Carotenoid Cleavage Dioxygenases
Chemical Genetics of the Carotenoid Cleavage Dioxygenases
The carotenoid cleavage dioxygenases are a family of non-heme iron-containing dioxygenase enzymes that catalyse the oxidative cleavage of carotenoid substrates. Most members of this family are found in plants, but they are also found in mammals and micro-organisms. The first identified enzyme in plants was 9-cis-epoxycarotenoid dioxygenase (NCED), which catalyses the 11,12-oxidative cleavage of 9-cis-neoxanthin, in the biosynthesis of plant hormone abscisic acid.

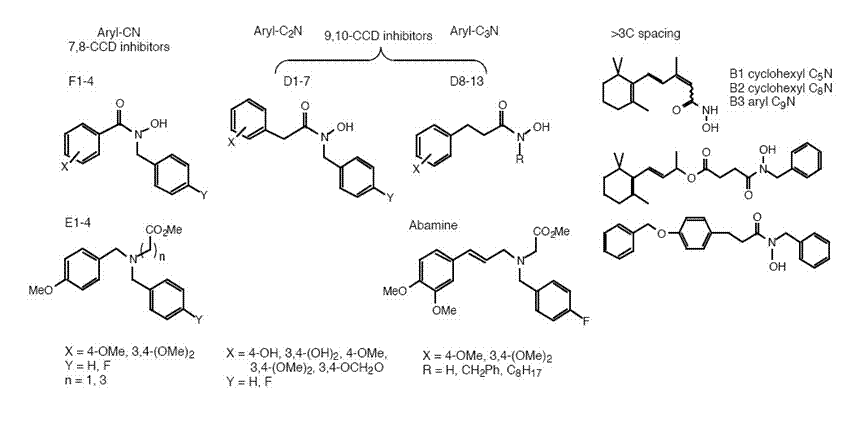
In collaboration with Dr. Andrew Thompson (Warwick HRI), we have used a chemical genetics approach to investigate the function of the carotenoid cleavage dioxygenases. Based on a model for CCD mechanism, we have synthesised two classes of CCD inhibitor: one group based upon a published inhibitor Abamine, and one group containing a hydroxamic acid metal chelator. By varying the functional group spacing in the inhibitor structure, we have prepared compounds designed to inhibit CCDs catalysing 7,8-, 9,10-, 11,12-, and 15,15’-oxidative cleavage. In vitroenzyme assays with recombinant 9,10- or 11,12-cleavage CCDs, combined with assays in carotenoid-producing E.colistrains that express various 9,10- and 15,15’-cleavage CCDs, have allowed us to identify several potent (IC500.8-2 µM) CCD inhibitors with a high degree of specificity for 9,10-cleavage.
Several of these compounds show effects upon shoot branching in Arabidopsis, comparable with a max3mutant, indicating inhibition of 9,10-cleavage in planta[M.J. Sergeant, J-J. Li, C. Fox, N. Brookbank, D. Rea, T.D.H. Bugg, and A.J. Thompson, J. Biol. Chem., 284, 5257-5264 (2009)]. We have also observed phenotypic effects of inhibitions upon flower colour in chrysanthemum and aroma in tomato, that can be rationalised by inhibition of specific CCD enzymes (unpublished results).