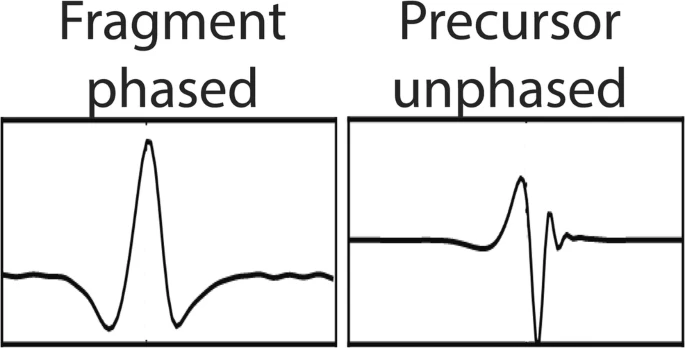
Christopher A. Wootton
Currently Head of MRMS R&D at Bruker Daltonics, Bremen, Germany
___________________________________________________________________________
Also Honorary Research Fellow Warwick FT-ICR Mass Spectrometry group

Department of Chemistry, The University of Warwick
Millburn Hill Road, Coventry CV4 7AL, UK
ORCID: orcid.org/0000-0002-3647-2611
LinkedIN: https://www.linkedin.com/in/christopher-wootton-7b986b6a/
Legacy Research Projects:
Developing new FT-ICR MS instrumentation and methods for the study of complex systems
To achieve reliable and routine analysis of biomolecules and complex samples high performance MS instrumentation is required. Current instrumentation has allowed the study of peptides, proteins, DNA, RNA and many species in between, in order to further this study, instrumentation must also develop to fulfill the requirements of such analysis.
We are currently investigating new instrumentation-based methods for ionising species of interest, manipulating their motion through mass spectrometers, improved detection aparatus, and implimentation of novel MS/MS techniques for comprehensive biomolecule characterisation.
Current development is based around a custom 12T FT-ICR mass spectrometer modified in house and will be suplimented with additional ionisation capabilities, ion optics, ICR cell upgrades, and new MS/MS capabilities. All controlled using a centralised and fully customisable NI LabView program developed in-house for routine and more complex (two-dimensional) MS experiments. Any questions? Please feel free to contact and discuss via email.
Development of 2-Dimensional MS processing software:
2DMS creates incredibly rich spectra of complex samples in a truely data-independent fashion. With big data comes big software requirements and advances in 2DMS data interpretation and analysis come in the form of our in-house developed software for this; True-2D (T2D). Please see our sub page on T2D here for more information.
Detailed characterisation of novel metallodrug interactions with biomolecules by high resolution Tandem-FT-ICR MS
Using ultra high resolution mass spectrometry to study the mechanism of action of organometallic and photoactivatable metal-based anticancer complexes, especially with respect to biomolecules such as peptides, proteins, and DNA.
A collaborative project between the O'Connor and Sadler Groups at the University of Warwick.
Background and research focus:
Metallodrugs can offer improved potency and selectivity over non-metal counterparts for a wide variety of diseases such as Malaria, Tuberculosis, and of course Cancer. Metallodrugs have so far offered the most effective anti-cancer compounds available and specifically platinum based chemotherapeutics are now used in over 50% of anti-cancer treatment regimes.
However it has been shown that over 90% of some traditional platinum based anti-cancer metallodrugs become protein bound within just one day of injection. The peptides and proteins within the blood and cells contain many residues that are excellent ligands for these complexes and binding to them has been shown to be extensive.
Unfortunately this can cause large conformational changes to tertiary structure, blocking of active sites and overall great decreases in function. This extensive biomolecule damage has been attributed to some of the side effects experienced by patients undergoing treatment. New metallodrugs currently synthesised have focused on reducing these side effects via better selectivity and new mechanisms of action against their target disease.
We are interested in studying how and why these new metallodrugs behave towards biomolecules they are likely to encounter in the body.
More specifically we are investigating a new range of photoactivatable metallodrugs synthesised by the Sadler group here at Warwick and studying their unique behaviour when activated in the presence of biomolecules. These new Platinum(IV) based compounds have shown to behave in very different ways to previous platinum based compounds, such as Cisplatin, and have shown not only different potencies, but also very different binding preferences and identities once activated.
