Victor Zammit
We are interested in three major enzyme systems which are all involved in the partitioning of fatty acid metabolism, and particularly in the sensing of the structure and composition of the membranes in which they occur. Carnitine palmitoyltransferases (CPT1A, -1B, and -1C) are polytopic proteins in which the N- and C-terminal parts of the molecules interact to different degrees depending on the composition and curvature of the membranes in which they occur. An extensive series of studies in out laboratory has established that this enables the properties of the membrane (particularly the mitochondrial outer membrane for CPT1A) to determine the 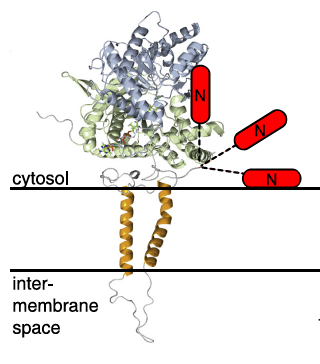 catalytic properties of the enzymes, and thus effectively to provide a mechanism through which the proteins sense the composition of the membrane which, in turn, is affected by hormonal and nutritional factors. Although CPT1 proteins have mostly been associated with mitochondrial fatty acid oxidation, the cloning and characterization of the brain-specific isoform CPT1C in our laboratory has indicated that this protein is associated with the endoplasmic reticular membrane, and has important functions in neurons; its loss-of -unction mutation results in severe ataxia in humans.
catalytic properties of the enzymes, and thus effectively to provide a mechanism through which the proteins sense the composition of the membrane which, in turn, is affected by hormonal and nutritional factors. Although CPT1 proteins have mostly been associated with mitochondrial fatty acid oxidation, the cloning and characterization of the brain-specific isoform CPT1C in our laboratory has indicated that this protein is associated with the endoplasmic reticular membrane, and has important functions in neurons; its loss-of -unction mutation results in severe ataxia in humans.
Diacylglycerol acyltransfereases 1 and 2 (DGAT1, and DGAT2) catalyse the same, final reaction in the synthesis of triglycverides, but our recent observations show that they are non-redundant and have specialised functions, In particular, DGAT2 esterifies de novo synthesised fatty acids to triglycerides, whereas DGAT1 is specialised for the synthesis of triglycerides from preformed, exogenously supplied fatty acids to cells. We have shown that DGAT2 links the metabolism of glucose to dyslipidaemia (in the liver, and to fatty acid-driven thermogenesis in brown adipose tissue. We also showed that DGAT1 in the liver is primarily concerned with the synthesis of triglycerides for secretion, and that its displays dual topology within the endoplasmic reticular membrane; the sidedness of its active site within the membrane is dependent on physiological state, and on hypolipidaemic pharmacological treatments, suggesting that DGAT1 may sense the composition of the endoplasmic reticular membrane in which it resides.
A third enzyme system that is studied in our laboratory is that of the acetyl-CoA carboxylases-1 and -2 (ACC-1 and -2). Both these isoforms catalyse the same reaction (the first reaction of fatty acid synthesis) converting acetyl-CoA to malonyl-CoA. This metabolite is the inhibitor of CPT1 (see above) that makes the latter the rate-limiting enzyme for the entry of long-chain fatty acids into mitochondria. Our research focusses on the hypothesis that the malonyl-CoA concentration in the micro-environment of CPT1 is determined by the reversible association of ACC-2 with the mitochondrial outer membrane where CPT1 is located. We have recently shown that ACC-2 is phosphorylated in an insulin-related manner at residues that may affect its reversible electrostatic association with the membrane.
Selected publications:
-
Villar, J., Ocheng, R., Gunier, R.B. et al (2022) Associatioin between fetal abdominalgrowth trajectories, maternal metabolite signatures early in pregnancy, and childhood growth and adiposity. Lancet Diabetes & Endocrinology doi. 10.1016/52213-8587(22)00215-7
-
Bonomini, M., Masola, V., Procino, G., Zammit, V., Divino-Filho, J.C., Arduini, A. and Gambaro, G., 2021. How to improve the biocompatibility of peritoneal dialysis solutions (without jeopardizing the patient’s health). International Journal of Molecular Sciences, 22(15), p.7955. doi.org/10.3390/ijms22157955
-
Lambie, M., Bonomini, M., Davies, S.J., Accili, D., Arduini, A. and Zammit, V., 2021. Insulin resistance in cardiovascular disease, uremia, and peritoneal dialysis. Trends in Endocrinology & Metabolism, 32(9), pp.721-730. doi.org/10.1016/j.tem.2021.06.001Link opens in a new window
-
Boachie, J., Adaikalakoteswari, A., Gázquez, A., Zammit, V., Larqué, E. and Saravanan, P., 2021. Vitamin B12 induces hepatic fatty infiltration through altered fatty acid metabolism. Cellular Physiology and Biochemistry, 55(3), pp.241-255. doi: 10.33594/000000368
-
Bonomini, M., Zammit, V., Divino-Filho, J.C., Davies, S.J., Di Liberato, L., Arduini, A. and Lambie, M., 2021. The osmo-metabolic approach: A novel and tantalizing glucose-sparing strategy in peritoneal dialysis. Journal of Nephrology, 34(2), pp.503-519. doi.org/10.1007/s40620-020-00804-2

