Introduction to Semiconductors
Essence of a semiconductor
A semiconductor can be considered a material having a conductivity ranging between that of an insulator and a metal. A crucial property of semiconductors is the band gap; a range of forbidden energies within the electronic structure of the material. Semiconductors typically have bandgaps ranging between 1 and 4 eV, whilst insulators have larger bandgaps, often greater than 5 eV [1]. The thermal energy available at room temperature, 300 K, is approximately 25 meV and is thus considerably smaller than the energy required to promote an electron across the bandgap. This means that there are a small number of carriers present at room temperature, due to the high energy tail of the Boltzmann-like thermal energy distribution. It is the ability to control the number of charge carriers that makes semiconductors of great technological importance.
Semiconducting materials are very sensitive to impurities in the crystal lattice as these can have a dramatic effect on the number of mobile charge carriers present. The controlled addition of these impurities is known as doping and allows the tuning of the electronic properties, an important requirement for technological applications. The properties of a pure semiconductor are called 'intrinsic', whilst those resulting from the introduction of dopants are called 'extrinsic'. This introduction of dopants results in the creation of new, intra-band, energy levels and the generation of either negative (electrons) or positive (holes) charge carriers. More detail on doping can be found on the electronic bandstructure webpage.
Semiconductor materials
Crystal structure
Clicking on a title will reveal the relevant images for that crystal structure. More information on crystallography can be found on this page.
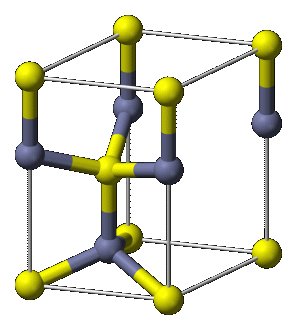
Cubic Structures
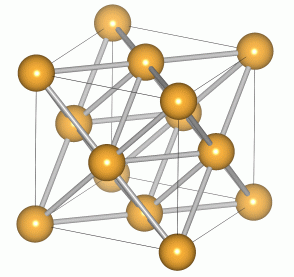
Cubic structures have the simple property that their unit cells take the shape of a cube. There are three main variants of the cubic crystal system: the simple cubic, the bodycentered cubic (BCC) and the face-centred cubic (FCC) structures. Of greatest interest is the face-centred cubic, as several derivatives of this structure are found amongst semiconducting materials.
The FCC lattice features an atom at the corners of the unit cell, in addition to one at the centre of each face. This structure is often adopted by metals as it maximises their co-ordination number.
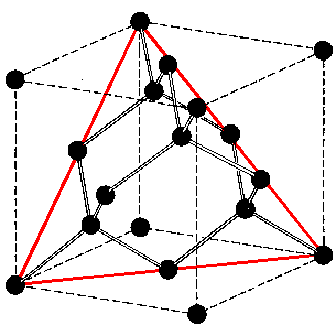
The strong covalent bonds that carbon forms with itself result in the tetrahedrally-bonded diamond structure. Each diamond atom is bonded to four neighbours and has a co-ordination number of 8. Diamond is the prototype material, however, other Group IV elements (Si, Ge and Sn) also have this structure.
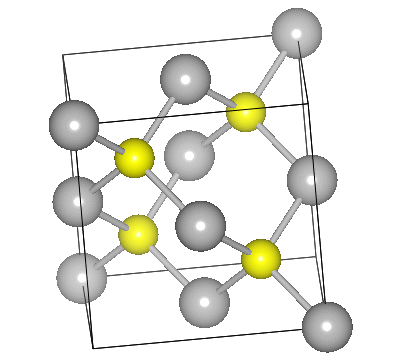
As with silicon and germanium, the III-V compound semiconductors form bonds with covalent characteristics. This results in a struture very similar to that of diamond, however, in diamond each carbon atom is bonded to another carbon atom. In the zincblende structure each atom is bonded to a different species of atom and results in a mixed lattice. The zincblende structure consists of two interpenetrating FCC lattices, where one lattice is offset by 1/4 of the unit cell. One type of atom occupies one set of lattice positions and the other species the second lattce.
A large number of III-V compound semiconductors adopt this structure, including AlAs, GaAs, GaP, InP and ZnSe.
Hexagonal Structures
Hexagonal structures have the property that they have hexagonal symmetry, with the unit cell assuming the shape of a rhombus.
Many III-V compound semiconductors can also be grown in a strucural phase known as wurtzite and is the hexagonal analogue to the zincblende structure.
References
- H. P. Myers, Introductory Solid State Physics, Taylor & Francis, 1990
Bibliography
- J.R. Hook and H.E. Hall (1991). Solid State Physics (2nd ed.) Wiley, Manchester Physics Series . ISBN 0-471-92805-4
- Charles Kittel (2005). Introduction to Solid State Physics (8th ed.). New York: Wiley. ISBN 0-471-11181-3
- Turley, Jim (2002). The Essential Guide to Semiconductors. Prentice Hall PTR. ISBN 0-13-046404-X.
- Yu, Peter Y.; Cardona, Manuel (2004). Fundamentals of Semiconductors : Physics and Materials Properties. Springer. ISBN 3-540-41323-5.
- John H. Davies (1998). The Physics of Low Dimensional Semiconductors - An Introduction Cambridge University Press. ISBN 0-521-48491-X
- Sze, Simon M. (1981). Physics of Semiconductor Devices (2nd ed.). John Wiley and Sons (WIE). ISBN 0-471-05661-8.
- Ben G. Steetman and Sanjay Kumar Banerjee (2006), Solid State Electronic Devices, Pearson Education Inc. ISBN 0-13-149726-X
- H. P. Myers (1990), Introductory Solid State Physics, Taylor & Francis. ISBN 0-85066-761-5