The concept is centred around injecting an inert prodrug into the body, usually a Platinum (IV) low spin d6 species, which can then be activated by certain wavelengths of light (tailored by the Metal centre and the ligands attached), this causes photo-induced cleavage of metal-ligand bonds and the creation of reactive Platinum(II) species which can then attack nearby groups and enact a potent cytotoxic effect upon cancer cells.
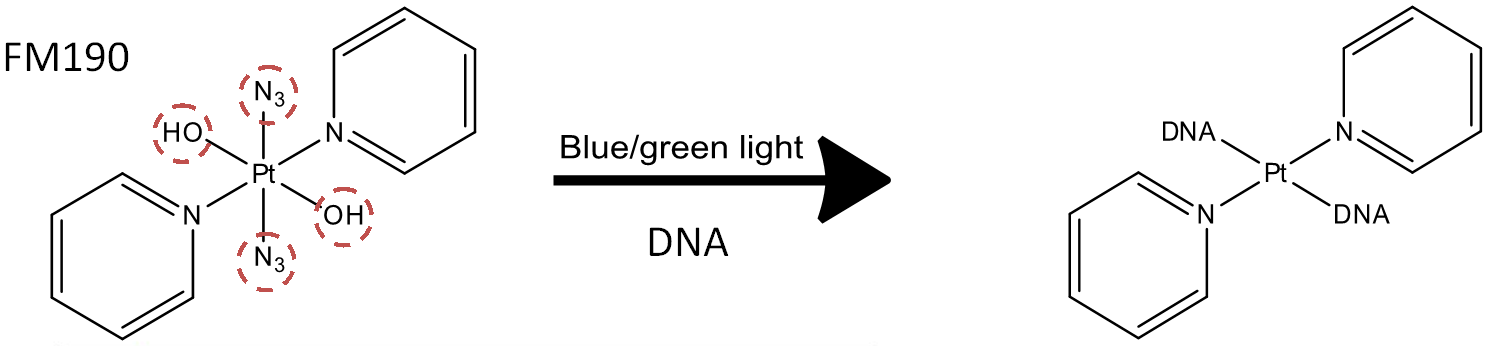
(reported reaction of FM190 with DNA under irradiation with blue light by NMR, the reaction mixture analysed by High-resolution FT-ICR Mass Spectrometry showed a larger number and variety of reaction products)
This Photoactivation process allows a great deal of temporal and spatial control over these potent compounds and will allow for localised activation of the metallodrug near/within tumours and will limit/minimise distribution of active species to other areas of the body which can cause some of the side effects of traditional chemotherapy.
These complexes have been shown to be very effective against a wide array of cancer cell lines, but more interestingly they are also effective against Cisplatin resistance strains of cancer. This interesting and very useful property makes these compounds highly desirable as a new generation of potent, but controllable chemotherapeutic agents.
Here we use ultra-high resolution mass spectrometry (FT-ICR MS) to study the interactions of these complexes with novel biomolecules and investigate the unique and interesting modifications these drugs carry out upon irradiation and try to discover and explain the behaviour and possible mechanisms of action of the new metallodrugs which have been shown to be so effective in combatting cancer.
We are particularly interested in how these metal based modifications behave during Electron Capture Dissociation (ECD) and Electron Detachment Dissociation (EDD) tandem mass spectrometry (MS/MS) and investigate how both the nature of the metal, and of the ligands bound, can affect the tuning of the ECD/EDD parameters and the fragments observed.
We are also investigating a range of other metal based therapeutics to study their binding locations, identities and ECD/EDD behaviour. We are currently investigating Platinum, Iridium, Ruthenium, Osmium, and Rhodium complexes but will be looking into many more in the future.
Please feel free to email me at C dot Wootton dot 1 at warwick dot ac dot uk if you have any questions about Top down peptide/protein/DNA fragmentations, Metal-biomolecule MS/MS, photoactivatable metallodrugs, or our work in general. The work above was based on our Chemical Science paper, 2018, see publication list below for more info.
