Jonathan Lamley
Structure and Dynamics of Proteins by Solid-State NMR
Supervisor: Dr Józef Lewandowski
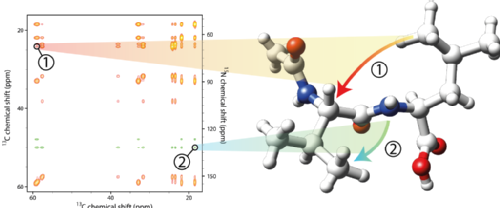
Solid-state NMR (SSNMR) is becoming a valuable tool for the characterisation of biomolecules that prove inaccessible to study by more established methods, such as solution-state NMR and x-ray crystallography. Targets such as membrane proteins and amyloid systems (linked with diseases such as Alzheimer's, Parkinson's) are key in a huge range of crucial biological processes, but often are insoluble and lack long-range order.
SSNMR offers an opportunity to study the structure and dynamics of such systems with atomic resolution in their native environment, from which conclusions regarding their complex functional mechanisms may be drawn. In practice, however, the extraction of much of this information is difficult, owing to a scarcity of observables and the inherent insensitivity of the technique.
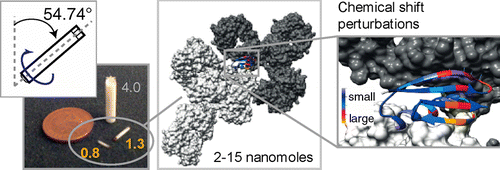
My project is aimed at the development of new NMR tools with which to probe the structural and dynamic characteristics of proteins in the solid state, and to explore links between their structure, dynamics and function. Of particular interest are "slow" motions of proteins, including anisotropic and collective motions, that are relevant to processes such as ligand binding and enzyme catalysis. I also intend to work on accelerated acquisition methods to enhance the efficiency of biological SSNMR experiments. Finally, much of my research is based on method development for the NMR study of protein complexes in the solid state. In solution, the slow molecular tumbling and correspondingly fast nuclear relaxation exhibited by large proteins and complexes inhibits their study. This limitation does not exist in SSNMR, making it an ideal candidate for studying the structures, dynamics and interactions of protein complexes that are beyond the reach of more "traditional" methods.
Publications:
Lamley, J. M.; Iuga, D.; Öster, C.; Sass, H. J.; Rogowski, M.; Oss, Andres.; Past, J.; Reinhold, A.; Grzesiek, S.; Samoson, A.; Lewandowski, J. R. J. Amer. Chem. Soc. 2014, 136, 16800.
Lamley, J. M.; Lewandowski, J. R. J. Magn. Reson. 2012, 218, 30.
Oral Presentations:
- "Protein Dynamics from Solid-State NMR Relaxation Measurements", delivered April 2013, ChemBio cluster meeting, University of Warwick, UK.
- "Applications of Fast Magic Angle Spinning to Protein NMR", delivered November 2013, Bruker user meeting, Coventry, UK.
- "Solid-State NMR of Nanomolar Quantities of Protein Complexes at 60-100 kHz Magic Angle Spinning", April 2014, 850 MHz NMR facility annual symposium, University of Warwick, UK.
- "Structure and Dynamics of Proteins by Solid-State NMR", May 2014, Chemistry postgraduate symposium, University of Warwick, UK.
Poster Presentations:
(The same poster was presented June 2012 at the RSC NMR discussion group meeting, University of Bristol, UK)
- "Protein Dynamics from Solid-State NMR Relaxation Measurements", May 2013, Department of Chemistry Postgraduate Research Symposium, University of Warwick, UK.
- "Site-Specific 13C R1ρ Relaxation Measurements in Proteins in the Solid State", September 2013, 8th Alpine Conference on Solid-State NMR, Chamonix, France.
- "Solid-State NMR of Nanomolar Quantities of Protein Complexes at 60-100 kHz Magic Angle Spinning", March 2014, 55th Experimental Nuclear Magnetic Resonance Conference, Boston, USA.
Courses Attended:
- Advanced School on Biological Solid-State NMR, October 7th-12th, Brno, Czech Republic.

Contact:
CV4 7AL