James Barnett
Research Projects 2009-2013
The dynamics of circulatory zinc handling and transport
This is a collaborative project between the Universities of St. Andrews and Warwick and is funded by the BBSRC. The work at Warwick will primarily use metalloproteomics approaches to examine the distribution of zinc in human blood plasma and how this is affected by factors such as fatty acids, that are known to affect zinc binding to serum albumin.
Metalloproteomics for marine cyanobacteria
In this project I have been developing metalloproteomics approaches to gain a better understanding of how marine cyanobacteria (using Synechococcus sp. WH8102 as a model organism) utilise zinc, and maintain cellular zinc homeostasis. Whilst focusing on zinc other important trace metal nutrients were studied including iron, copper, nickel and cobalt. A significant part of the project involved establishing robust methodologies for defining metalloproteomes including the use various protein fractionation techniques coupled with ICP-MS for metal analysis and MALDI-TOF/TOF for protein identification. An example of some of the data obtained is shown below. The 3D plots show the resolution of major iron and nickel peaks following fractionation of the soluble proteome of Synechococcus sp. WH8102 by gel filtration and anion exchange chromatography. Proteins present in these peaks are identified by standard peptide mass fingerprinting methods. 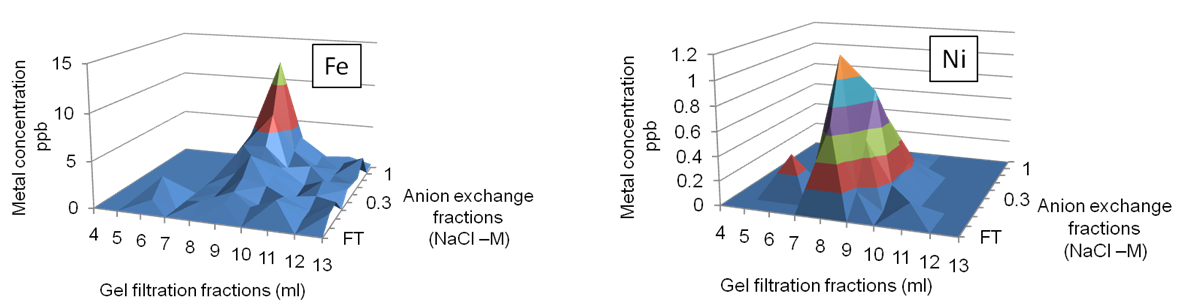
ICP-MS data showing the elution of iron and nickel following a 2-dimensional chromatographic separation on the soluble proteome of Synechococcus sp. WH8102.
Organisation and mechanism of the bacterial Twin-Arginine Translocation (Tat) pathway
I completed my PhD in the group of Prof. Colin Robinson where my research focused on understanding the mechanism of Tat-dependent protein translocation using Escherichia coli and Bacillus subtilis as model organisms for Gram-negative and Gram-positive bacteria.
The Tat (Twin-Arginine Translocation) pathway is responsible for the translocation of fully folded proteins across the bacterial plasma membrane and has been most widely studied using E. coli as a model organism. In Gram-negative bacteria 3 integral membrane proteins (TatA, TatB, and TatC) are required for translocation. These 3 proteins are organised into two major types of complex within the membrane, a substrate binding TatABC complex and seperate TatA complexes that are thought to serve as the channel through which substrates can cross the membrane.
The Tat pathway in Gram-positive bacteria exhibits some interesting differences to Gram-negative systems, most notably the absence of a separate TatB component. This suggested possible differences in translocation mechanism to Gram-negative bacteria and my work focussed on studying the B. subtilis Tat pathway to try and address this hypothesis. My work has resulted in a number of important findings and these have been published in the journal articles listed.
More recently I have developed an interest in the Tat pathway in Cyanobacteria where proteins are targeted to both the plasma and thylakoid membranes. How proteins are sorted to each membrane is not currently known. Linking in with my current research the Tat pathway has been found to be inportant for the biosynthesis of metalloproteins in cyanobacteria, by compartmentalising protein folding to the cytoplasm.
Publications
1. Barnett J.P., Blindauer C. A. , Kassaar O., Khazaipoul S., Martin E. M., Sadler P. J., and Stewart A. J. (2013) Allosteric modulation of zinc speciation by fatty acids. Biochim Biophys Acta. 12, 5456-5464.
2. van der Ploeg, R., Monteferrante, C. G., Piersma, S., Barnett, J. P., Kouwen, T.R., Robinson, C., and van Dijl, J. M. (2012) High salinity growth conditions promote Tat-independent secretion of Tat substrates in Bacillus subtilis. Appl Environ Microbiol. 78, 7733-7744.
3. Barnett, J. P., Millard, A., Ksibe, A., Scanlan, D. J., Schmid, R., and Blindauer C.A. (2012) Mining genomes of cyanobacteria for elements of zinc homeostasis. Front Microbiol Chem. 3, 1-21.
4. Barnett, J. P., Scanlan, D. J., and Blindauer C. A. (2012) Protein fractionation and detection for metalloproteomics: challenges and approaches. Anal Bioanal chem. 402, 3311-3322.
5. Barnett, J. P., Scanlan, D. J., and Blindauer C. A. (2012) Fractionation and identification of metalloproteins from a marine cyanobacterium. Anal Bioanal Chem. 402, 3371-3377.
6. van der Ploeg R., Barnett, J.P., Vasisht, N., Goosens, V. J., Poether, D. C., Robinson, C., and van Dijl, J. M. (2011) Salt-sensitivity of minimal Twin-arginine translocases. J Biol Chem. 286, 43759-70.
7. Barnett, J. P., Robinson C., Scanlan D. J., and Blindauer C (2011) The Tat protein export pathway and its role in cyanobacterial metalloprotein biosynthesis. FEMS Microbiol Lett. 325, 1-9.
8. Barnett, J. P., Lawrence, J., Mendel, S., and Robinson C (2011). Expression of the bifunctional Bacillus subtilis TatAd protein in Escherichia coli reveals distinct TatA/B-family and TatB specific domains. Arch Microbiol 193, 583-94.
9. Barnett, J. P., van der Ploeg R., Eijlander R. T., Nenninger A., Mendel S., Rozeboom R., Kuipers O. P., van Dijl JM., and Robinson C (2009). The twin-arginine translocation (Tat) systems from Bacillus subtilis display a conserved mode of complex organization and similar substrate recognition requirements. FEBS J 276, 232-43.
10. Mendel S., McCarthy A., Barnett, J. P., Eijlander R. T, Nenninger A, Kuipers O. P., and Robinson C (2008). The Escherichia coli TatABC system and a Bacillus subtilis TatAC-type system recognise three distinct targeting determinants in twin-arginine signal peptides. J Mol Biol 375, 661-72.
11. Barnett, J. P., Eijlander R. T., Kuipers O. P., and Robinson C (2008). A minimal Tat system from a gram-positive organism: a bifunctional TatA subunit participates in discrete TatAC and TatA complexes. J Biol Chem 283, 2534-42.
12. Oates J., Barrett C. M., Barnett, J. P., Byrne K. G., Bolhuis A., and Robinson C (2005). The Escherichia coli twin-arginine translocation apparatus incorporates a distinct form of TatABC complex, spectrum of modular TatA complexes and minor TatAB complex. J Mol Biol 346, 295-305.
Publications in Google Scholar
