Andrew McAinsh
Technical Summary
Cell division is a beautifully choreographed mechanical process in which the cell constructs a microscopic microtubule-based structure called the mitotic spindle. This facilitates physical segregation of chromosomes into daughter cells. Our work is focused on the kinetochore - an adaptive, multi-layered mechanochemical machines that assemble at the centromere of each sister chromatid and engage on their outer face with the plus ends of k-fibres, microtubule bundles that emanate from the spindle poles.
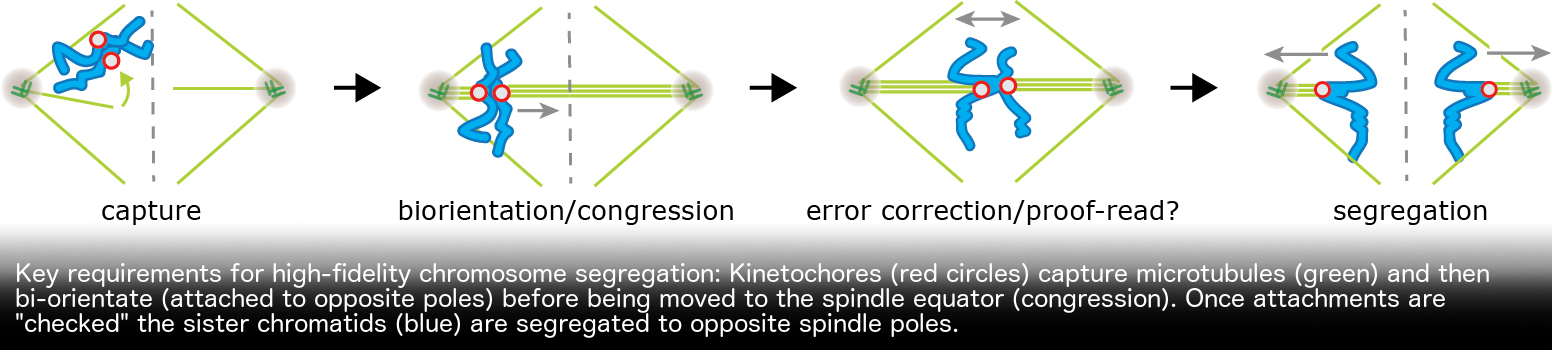 We are working to address three key questions in the cell division field: 1) how do kinetochore-microtubule interactions generate the pushing and pulling forces nessessary for chromosome movement? 2) How do kinetochores sense changes in attachment and convert this into checkpoint signals 3) What is the origin of chromosome segregation errors in human mitosis and meiosis? Approaches in the McAinsh lab include live-cell microscopy, computational image analysis, mathematical modelling and in vitro reconstitution.
We are working to address three key questions in the cell division field: 1) how do kinetochore-microtubule interactions generate the pushing and pulling forces nessessary for chromosome movement? 2) How do kinetochores sense changes in attachment and convert this into checkpoint signals 3) What is the origin of chromosome segregation errors in human mitosis and meiosis? Approaches in the McAinsh lab include live-cell microscopy, computational image analysis, mathematical modelling and in vitro reconstitution.
We study these questions in both human somatic cells, and also in human Eggs & Embryos which enables us to ask fundamental questions concerning how aneuploidy shapes pre-implantation development of human beings.
Our work on chromosome segregation is supported by Wellcome and recognised by award of the Hooke Medal in 2018 and Royal Society Wolfson Research Merit Award in 2015.
Latest works:
Nat Commun. 2022 Nov 8;13(1):6755. doi: 10.1038/s41467-022-34294-6. PMID: 36347869 Free PMC article.
Dev Cell. 2021 Dec 20;56(24):3405. doi: 10.1016/j.devcel.2021.11.023.
Ensemble-Level Organization of Human Kinetochores and Evidence for Distinct Tension and Attachment Sensors.
Cell Rep. 2020 Apr 28;31(4):107535. doi: 10.1016/j.celrep.2020.107535. PMID: 32348762
Auckland P.A., Roscioli E. and McAinsh, A.D. (2020)
CENP-F stabilizes kinetochore-microtubule attachments and limits dynein stripping of corona cargoes.
Journal of Cell Biology 219:1623-1639 PMID: 32207772
New Technology:
CelFDrive: Artificial Intelligence assisted microscopy for automated detection of rare events
Review articles:
AD McAinsh, GJPL Kops (2023)
Principles and dynamics of spindle assembly checkpoint signalling
Nature Reviews Molecular Cell Biology 24 (8), 543-559
Annu Rev Genet. Nov 30;56:279-314. doi: 10.1146/annurev-genet-072820-034559. PMID: 3605565