Publications:
- Christopher A. Wootton, Carlos Sanchez-Cano, Hong-Ke Liu, Mark P. Barrow, Peter J. Sadler, and Peter B. O’Connor. Binding of an organo–osmium(II) anticancer complex to guanine and cytosine on DNA revealed by electron-based dissociations in high resolution Top–Down FT-ICR mass spectrometry, Dalton Transactions, 2015, 44, 3624 - 3632, DOI: 10.1039/c4dt03819cLink opens in a new window

- Maria A. van Agthoven, Mark P. Barrow, Lionel Chiron, Marie-Aude Coutouly, David Kilgour, Christopher A. Wootton, Juan Wei, Andrew Soulby, Marc-André Delsuc, Christian Rolando, Peter B. O’Connor. Differentiating Fragmentation Pathways of Cholesterol by Two-Dimensional Fourier Transform Ion Cyclotron Resonance Mass Spectrometry, JASMS, 2015, 1044-0305, DOI:10.1007/s13361-015-1226-7
- Maria A. van Agthoven, Christopher A. Wootton, Lionel Chiron, Marie-Aude Coutouly, Andrew J
Soulby, Juan Wei, Mark P. Barrow, Marc-André Delsuc, Christian Rolando, and Peter B O'Connor. Two-Dimensional Mass Spectrometry for Proteomics, a Comparative Study with Cytochrome C, Analytical Chemistry, 2016, DOI:10.1021/acs.analchem.5b04878
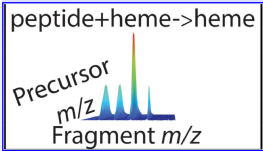
- Floris, F.; van Aghtoven, M.A.; Chiron L.; Wootton C. A.; Lam P.Y.; Delsuc M.-A.; Barrow P.M.; O'Connor, P.B. - Two-dimensional Fourier Transform Ion Cyclotron Resonance Mass Spectrometry of Calmodulin: a top-down and bottom-up approach. J. Am. Soc. Mass Spectrom. (2016). DOI:10.1007/s13361-016-1431-z
- Christopher A. Wootton, Yuko P. Y. Lam, Matthew Willetts, Maria A. van Agthoven, Mark P. Barrow, Peter J. Sadler* and Peter B. O’Connor - Automatic Assignment of Metal-containing Peptides in Proteomic LC-MS and MS/MS Datasets, Analyst (2017), DOI: 10.1039/c7an00075h.
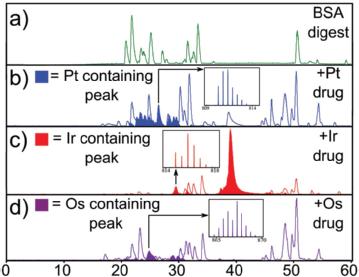
- Christopher A. Wootton, Carlos Sanchez-Cano, Andrea F. Lopez-Clavijo, Evyenia Shaili, Mark P. Barrow, Peter J. Sadler, and Peter B. O’Connor - Sequence-Dependent Attack on Peptides by Photoactivated Platinum Anticancer Complexes, Chemical science (2018), DOI: 10.1039/c7sc05135b.
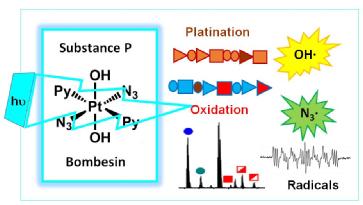
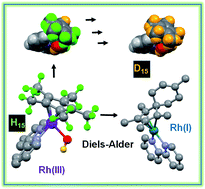
- Samya Banerjee, Joan J. Soldevila-Barreda, Juliusz A. Wolny, Christopher A. Wootton, Abraha Habtemariam, Isolda Romero-Canel´on, Feng Chen, Guy J. Clarkson, Ivan Prokes, Lijiang Song, Peter B. O'Connor, Volker Schunemann, and Peter J. Sadler* - New activation mechanism for half-sandwich organometallic anticancer complexes, Chemical Science (2018), DOI: 10.1039/c7sc05058e.
