Journal Club Week 4 Answers
Follow us on twitter for updates and helpful hints! @physics_journal
A. K. Geim (2011), Nobel Lecture: Random walk to graphene, Reviews of Modern Physics, 83, 851
The paper can be found at https://journals.aps.org/rmp/pdf/10.1103/RevModPhys.83.851
Below are some of the amazing answers we've received this week from students. There are student answers to the four suggested skim-read questions as well as answers to questions thought up by students. There are lots of excellent summary answers this week too. Answers can be downloaded in a Cornell notes style here.
Special congratulations to Rugby School for having the most entries from one school!
SKIM-READ QUESTIONS
What is graphene?
Graphene is an allotrope of carbon and its atoms are hexagonally arranged. Moreover, it resembles a single layer of graphite which is one atom thick but incredibly strong as it has a large number of carbon atoms covalently bonded together. Like graphite, it conducts electricity well as it has delocalised electrons which are free to move to carry a charge.
Katie
What is microfabrication?
This is the process of making structures that are extremely small - one-millionth of a metre or even smaller. An example of when this is used in making integrated circuits or microchips
Alex
What is diamagnetism?
Diamagnetism is a type of magnetism. It is significantly different to the two other types of magnetism: ferromagnetism and paramagnetism. Both ferromagnetic and paramagnetic objects are attracted to magnetic fields. Diamagnetic materials, however, are repelled by magnetic fields. This is because when a magnetic field is applied, it creates an induced magnetic field in them in the opposite direction. This causes a repulsive force.
Adam
What is the electric field effect?
A phenomenon where applying an external electric field affects the electrical conductivity of a material. Allowing the control of the material’s electrical conductivity. In metals, the electron density is too high for sufficient penetration into the metal by an electric field, for the electric field effect. Alternatively, a semiconductor has a sufficiently lower density of electrons, allowing an electric field to penetrate for enough to alter the conductivity of the said semiconductor.
Tilen
What are two-dimensional electron gases (2DEGs)?
A 2DEG is an electron gas that is free to move in two dimensions, but tightly confined in the third. This tight confinement leads to restricted energy levels for motion in the third direction – the electrons can only move in 2D. Thus, the electrons appear to be a 2D sheet embedded in a 3D world: they can move freely in two directions but are restricted in the third direction.
Ayesha
SUMMARY QUESTIONS
What makes graphene such a momentous material?
Graphene is a momentous material as not only does it exhibit astonishing physical and electronic properties, but it also displays exotic new physics that may help us with discoveries in other fields such as superconductivity. For example, graphene is the strongest material ever discovered and displays an exceptional electronic quality in terms of charge carrier mobility and has a remarkable electric field effect. Some of the physics behind the electronic properties can be deduced by the fact that the charge carriers in a monolayer are massless Dirac fermions whereas they are massive in a graphene bilayer. Recent research with the material is proving to show new discoveries, for example recently there was the discovery of the superconducting state in twisted bilayer graphene. Uncovering the origin of this could help understand the physics at play in high temperature superconductors and allow us to produce superconductors that operate at near room temperature.
Jared
I believe graphene is a momentous because of its versatility. It has novel uses in many different industries, from electronics to membranes for filtration. This versatility, combined with its high strength, and that the described tape method is simple enough to isolate that “even school children could probably do it”, makes it a significant discovery that could prove extremely useful in the future. In a world so rapidly technologically advancing, graphene may be a substance that will become widely used, as its numerous qualities outperform those of substances that we use at the moment – for example, it’s qualities as a semi-conductor could mean it replaces silicon in transistors and integrated circuits.
Johan
Which piece of Geim’s research stood out most to you? Summarise it in three sentences or less.
Although Geim doesn't consider it his real 'eureka moment', I think there is a lot to be said for his use of sellotape to procure thin samples of graphene, rather using the polishing technique. I imagine the constant pressures on scientists to publish meaningful research creates a stressful environment in which little value is given to reflecting on methods that are already used and accepted, meaning that thinking 'outside the box' is rare. For me, the importance of Geim's discovery is less the discovery itself and more the idea that taking time to consider problems from other viewpoints can be extremely beneficial and could even potentially lead to a Nobel Prize.
