Keith Leppard - My Research (Public Understanding)

Welcome to my website! This page is intended to explain my research interests to people who are not trained research scientists. If you have any comments or suggestions for additions / improvements, or would like to hear more about what I do, please contact me (contact details).
A few basics:
(If you remember some biology from school, then you may not need this bit.)
All living things are made up of cells. For some species (eg most bacteria), an individual is just a single cell, but for plants and animals, each individual is built of lots of cells. These cells (with just a few exceptions) each contain a complete set of the genes of the organism, and these genes are made up of long pieces of DNA.
Each cell has some basic features in common, but in complex organisms like people, this basic pattern is adapted to give lots of different cell types. These are used to build the different organs and tissues of the body, and usually have highly specialised functions.
Viruses don't fit into this description of living things, as they are not made of cells. A virus only really has 'life' while it is within an organism that is made of cells. So, what viruses do is to infect a living organism (the host), get inside its cells and 'borrow' resources from it in order to make more virus, in other words to reproduce. While they are doing this, the host reacts to try to stop the virus growing, and between the effects of virus itself on the host and the effects of the host's reaction to the virus, the host often gets sick.
Because cells are specialised, most viruses specialise too, infecting just some types of cell and consequently causing specific disease symptoms.
About adenoviruses:
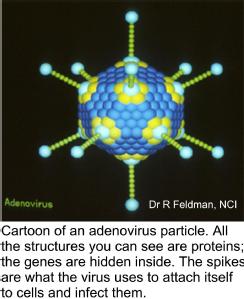
Much of the research I do is tied up with adenoviruses, so a few words of explanation about them might be useful.
Adenoviruses comprise a large family of infectious agents - there are examples able to infect just about any animal you can think of. We know most about the ones that infect people (about 60 different ones) that variously cause respiratory disease (cough/cold or more severe, including pneumonia), conjunctivitis (eye infections that can cause lasting damage), gastroenteritis (vomiting and diarrhoea) and urinary tract infections (cystitis). Some of these are a particular problem in patients undergoing organ/tissue transplants.
Despite this impressive list of diseases, most of the work done on adenoviruses is not primarily aimed at solving the health problems they cause. This is mainly because, although all these diseases are important - especially to anyone suffering from one of them, people usually recover well. Also, adenoviruses in each case are not the major cause of that type of disease - there are other viruses and bacteria that are more frequently the problem in someone suffering from these symptoms. Instead, people research adenoviruses to learn more about how the cells of the host work, or else to harness the power of the virus to make it work for us.
What are my research interests?
The work my group does encompasses both of these reasons for studying adenovirus. On one hand, we are interested in an aspect of the very early response that cells make to becoming infected, which involves a protein called PML, and on the other we are working to understand precisely how the virus uses the genes it carries during the course of an infection, so we can manipulate this behaviour to make a better gene delivery vector. If you want to know more about these, read on....
1: Understanding adenovirus to make better vectors
Getting genes (DNA) into cells efficiently has the potential to improve medicine in lots of ways. Some people suffer illness as a consequence of an inherited mutation in one of their genes. One vision is to be able to treat such people by introducing a 'good' copy of the faulty gene into their cells - gene therapy. Another possible impact is in vaccination, to protect against infections. If a gene that makes part of a dangerous virus or bacteria could be incorporated into a safe viral vector, then this could be used to protect people against the dangerous virus/bacteria. And finally, there are lots of clever ideas for treating cancer by carrying particular genes into cancer cells.

The key thing for all this is efficiency of gene delivery - getting DNA into enough cells. This is very difficult, and for most possible applications of gene delivery is still not possible. Which brings us to gene delivery vectors and making them better...
The reason why viruses can be used as gene delivery vectors is that the outer surface of the virus is already designed to attach to the surface of a cell and - crucially - to get the contents of the virus particle INSIDE the cell. Normally these contents are the genes of the virus. If you can replace some of these genes with another piece of DNA, you can destroy the ability of the virus (now termed a vector) to spread and cause disease and at the same time trick it into taking that piece of DNA into the cell instead. Without this kind of help, it is very difficult to get DNA into cells.