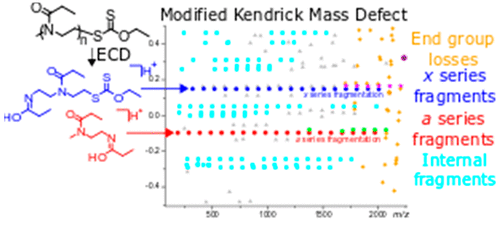
- Tomos E. Morgan, Sean H. Ellacott, Christopher A. Wootton, Mark P. Barrow, Anthony W. T. Bristow, Sebastien Perrier, Peter B. O'Connor, Coupling Electron Capture Dissociation and the modified Kendrick mass defect for sequencing of a poly(2-ethyl-2-oxazoline) polymer., Analytical Chemistry 2018, 90, 19, 11710-11715. DOI: 10.1021/acs.analchem.8b03591
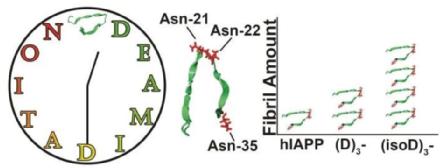
- Pui Yiu Lam, Christopher A. Wootton, Ian Hands-Portman, Juan Wei, Cookson K C Chiu, Isolda Romero-Canelon, Mark Barrow, Frederik Lermyte and Peter O' Connor. Does Deamidation of Islet Amyloid Polypeptide Accelerate Amyloid Fibril Formation? Chemical Communications, 2018, DOI: 10.1039/C8CC06675B
- Maria A. van Agthoven, Alice M. Lynch, Tomos E. Morgan, Christopher A. Wootton, Yuko P. Y. Lam, Lionel Chiron, Mark P. Barrow, Marc-Andre Delsuc, Peter B. O'Connor,Can Two-Dimensional IR-ECD Mass Spectrometry Improve Peptide de Novo Sequencing?, Anal. Chem., 2018, 90, 3496. DOI:10.1021/acs.analchem.7b05324,
- Christopher A. Wootton, Adam Millett, Andrea Lopez, Cookson K C Chiu, Mark Barrow, Guy J Clarkson, Peter J Sadler and Peter O' Connor. Structural analysis of peptides modified with organo-iridium complexes, opportunities from multi-mode fragmentation. Analyst 2019, DOI: 10.1039/C8AN02094A
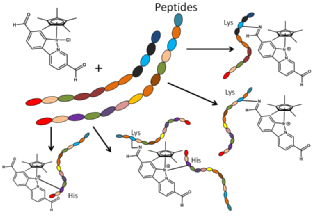
- Frederik Lermyte, James Everett, Yuko PY Lam, Christopher A. Wootton, Jake Brooks, Mark P Barrow, Neil D Telling, Peter J Sadler, Peter B O’Connor, Joanna F Collingwood. Metal Ion Binding to the Amyloid β Monomer Studied by Native Top-Down FTICR Mass Spectrometry, JASMS 2019, DOI:10.1007/s13361-019-02283-7Link opens in a new window
- Maria A. van Agthoven, David P. A. Kilgour, Alice M. Lynch, Mark P. Barrow, Tomos E. Morgan, Christopher A. Wootton, Lionel Chiron, Marc-Andre Delsuc, Peter B. O'Connor, Journal of The American Society for Mass Spectrometry, 2019, 30, 12, 2594-2607.
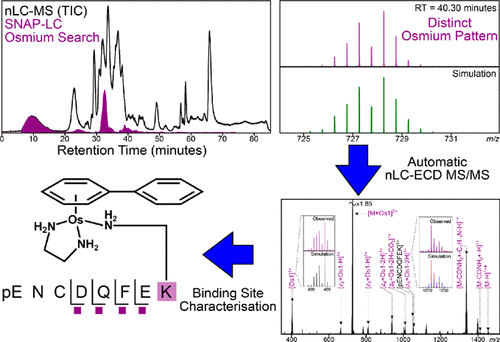
- Cookson K. C. Chiu, Yuko P. Y. Lam, Christopher A. Wootton, Mark P. Barrow, Peter J. Sadler, and Peter B. O’Connor. Metallocomplex–Peptide Interactions Studied by Ultrahigh Resolution Mass Spectrometry. J. Am. Soc. Mass Spectrom. 2020, 31, 3, 594–601.
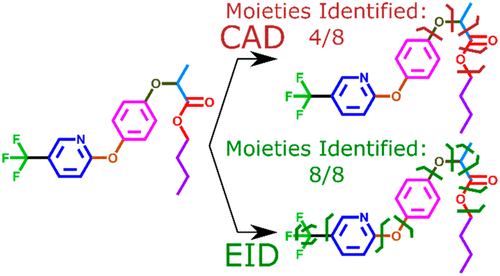
- Bryan P. Marzullo, Tomos E. Morgan, Christopher A. Wootton, Meng Li, Simon J. Perry, Mansoor Saeed, Mark P. Barrow, Peter B. O'Connor, Comparison of Fragmentation Techniques for the structural characterisation of singly charged agrochemicals, Anal. Chem., 2020, 92, 4, 3143-3151.