Kitty
Geim’s work with water in incredibly powerful magnets was based around simple curiosity, yet led to an extension of the principles of diamagnetism. This idea that a sufficient magnetic field could overcome the gravitational force of the water wasn’t a completely new idea, but by playing around with it in a way that others hadn’t, Geim allowed new applications.
Acacia
The piece that stood out most for me was when Gein and Dubonos used inspiration from an animal to create a material to help with their research (a gecko’s feet) in ‘Friday night experiments’. Even though in did not work as well as they hoped, it still inspired further work in the field.
Alice
The precise measurements of the electronic properties of graphene, which caused Geim and Novoselov’s research to win them a Nobel prize, had to be taken with very sensitive and elaborate microfabricated devices. However, the potential of graphene was discovered using only scotch tape and a hand-made electronic circuit made under a microscope.
Ambre
The standout piece of research for me was when Geim described his 6 month stay at Nottingham University as he was granted a visiting fellowship. Although he could only study and research readily available devices, he considered them gold. Geim studied GaAs wires from previous experiments and although they were considered ‘exhausted’ equipment, he was able to do 100 hours of research a week which he describes as ‘relentless’ and ‘inspiring’ (this research perhaps aiding him in future endeavours).
Phinehas
What have you learned about the workings of a scientific research group from this article?
The importance of reproducibility and repeatability of an experiment, because it’s constantly thrown around in A-level physics and no one really pays attention to it, especially during practical work when many scoff at it as it makes the process more monotonous than it should be. However, that was the issue I’ve only thought about it as an A-level student doing experiments that have been already done countless times over decades or even centuries. So, reading this article finally made me see this fact through the eyes of someone who is researching something at the forefront of their field at the most professional stage.
Tilen
To me this article really highlighted the importance of collaboration to researchers in science. Geim said that he survived scientifically during his early years at Nijmegen due to help from colleagues at Nottingham and Chernogolovka, sending 2DEGs and the resulting microfabricated devices. To think, a Nobel prize winner’s scientific career may once have been in jeopardy! The international help from previous connections and colleagues seems to have been crucial for Geim’s early career. Additionally, for a lot of Geim’s ‘Friday night experiments’ he worked with PhD students as well as colleagues from the university, a lot of that work would not have been able to be done without that support, and it is these lateral thinking experiments that eventually led Geim to graphene and the Nobel prize. In most media the scientist is often portrayed as a loner, working completely independently on their research all of the time, but Geim paints a different picture in this article, one where research is a group effort and the success of one is possible due to the support of many.
Molly
I have learnt that discoveries are rarely made by a single person, moreover, it is often a team of scientists working together to achieve a common goal that allows the discoveries to be made. Furthermore, when a discovery is made, the scientific community will not be fully interested in the discovery unless sufficient study of the discovery has been made.
Thomas
Why do you think Geim believes that “Chances of success are much higher where the field is new”? Is there any reason why this might not be the case?
In new fields, there is much that is unknown and therefore would be viewed as a new discovery. This means that there will be higher chances of success as it is an uncertain field. Although it could mean that the chances of achieving successes are lower as the field will be difficult as so many variables will be unknown or uncertain. I believe that the chances of success would be higher, but it may take longer to discover something different as it is a field that is still being researched.
Devasena
I think that Geim believes in this because it can be difficult to contribute more to a field already well-explored without any new knowledge or extremely high level equipment, especially since everyone already working within that field is already an expert with similar backgrounds and approach to the subject. However, as Geim himself admits, there is often an element of chance and exploring the field further can still lead to valuable research because the conditions are always different. Furthermore, a well-established field could reveal a number of possibilities due to the new research happening at all times, and applications or insights could be discovered that have not been considered yet. To apply the reverse of this argument, there could also be cases where a new and interesting area of research could seem promising but actually can only fulfil its potential and yield success once more developments have occurred, either in our understanding of the subject or in equipment.
Angela
I believe that although chances of success are higher in a new field as whatever you discover is new it does not mean that success is certain. Einstein won his Nobel prize due to his work on the photoelectric effect which was discovered by Rudolf Hertz. Einstein did not win his prize for the ground-breaking research that he did on relativity. This shows that although Einstein had huge success in a new field his prize was gained for the work he did in another previously discovered field. Success in a new field may be higher in chance but it is also more likely to be ridiculed and rejected. It is human nature to trust the thing we already know so adding to the previous knowledge is more accepted. When Darwin published the theory of evolution is has heavily ridiculed and many rejected his finding. This shows that although you chance of success are higher in a new field simply because everything you find is new, so are your chances for failure as there is nothing to base your new discoveries on.