Adenoviruses have already been made into vectors that have seen use in the clinic. But the vectors still struggle to do enough to be effective, and one problem is that the host still 'sees' it like it would an incoming virus and so responds to try to stop the infection. Our interest is to re-engineer adenovirus vectors to get around this problem.
Our work on this topic was funded from 2004-2010 by the Biotechnology and Biological Sciences Research Council (BBSRC) in the UK and from 2011-2014 by the Wellcome Trust. Here is the summary of that project, written for a lay audience:
Understanding and re-engineering adenovirus late gene expression: controlling the gatekeeper to productive infection
Successful gene therapy requires a vector to take DNA through the barrier of the cell surface. Several viruses have been modified as vectors, including adenovirus, which can be made incapable of growing and causing disease by removing selected parts of its genome. Another gene can then be inserted instead to create a delivery vector for the added gene. When the vector infects a cell, this gene becomes operative and has a beneficial effect. However, although adenovirus vectors can achieve this, the effect does not last very long because they are eliminated by immune responses that are generated to small amounts of viral proteins that are still made from genes that remain in the vector genome. We have been working on a new way to prevent the production of these proteins when a vector is used in therapy, without actually removing all the genes. This means that the vector can still be grown easily in the laboratory. The trick is to prevent production of just two late proteins, which we have discovered turn on the production of all the others. When these two proteins are provided, the vector grows well but otherwise, none of the late genes work.
2: PML and host responses to infection
You're perhaps familiar with the idea that you have an immune system that responds specifically to any viral or other infection that you suffer, helping your body to get rid of the infection and often preventing that same infection from affecting you again (this is the same principle that underlies vaccination). What is less well known is that this specific response takes several days to get going, and long before that, you are being protected by less specific systems which are known as innate and intrinsic immunity.
One aspect of innate immunity has been known for years - the production of interferon by a virus-infected cell. The release of this molecule spreads news of the infection to neighbouring cells and prepares them to resist infection, thus slowing the virus down. But the way in which a cell first becomes aware it is infected so as to trigger the production of interferon has only recently become clear, and as for intrinsic immunity - another whole range of molecules that slow down or block infection within a cell - this is still an unfolding story.
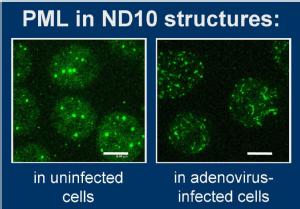
PML proteins are fascinating in lots of different ways. They got their name from being involved in one specific type of leukaemia - which of course makes them important straight away. But scientists working in many other fields of biology research have also found them to be involved in the phenomena they study. There are also lots of different forms of PML protein - not just one - which makes them difficult (but also exciting) to work with.
PML proteins are somehow part of the innate/intrinsic response to infection. Several research groups, working on very different viruses, have noticed that their virus does something to disrupt the normal behaviour of these proteins and adenovirus is one of these. It completely alters the way the PML is organised in the nucleus of the cell and this helps the virus to fight the cell's innate/intrinsic responses and so to reproduce successfully. We now know precisely which molecule from the virus does this, and which of the many forms of PML protein it acts upon. And we have recently worked out how PML contributes to innate immunity - it allows active gene expression from interferon genes and all the genes that interferon induces. So without PML, you get only a weak innate immune response. When adenovirus disrupts PML in the nucleus as you can see in the picture, the interaction it makes with the PML blocks its function in innate immunity - meaning that the virus can reproduce further and faster.
We've also recently discovered a connection between our work on PML and viruses, and how cells respond to stresses like being too warm or not having enough nutrients. Whenever a cell is stressed it produces a set of proteins that help it to cope with the stress: these are the HSPs (heat-shock proteins). It turns out that removing the PML protein needed for efficient innate immunity increases the production of an HSP (known as HSP70), even when the cell is growing normally. And that extra HSP70 is beneficial to adenovirus infection, so disrupting PML helps adenovirus to succeed in two ways - first by preventing a good innate immune response and second by causing production of HSP70.
