Metallothioneins: an overview
Metallothioneins are ubiquitous in all kingdoms. Mammalian MTs have been discovered as Cd-binding proteins in horse liver by Margoshes and Vallee in 1957, but in the meantime, it has become clear that these intriguing proteins play much more fundamental roles than dealing with cadmium toxicity. They are thought to play an integral part in the homeostasis of essential metal ions, and thus have an impact on a range of physiological processes. Whilst a large body of knowledge has been accumulated for vertebrate MT both in vitro and in vivo, we know much less about metallothioneins from other species.
The schematic below illustrates the distribution of metallothioneins across a part of the tree of life.
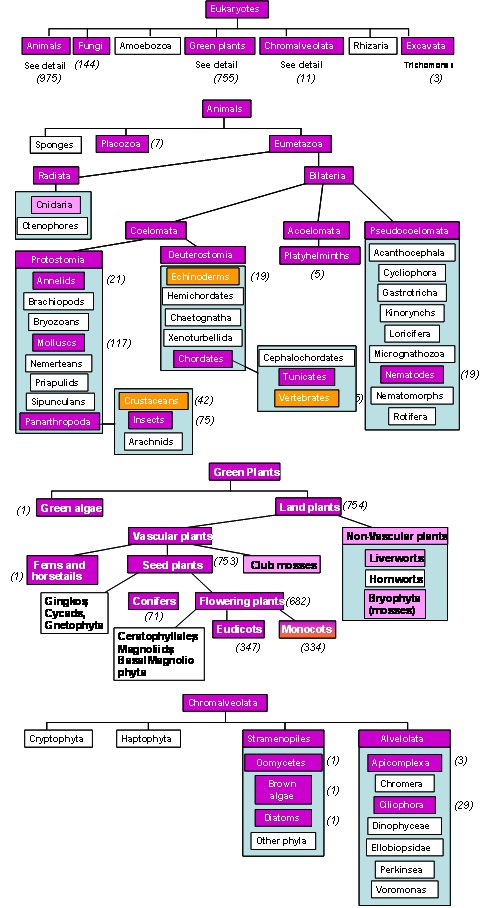 Boxes in magenta indicate evidence for metallothioneins (or metallothionein-like proteins) at nucleotide level, and pink boxes refer to EST (expressed sequence tag) data. The numbers in brackets indicate the number of sequences deposited in the "Nucleotide" database at NCBI (as of December 2009). Bacterial MTs are not included in the overview, see Bacterial Metallothioneins for details.
Boxes in magenta indicate evidence for metallothioneins (or metallothionein-like proteins) at nucleotide level, and pink boxes refer to EST (expressed sequence tag) data. The numbers in brackets indicate the number of sequences deposited in the "Nucleotide" database at NCBI (as of December 2009). Bacterial MTs are not included in the overview, see Bacterial Metallothioneins for details.
Orange boxes indicate the availability of 3D structural data. Only six different folds for metallothioneins have been determined so far (beside vertebrate, echinoderm, and crustacean MTs, these are pdb entries for baker's yeast Cup1, bacterial SmtA, and one domain of the wheat EC protein).
Metallothioneins from different phyla appear to be evolutionarily unrelated, and hence, it is not possible to predict structure and properties of the majority of MTs listed here. The scarcity of orange boxes clearly illustrates the wealth of knowledge still to be uncovered. We maintain that a comprehensive understanding of the function of a protein benefits enormously from the knowledge of its 3D structure - even though we also acknowledge that this is just a starting point to understand the reactivity of a molecule.