Izzy
Below are answers to the rest of this week's questions.
Zombie management
|
(P1, C1) ‘Web of Science soberly reveals that the papers were cited twice’. What is a citation and why is it sobering that they were only cited twice? |
A citation is a reference to a source. Every time a new journal article cites your work, it is counted up by large indexes to quantify how ‘important’ it is. It is certainly sobering to realise that five years work has amassed two citations in nearly 20years, but this is often the case with scientific research. |
|
(P1, C1) What does Geim mean by the phrase ‘zombie project’? |
Research in a field that is dead and is no longer of consequence. It should be noted that research might be considered ‘dead’ at a particular time if there’s little interest in it, but that doesn’t mean it will remain this way in the future. |
|
(P1, C1) Thinking back to week 3: ‘the superconductor served only to condense an external magnetic field into an array of vortices’. What type of superconductor must this have been? |
A type-II superconductor. Above their first critical field, a type-II superconductor will allow magnetic field lines to penetrate the bulk of the material, but only in the form of ‘tubes’ of quantised flux. A type-I superconductor in its superconducting state will never let a magnetic field penetrate its bulk (but does allow a magnetic field to penetrate on the surface a short way – the London penetration depth). |
|
(P1, C1) What does inhomogenous mean? |
Not uniform. As the magnetic field penetrates the superconductor in discrete regions, it demonstrates significant variations across the ‘sandwich’ device. |
|
(P1, C1) What is electron transport and why is it affected by a magnetic field?
|
Electron transport is simply the movement of electrons through a material (so movement on the macroscopic scale). It is affected by a magnetic field as a moving electron is a current, and around any current circulates a magnetic field. Therefore a moving electron is inherently magnetic and is affected by an applied magnetic field. |
One man’s junk, another man’s gold
|
(P1, C2) What does ‘submicron’ mean? |
Something smaller than a micrometer (1 µm=1×10-6 m) in size. |
|
(P1, C2) What is a ‘h index’? |
It’s a metric for comparing the research output of academics https://en.wikipedia.org/wiki/H-index A ‘h index’ of 1 is low. |
|
(P1, C2) What does mesoscopic mean? How does this link to Anderson’s paper More Is Different from week one? |
Mesoscopic physics studies an intermediate scale of physics in between nanophysics (on the smaller side) and studies of materials on the micrometer scale (on the larger side). It’s a realm of size where quantum mechanics begins to enter. This realm of physics is very much what Anderson was discussing when talking of emergence - the physics of systems that are on the transition between one scale (and one set of rules) and another (with a new set of rules). |
|
(P1, C2) What is a 2D electron gas? |
We can think of a 2D electron gas as a collection of electrons that are free to move around in a plane (hence 2D) but are confined from moving in the third dimension. |
|
We are probably not familiar with all of the effects and techniques that Geim mentions – quantum point contacts, resonant tunneling, the quantum Hall effect, molecular beam epitaxy, electron-beam lithography etc. but that’s okay. They’re not crucial to our understanding of the article so we can ignore them for now. It takes time to realise which bits are important and which bits we can ignore for now – we just need to gain experience at reading papers. |
|
Dutch comfort
|
(P2, C1) Why would scientists want to study the individual vortices within a type-II superconductor (think back to week 3).
|
Because the superconducting mechanism of type-II superconductors is still unknown. Scientists have been using experiments that approach the subject from every possible angle to try and understand why type-II superconductors become superconducting. Also, understanding these magnetic vortices could be important alone as they could have technological uses if they could be readily manipulated.
