphene Oxide Revealed: Stripping the Oxidative Debris from the Graphene-like Sheets by Dr. Jonathan P. Rourke, Priyanka A. Pandey, Joseph J. Moore, Matthew Bates, Neil R Wilson (all of the University of Warwick), and Dr Ian A. Kinloch, Prof. Robert J. Young (The University of Manchester), has just been published in Angewandte Chemie DOI: 10.1002/anie.201007520. Notes for editors: The researchers thank Dave Hammond for help with thermogravimetric analysis (TGA), Lijiang Song for help with mass spectrometry, and Ajay Shukla for help with X-ray photoelectron spectroscopy (XPS), the Midlands Physics Alliance Graduate School for a scholarship. The TEM, TGA, and XPS instruments as well as the mass spectrometer used in this research were purchased with support from Advantage West Midlands (part funded by the European Regional Development Fund) as part of the Science City programme. For further information please contact: Dr Jonathan P. Rourke Peter Dunn, Head of Communications PR23 8th March 2011 |
Inorganic Materials book series
The fifth volume, Energy Materials, co-edited by Richard Walton, with Duncan Bruce (York) and Dermot O’Hare (Oxford), has been published this week by Wiley.
http://eu.wiley.com/WileyCDA/WileyTitle/productCd-0470997524.html

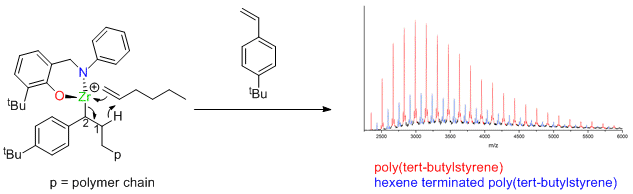
Zirconium catalysts not dead, just resting.
Dormant polymerization catalysts are given a rude awakening by Giles Theaker, Peter Scott and Warwick Chemistry Alumnus Colin Morton at Infineum. The results indicate how many old catalysts, thought to be dead, are just in a dormant state. The results published in the American Chemical Society journal Macromolecules describe a new mechanism for polymerization of styrenes.
JACS cover for Challis group
Prof. Greg Challis and his team together with Thomson and co-workers feature on this weeks cover of the Journal of the American Chemical Society. The absolute and relative stereochemistry of streptorubin B, a brightly colored prodiginine antibiotic, has been determined. Challis and co-workers utilized a mutant of Streptomyces coelicolor to conduct a mutasynthesis using enantioenriched deuterium-labeled biosynthetic precursors, while Thomson and co-workers developed an enantioselective total synthesis via a 10-membered pyrrolophane intermediate. See Challis and co-workers, p 1793, and Thomson and co-workers, p 1799. View the article.

