X-ray Photoelectron Spectroscopy (XPS)
In X-ray photoelectron spectroscopy (XPS) an incident X-ray photon interacts with a core electron in a solid, often resulting in its excitation from the atom and escaping into the surrounding material. As these core electrons are in unique electronic environments depending upon the atomic species the electron is bound to, XPS provides a detailed method for probing the composition of a material. In order to detect these electrons three things are necessary: an X-ray source, a sample and an electron detector. In order to maximise the free path of electrons and increase the probability of detection, this must be performed in an evacuated environment. As such, ultra-high vacuum is often required in order to perform XPS and many other photoelectron spectroscopies.
An X-ray photon of energy is incident on a surface and is absorbed by an atom with binding energy
. If the incident energy of the photon is larger than
then the electron is ejected from the atom with energy
. This is then detected in an analyser and the binding energy of that electron is determined by:
where is the work function of the electron analyser, not the species in question.
This means that any detected electrons provide a specific fingerprint of the atomic species in the near surface region. Slight variations in the binding energy known as chemical shifts, allow the chemical environment of atoms in the near surface region to be determined. However, due to varying x-ray absorption probabilities of different states the picture is slightly more complex. This process is shown schematically below:
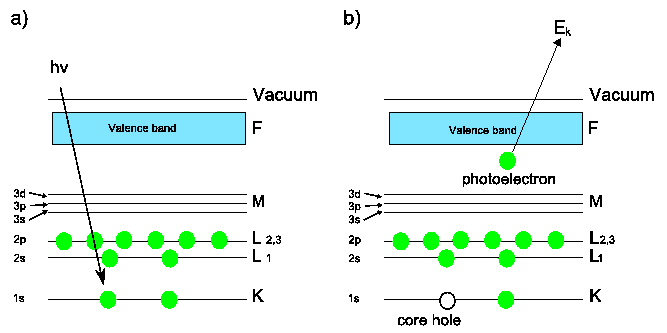
Schematic of the photoemission process in XPS. a) Incident photon energy is absorbed by a core level electron. b) If
then the electron is emitted and a core hole is left behind. This core hole may be filled by an electron from a higher state, causing emission of a secondary photon, or an Auger electron
A typical XPS spectrum is illustrated below. The spectrum shows a series of sharp peaks corresponding to core states of surface atoms. These peaks are accompanied by inelastic loss tail features, arising from interactions before the electrons emerge from the surface, thus reducing their energy. This feature in itself can be used to obtain chemical depth profiles of the sample [1].
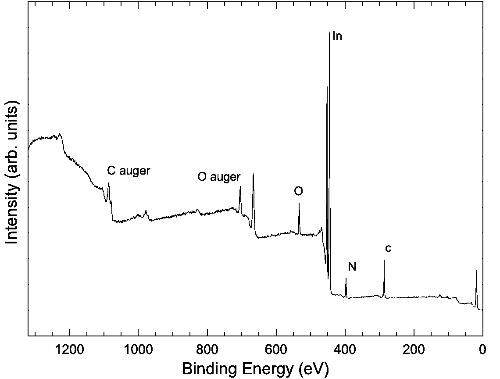
A typical XPS spectrum of an as-loaded InN sample, taken with a monochromated 1486.6 eV Alk-alpha source. Stepped structure of the background is due to inelastic processes.
Increasing the surface specificity of XPS can be achieved by using smaller angles of incidence. For quantitative XPS analysis broadening due to the energy width of electronic state, phonons and the instrumental resolution must be taken into account. Despite this XPS remains an excellent tool for qualitative and quantitative analysis.
Return to electronic characterisation
References
- L. Rosenberger, R. Baird, E. McCullen, G. Auner, and G. Shreve, Surf. Interface Anal. 40, 1254 (2008).