|
A spell of levity
|
(P2, C2) In the first paragraph of ‘a spell of levity’ what does Geim highlight about the political nature of science? |
That sometimes you have to do things to justify the existence of a facility/department. A lot of time and effort had gone into creating a 20T magnet, but it wasn’t being used (at the time) to its full potential – it is used more regularly now as the focus of condensed matter research shifts over time. |
|
(P2, C2) What advantage did Geim have in studying ‘magnetic water’? |
The strong magnetic field of the 20T magnet should make any ‘magnetic water’ effect much more prevalent than any standard magnet (whose field is typically 200times smaller). |
|
(P2, C2) What is diamagnetism? |
The effect of an external magnetic field acting on certain materials to create an opposing magnetic field within the material itself (these materials are called diamagnetic materials). This field is repelled by the original magnetic field. |
|
(P2, C2) Why did the water ‘float’ inside the 20T magnetic field? |
Water is a diamagnetic material. When an external field is applied to it, a magnetic field is created within the water in the opposite direction to the applied field. In the experimental setup that Geim had, the resulting repulsion was enough to balance the weight of small droplets of water. |
|
(P2, C2) Draw a force diagram for a droplet of water inside Geim’s magnetic field. |
|
| (P2, C2) Why can’t you see levitating water with a normal magnet? | Because the magnetic response of water is very weak (its magnetic susceptibility is 9.0x10-6) and so a standard magnet is not strong enough to create a large enough magnetic force to counterbalance the weight. |
|
(P3, C1) Why do you think a frog can also be levitated within a magnetic field? |
Since a frog is mostly water (as are most living things), they also have a diamagnetic response. If we had a magnetic field large enough, you can levitate humans too. |
Friday night experiments
|
(P3, C2) A van der Waals force is an intermolecular force – what does this mean? |
It’s a force between different molecules (not forces within a single molecule which has the term intramolecular). |
|
(P3, C2) Van der Waals forces are incredibly weak. How can such weak forces manage to suspend a gecko? |
The van der Vaals force arises between the hairs on the gecko’s feet and the surface it walks on. Whilst the force between a single hait and the surface is tiny (in the nN range), a gecko has millions of hairs and these forces add up. |
|
(P3, C2) Why would a large diamagnetic response be an indicator of possible superconductivity? (Think back to week three)
|
The Meissner effect in superconducting systems is a case of perfect diamagnetism as the applied magnetic field is prevented from penetrating the bulk. This is because superconducting currents are created on the surface which lead to an opposing magnetic field, shielding the bulk. So if we see a large diamagnetic response in a material, it may be because the material is superconducting and creating such opposing superconducting currents. |
Mancunian way
|
(P4, C1) How have Geim’s previous experiences in research shaped his attitudes to spending his grant money? |
Given that he’s worked in situations where money has been tight and resources scant, he is more than happy to forgo new buildings or rooms or large infrastructure and instead spend his money on the necessary equipment to get results. |
Three little clouds
|
(P4, C2) In terms of free electrons, what is the difference between a metal and a semiconductor? |
A metal has free electrons than can conduct electricity and a current can run even with a very small potential difference. A semiconductor does not have free electrons unless a large enough potential difference is applied to it (think of the I-V curve of a LED). |
|
(P4, C2) What is the electric field effect (also known as the field effect)? |
The alteration in electrical conductivity of a material through the application of an electric field. In a metal, the number of current carriers is large and so electric fields don’t penetrate far into a material. In a semiconductor, though, the smaller number of carriers means that the electric field can penetrate further into the material and also alter the number of carriers on the surface of the semiconductor. |
|
(P4, C2) In terms of free electrons, what is the difference between a metal and a semiconductor? |
A metal has free electrons than can conduct electricity and a current can run even with a very small potential difference. A semiconductor does not have free electrons unless a large enough potential difference is applied to it (think of the I-V curve of a LED). |
|
(P4, C2) In terms of the number of free electrons, what is the difference between a metal and a semimetal? |
Semimetals have fewer free electrons (a smaller ‘carrier concentration’) than metals. They don’t have a threshold potential difference to conduct like semiconductors, but their conductivities aren’t as large as metals. |
|
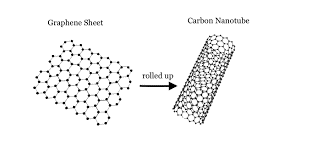
(P5, C1) What is a carbon nanotube? What is its relationship to graphene? |
A carbon nanotube is tube whose walls are a one atomic layer thick and are entirely made of carbon atoms. A carbon nanotube can be considered to be a rolled up piece of graphene.
|
|
(P5, C1) What was Geim hoping to achieve by getting his student to look into thin films of graphite? |
His main aim was to look at the electric field effect in metals (even though he knew the effect would be very small). His secondary aim was to look at some of the unusual properties of graphite to try to better understand the material.