Understanding Stimuli-Responsive Biomaterials
Matthew Gibson and collaborators investigate in detail the behaviour of stimuli-responsive polymer-protein conjugates in Polymer Chemistry. It is shown that at in vivo concentrations and when measured in blood rather than water, the behaviour of these materials deviates significantly from what is normally expected.
and collaborators investigate in detail the behaviour of stimuli-responsive polymer-protein conjugates in Polymer Chemistry. It is shown that at in vivo concentrations and when measured in blood rather than water, the behaviour of these materials deviates significantly from what is normally expected.
Konstantinos Bebis, Mathew W. Jones, David M. Haddleton and Matthew I. Gibson*. Polymer Chemistry, 2011, DOI: 10.1039/C0PY00408A
Read how the Bonlab armors polymer vesicles with colloids in JACS
|
The ability of some forms of plankton and bacteria to build an extra natural layer of nanoparticle-like armour has inspired chemists at the University of Warwick to devise a startlingly simple way to give drug bearing polymer vesicles (microscopic polymer based sacs of liquid) their own armoured protection. The Warwick researchers have been able to decorate these hollow structures with a variety of nanoparticles opening a new strategy in the design of vehicles for drug release, for example by giving the vesicle “stealth” capabilities which can avoid the body’s defences while releasing the drug. Advances in polymerisation have led to a surge in the creation of vesicles made from polymer molecules. Such vesicles have interesting chemical and physical properties which makes these hollow structures potential drug delivery vehicles. The University of Warwick team were convinced that even more strength, and interesting tailored properties, could be given to the vesicles if they could add an additional layer of colloidal armour made from a variety of nanoparticles.
“We took our inspiration from nature, in how it adds protection and mechanical strength in certain classes of cells and organisms. In addition to the mechanical strength provided by the cytoskeleton of the cell, plants, fungi, and certain bacteria have an additional cell wall as outermost boundary. Organisms that particularly attracted our interest were those with a cell wall composed of an armour of colloidal objects – for instance bacteria coated with S-layer proteins, or phytoplankton, such as the coccolithophorids, which have their own CaCO3-based nano-patterned colloidal armour” The Warwick researchers hit on a surprisingly simple and highly effective method of adding a range of different types of additional armour to the polymer based vesicles. One of those armour types was a highly regular packed layer of microscopic polystyrene balls. This configuration meant the researchers could design a vesicle which had an additional and precise permeable reinforced barrier for drug release, as a result of the crystalline-like ordered structure of the polystyrene balls. The researchers also succeeded in using the same technique to add a gelatine-like polymer to provide a “stealth” armour to shield vesicles from unwanted attention from the body’s immune system while it slowly released its drug treatment. This particular coating (a poly((ethyl acrylate)-co-(methacrylic acid)) hydrogel) absorbs so much surrounding water into its outer structure that it may be able to fool the body’s defence mechanism into believing it is in fact just water.
The research has just been published in a paper entitled Polymer Vesicles with a Colloidal Armor of Nanoparticles by Rong Chen, Daniel J. G. Pearce, Sara Fortuna, David L. Cheung, and Stefan A. F. Bon* Department of Chemistry, University of Warwick in the current Journal of the American Chemical Society http://dx.doi.org/10.1021/ja110359f Note for Editors: The cryo electron microscope used for the research have been funded by the Science City Research Alliance (SCRA) which is part of a larger investment by Advantage West Midlands and ERDF in the research infrastructure of the West Midlands region, which unites the University of Warwick and the University of Birmingham and the in a strategic research partnership – SCRA – formed under the Birmingham Science City initiative. Birmingham Science City, funded by Advantage West Midlands, is a region-wide partnership of public sector, businesses and the research base, which is facilitating the use of science and technology to improve the quality of life and prosperity of the West Midlands. For further information please contact: Peter Dunn, Head of Communications, Communications Office, PR10 31st January 2011 |
|
Dr Jósef Lewandowski joins Warwick Chemistry
We are delighted to announce that Dr Jósef Lewandowski will be joining us as Assistant Professor in Physical Chemistry from 1 September 2011.
Dr Lewandowski's research interests are primarily in the area of solid state NMR.
Jósef comes to us from the European Center for high Field NMR in Lyon, France where he is currently completing a postdoctoral stay with Prof Lyndon Emsley.
Walton group's research highlighted on the front cover of Journal of Applied Crystallography
Recent work by the group of Richard Walton in collaboration with the group of Professor Pam Thomas of the Department of Physics at Warwick (Lethbridge et al. J. Appl. Crystallogr. 43 (2010) 168-175 ) has been selected as the cover image for the Journal of Applied Crystallography for 2011. The research involved the synthesis of usually large crystals of microporous zeolites, whose behaviour on heating and cooling was then examined using birefringence microscopy. This allowed new insights into the materials’ stability and structure as a function of temperature, including the migration of organic guest species through their structures.

Rachel O'Reilly and Andrew Dove guest edit themed issue of Polymer Chemistry
Rachel O'Reilly and Andrew Dove introduce the first themed issue of the journal Polymer Chemistry by the RSC (Royal Society of Chemistry) as guest Editors. The issue contains 2 reviews, 4 communications and 18 full papers of work by emerging investigators in the area of polymer chemistry. You can read this special issue of the journal here.
Scott Group detects chiral building blocks for new materials
Pairs of building blocks are shown using spectroscopic and electrochemical techniques to associate in solution before forming new types of charge transfer material relevant to the search for chiral conductors. The work forms part of the PhD research by former Warwick Postgraduate Research Fellow , Dr Nikola Chmel
, Dr Nikola Chmel .
.
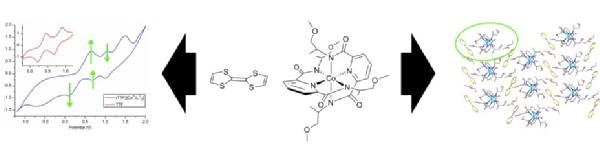
read the paper: http://dx.doi.org/10.1039/C0DT01184C






