News Library
Macpherson and Unwin and co-workers featured in "Chemistry World" and Chemical Communications
Macpherson and Unwin and co-workers Challenge the Current Consensus on the Electrochemical Properties of Nanotubes as featured in "Chemistry World" http://www.rsc.org/chemistryworld/2012/06/challenging-consensus-nanotube-electrochemistry and Chemical Communications http://pubs.rsc.org/en/content/articlelanding/2012/cc/c2cc32890a
and Chemical Communications http://pubs.rsc.org/en/content/articlelanding/2012/cc/c2cc32890a .
.
Unwin and O'Reilly win prestigious RSC awards
Pat Unwin and Rachel O’Reilly win 2012 Royal Society of Chemistry Awards in recognition of significant contributions to their research fields
Inverted membranes by ion soft landing
Costantini and co-workers report in Advanced Materials on the fabrication of inverted crystalline membranes of sodium dodecyl sulfate by ESI deposition.
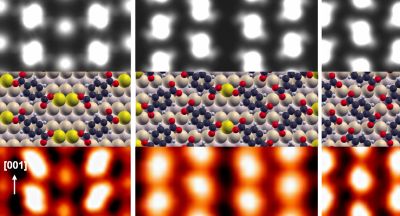
Structural studies on a Meningitis B vaccine
Results by MOAC and chemistry PhD student Angela Martino and her supervisor Alison Rodger were published this week on 4CMenB (a new meningococcal B vaccine).
At surfaces it's different
Costantini and collaborators have reported in a special themed issue of Chemical Communications about a novel chemical pathway observed only in the presence of a metal substrate. In solution chemistry, assuming no kinetic limitations, the thermodynamic product is formed independently of the absolute reactant concentration. However, inclusion of a metallic substrate introduces a further variable which ultimately defines the chemistry observed. In their recent work, terephthalic acid was deposited onto a Cu(110) substrate, where, at low surface coverages, 2-dimensional metal-organic structures form. However, with increasing coverage, the interaction between molecule and metal induces the formation of a denser, less energetically-favoured hydrogen-bonded network.
The article can be read here .
.
Prof. Fred McLafferty Officially Opens the new Ion Cyclotron Resonance Laboratory
Costantini and co-workers on the formation of chiral metal-organic structures at surfaces
 ). The supramolecular structures are organized on successive hierarchical levels with chiral properties developing only at the latest assembly step. The driving forces for the generation of these high-order chiral architectures are identified as competing coordination bonding within the metal-organic complexes and hydrogen bonding among them.
). The supramolecular structures are organized on successive hierarchical levels with chiral properties developing only at the latest assembly step. The driving forces for the generation of these high-order chiral architectures are identified as competing coordination bonding within the metal-organic complexes and hydrogen bonding among them.
"A barrel load of compounds" - Mark Barrow interviewed for Chemistry World
>http://www.rsc.org/images/Petroleomics_tcm18-180507.pdf

Giovanni Costantini and collaborators on reversing the shape transition of InAs/GaAs (001) quantum dots by in situ etching
 ) reports on the shape evolution of epitaxially grown InAs/GaAs(001) quantum dots after the controlled removal of material by in situ etching. An atomic force and scanning tunnelling microscopy investigation shows that a reversal of the shape transition that occurs during growth takes place. This reversibility impressively confirms that both the growth process and the etching process are dominated by thermodynamic factors. It is further found that the evolution of the quantum dots is not determined by direct etching but is caused by the removal of the wetting layer and the subsequent diffusion of In atoms from the quantum dots onto the bare GaAs.
) reports on the shape evolution of epitaxially grown InAs/GaAs(001) quantum dots after the controlled removal of material by in situ etching. An atomic force and scanning tunnelling microscopy investigation shows that a reversal of the shape transition that occurs during growth takes place. This reversibility impressively confirms that both the growth process and the etching process are dominated by thermodynamic factors. It is further found that the evolution of the quantum dots is not determined by direct etching but is caused by the removal of the wetting layer and the subsequent diffusion of In atoms from the quantum dots onto the bare GaAs.
Protein aging can be analysed by new mass spectrometry methods
Proteins age in many ways, but one of them involves deamidation of asparagine and glutamine to aspartic and glutamic acids respectively. When this occurs, two isomers of the acidic species are generated. Glutamine deamidation of proteins is specifically studied by the O'Connor group in a new report in Analytical chemistry, with the result that the two isomers can be readily differentiated with a new fragmentation technique called Electron Capture Dissociation.
http://dx.doi.org/10.1021/ac9028467
Mass spectrometry unravels the aquatic environmental impact of the Canadian Athabasca oil sands

Pat Unwin's and Julie Macpherson's electrochemistry group make the cover of Chemical Communications
Ioana Dumitrescu, Patrick R. Unwin and Julie V. Macpherson make the cover of Chem.Commun with their feature article on Electrochemistry at carbon nanotubes (CNTs): It is a large and growing field, but one in which there is still uncertainty about the fundamental activity of CNTs as electrode materials. On the one hand, there are many reports which focus on the favourable electrochemical properties of CNT electrodes, such as enhanced detection sensitivity, electrocatalytic effects and reduced fouling. On the other hand, other studies suggest that CNTs may be no more electroactive than graphitic powder. Furthermore, it has been proposed that the catalytic nanoparticles from which CNTs are formed may dominate the electrochemical characteristics in some instances. A considerable body of the literature presumes that the CNT sidewall is inert and that edge-plane-graphite-like open ends and defect sites are responsible for the electron transfer activity observed. In contrast, studies of well characterised single-walled nanotube (SWNT) electrodes, either as individual tubes or as two-dimensional networks, suggest sidewall activity. This review highlights how the various discrepancies in CNT electrochemistry may have arisen, by taking a historical view of the field and identifying crucial issues that still need to be solved. When assessing the behaviour of CNT electrodes, it is vitally important that careful consideration is given to the type of CNT used (SWNT or multi-walled), the quality of the material (presence of impurities), the effect of chemical processing steps in the fabrication of electrodes and the experimental arrangements adopted. Understanding these key features is an essential requirement to develop a fundamental understanding of CNT electrochemistry, to allow a wide range of electroanalytical applications, and to move the field forward rationally. As part of this process, high resolution electrochemical and electrical imaging techniques are expected to play a significant role in the future, as well as theoretical developments which examine the fundamentals of electron transfer at different types of CNTs and their characteristic surface sites. http://dx.doi.org/10.1039/b909734a