|
Legend of Scotch tape
|
(P5, C1) What is pyrolytic graphite (or pyrolytic carbon) and what’s its advantage over natural graphite when trying to produce graphene? |
Pyrolytic carbon is a manmade form of carbon which can be understood now as individual sheets of graphene with some covalent bonding between the sheets. Thus it is much easier to cleave pyrolytic carbon into graphene sheets compared to natural graphite which crystallises into randomly oriented small domains. |
|
(P5, C1) In polishing the sample, what were they hoping to achieve? |
To polish down a piece of the pyrolytic graphite to be as thin as possible (hopefully atomically thin). |
|
(P5, C1) What might the advantage of highly oriented pyrolytic graphite be over the sample that they originally used? |
In HOPG, the crystallographic planes should be all in the same direction and the bonding between layers should be weaker than the high-density graphite (so that it’s easier to form thin samples). |
|
(P5, C2) How does sellotape create thinner samples of graphite than polishing? |
The sellotape can be used to simply peel away a layer of graphite from the larger sample. The bond formed between the graphite and the sellotape is stronger than the bond between one layer of graphite and the next, and this very gentle method allows you to simply isolate a very smaller number of layers of graphite. |
|
(P5, C2) If you’ve read any Shakespeare, compare and contrast the styles of the bard with Geim in this article so far (this is not a serious question...). |
...I've not read enough Shakespeare to do this justice. |
|
(P6, C1) What is a substrate in crystallography? |
An underlying material which is used as a surface on which to place or grow a sample that you want to be flat. |
|
(P6, C1) Why does being optically transparent indicate that a sample is thin? |
The thinner a sample is, the more likely it is that photons will pass through without scattering from the material itself. |
|
(P6, C1 and Figure 2d) Describe what is shown in Figure 2d. |
In the centre of the image, the green blob with red lines is the sample of graphene (it’s unclear whether this is atomically thin and therefor truly graphene). The four, brown crystally looking objects are the four silver contacts that Geim discusses that have been attached to look at the electrical properties of the sample. |
|
(P6, C1 and Figure 2d) 50µm is a difficult size to imagine. Find a comparison to understand how big 50µm is. |
Human hairs lie in region of thickness between 20µm and 180µm so these samples are the width of thin hairs and not much longer. Imagine trying to attach four electrical contacts to a sample that has a width, thickness and height that are all comparable to a thin hair! |
Eureka moment
|
(P6, C1) Rather than using sellotape to create graphene, what does Geim describe as his real Eureka moment? |
After noticing the electric field effect in his crude samples of graphene, the Eureka moment was to imagine what could be done if he used the ‘full arsenal of microfabrication facilities’ to make samples. |
|
(P6, C2) In the simplest of terms, what does Geim mean when he talks about moving ‘from multilayers to monolayers and from hand- made to lithography devices’. |
His samples had got thinner so they were now one layer thick instead of many layers and the electrical contacts were now made through more precise techniques (lithography). |
|
(P6, C2) How did Geim and his team ensure that any of their conclusions about the electrical properties of graphene were valid? |
They ensured that they were reproducible by only considering data that was consistent between many devices. |
|
(P6, C2) Studying 50 samples doesn’t initially sound too taxing... why is it such hard work. |
Because the samples are so small and fiddly and the measurements they are making are very precise. |
|
(P6, C2) What does Geim highlight about the review process for getting articles published? |
That it is hard work and not all referees agree with what constitutes an advancement in science. |
Defiant existence
|
(P7, C1) What does Geim mean by a continuous monolayer? |
In the case of graphene, a continuous monolayer is a piece of graphene (this is the monolayer part) that maintains a constant crystallographic structure over the size of the sample – the familiar hexagonal shape structure of graphene – rather than breaking up into different regions (called domains). Continuous monolayer:
In Figure 3, d, you can see how graphene might form little islands is different orientation if you look carefully at the largest blocks. |
|
(P7, C1) Why does a system often prefer to small regions rather than a continuous layer? |
To reduce its surface energy. Surface effects are incredibly unusual – just think about the way water can behave when it’s in small droplets (where it has a greater surface to volume ratio). |
|
(P7, C1) Why was it predicted that small, flat graphene sheets were unlikely to occur? |
Because theory told us that the atoms should prefer to exist in a 3D structure to minimise their energy. |
|
The third argument for why the existence of graphene is surprising is a little more involved so we won’t dwell on it. To grow a sample, the atoms need cool into a stable arrangement. If we're trying to grow a 2D sample, this is problematic as thermal fluctuations have a greater effect. Combined with the need for a substrate to grow it on, this limits the predicted size of graphene samples. |
|
|
(P7, C1) What is a theoretical disadvantage of graphene when it comes its existence in air? |
It has two surfaces and would therefore seem to stand more chance of being reactive in ambient conditions |
|
(P7, C1) Summarise (in as much detail as you feel comfortable with) the reasons that people would have been surprised that small, flat, continuous, monolayer sheets of graphene exist. |
|
For our purposes, we can largely ignore the section Graphene incarnations (P8 and most of P9, C1), but you may wish to read it for your own interest.
Planet graphene (The Greek letters spell ‘planet’)
|
(P9, C2) Why does Geim believe people were so interested in his paper? |
For the results of the electronic properties of graphene rather than the simpler fact of creating large monolayer samples of it. |
|
(P9, C2) Looking back, what is a typical value for the percentage change in the number of electronic carriers if a material displays the electric field effect? |
Around a 1% change in the number of carriers. Graphene surpasses this by a factor of 1000! |
|
(P9, C2) What does ‘charge carrier mobility’ mean? |
How quickly a charge carrier can move through a material when propelled by an electric field. |
|
(P9, C2) Why does Geim refer to ‘ballistic transport’ for graphene? |
Because the carriers can travel without scattering (and so travel very quickly) – charge carrier scattering is the origin of a material’s resistivity. |
|
(P9, C2) Summarise some of the remarkable electronic properties of graphene. |
It shows a huge electric field effect – by altering the potential difference across the graphene, you can tune if from having so many electronic carriers that it seems to be metallic, to having so few that it looks like a semiconductor. Remember that previously, the electric field effect would only change the number of carriers by around 1% in other materials. The graphene sheet remained conducting regardless of the contacts and surface treatments and being left in ambient conditions. The surface of materials is renowned for being reactive and terrible at conducting when exposed to air, yet graphene (despite it all being on the surface) bucks this trend. Graphene has an incredibly high charge carrier mobility. |
The section ‘Magic of flat carbon’ gives some of the newer findings about graphene’s capabilities. These are a little more specialized and aren’t discussed in enough detail for us to truly understand so we’ll leave our questions for this section.
Ode to one
|
(P10, C1) Other than its electronic properties, in what other ways in graphene remarkable? |
It displays an incredibly high strength. Graphene chemistry is made possible by its unusual bonding properties with other materials. |
|
(P10, C2) Why is graphite (and bilayers of graphene) much weaker than monolayer graphene? |
Because the layers can slide against one another. The inter-layer bonds are much weaker than the intra-layer bonds. |
|
(P10, C2) What reason does Geim give for graphite (and multilayer graphene) having significantly different electronic properties? |
Electric fields are screened by the electrons within graphite (and multilayer graphene) so that the electric field doesn’t penetrate more than a couple of layers into the material. As graphene is just a single layer, there is no such screening. |
FURTHER READING
More information on the levitation of frogs can be found here
https://physicstoday.scitation.org/doi/pdf/10.1063/1.882437
Discovery and characterisation of graphene (this is actually a paper in the journal Science but a version is available on the arXiv that does not require an academic subscription)
https://arxiv.org/pdf/cond-mat/0410550.pdf
A more detailed review of graphene by Geim and Novoselov can be found here
GENERAL INFORMATION
Remember, reading a paper isn't like reading a piece of fiction or a newspaper article. Don't get frustrated if it doesn't immediately make sense - you might need to do a little research of your own to understand some of the ideas. This article gives you an idea of how scientists read differently.
Each question refers to a specific part of the paper e.g. Page 2, Column 3 is written as (P2, C3).
Next week, we'll publish solutions to the questions and the best submitted summaries from students across the country.
NEXT WEEK
We're going to be looking at the discovery of nuclear fission. This is a rich story from a historical viewpoint as well as a scientific and technological one and we'll look into the life of one of the scientists, Lise Meitner.
This video explains, in basic terms, what Lise Meitner and Otto Hahn found. This video gives a very brief history of Lise's life. This article gives a nice overview of the link between her scientific discoveries and its relationship to history.