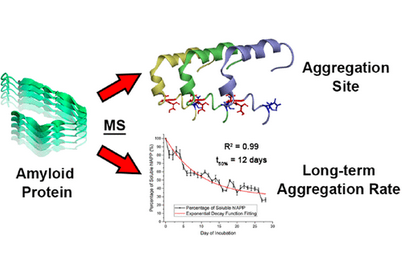
- Yuko P. Y. Lam, Christopher A. Wootton, Ian Hands-Portman, Juan Wei, Cookson K. C. Chiu, Isolda Romero-Canelon, Mark Barrow, Frederik Lermyte, and Peter O'Connor, Determination of the Aggregate Binding Site of Amyloid Protofibrils Using Electron Capture Dissociation Tandem Mass Spectrometry. Journal of the American Society for Mass Spectrometry, 2020, DOI: 10.1021/jasms.9b00053
- Johanna Paris, Tomos E. Morgan, Christopher A. Wootton, Mark P. Barrow, John O'Hara, Peter B. O'Connor, Facile determination of phosphorylation sites in peptides using 2DMS, Anal. Chem. 2020, 92, 10, 6817–6 https://doi.org/10.1021/acs.analchem.0c00884
-
Yuko P. Y. Lam, Cookson K. C. Chiu, Christopher A. Wootton, Ian Hands-Portman, Meng Li, Mark P. Barrow, and Peter B. O'Connor, Does deamidation affect inhibitory mechanisms towards amyloid protein aggregation? Chem Comm. 2020 56, 9787-9790 DOI: https://doi.org/10.1039/D0CC03548C

-
Bryan P. Marzullo, Tomos E. Morgan, Christopher A. Wootton, Simon J. Perry, Mansoor Saeed, Mark P. Barrow, and Peter B. O’Connor. Advantages of Two-Dimensional Electron-Induced Dissociation and Infrared Multiphoton Dissociation Mass Spectrometry for the Analysis of Agrochemicals. Anal. Chem. 2020, 92, 17, 11687–11695, DOI:https://doi.org/10.1021/acs.analchem.0c01585
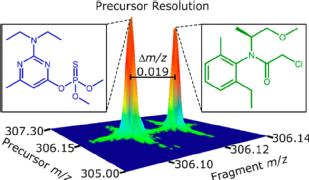
-
Two-Dimensional Mass Spectrometry Analysis of IgG1 Antibodies, Johanna Paris, Tomos E. Morgan, Bryan P. Marzullo, Christopher A. Wootton, Mark P. Barrow, John O’Hara, and Peter B. O’Connor, J. Am. Soc. Mass Spectrom. 2021, 32, 7, 1716–1724. DOI: https://doi.org/10.1021/jasms.1c00096
- Bryan P. Marzullo, Tomos E. Morgan, Alina Theisen, Anisha Haris, Christopher A. Wootton, Simon J. Perry, Mansoor Saeed, Mark P. Barrow, and Peter B. O’Connor, Combining Ultraviolet Photodissociation and Two-Dimensional Mass Spectrometry: A Contemporary Approach for Characterizing Singly Charged Agrochemicals. Anal. Chem. 2021, 93, 27, 9462–9470 https://doi.org/10.1021/acs.analchem.1c01185

-
Tomos E. Morgan, Christopher A. Wootton, Bryan Marzullo, Johanna Paris, Andrew Kerr, Sean H. Ellacott, Maria A. van Agthoven, Mark P. Barrow, Anthony W. T. Bristow, Sebastien Perrier, and Peter B. O’Connor, Characterization Across a Dispersity: Polymer Mass Spectrometry in the Second Dimension. J. Am. Soc. Mass Spectrom. 2021, 32, 8, 2153–2161 https://doi.org/10.1021/jasms.1c00106

-
Fanny C. Liu, Mark E. Ridgeway, J. S. Raaj Vellore Winfred, Nicolas C. Polfer, Jusung Lee, Alina Theisen, Christopher A. Wootton, Melvin A. Park, Christian Bleiholder, Tandem-trapped ion mobility spectrometry/mass spectrometry coupled with ultraviolet photodissociation. RCMS 2021, 35, 22 https://doi.org/10.1002/rcm.9192
- Diana Catalina Palacio Lozano, Hugh E. Jones, Remy Gavard, Mary J. Thomas, Claudia X. Ramírez, Christopher A. Wootton, José Aristóbulo Sarmiento Chaparro, Peter B. O’Connor, Simon E. F. Spencer, David Rossell, Enrique Mejia-Ospino, Matthias Witt, and Mark P. Barrow, Revealing the Reactivity of Individual Chemical Entities in Complex Mixtures: the Chemistry Behind Bio-Oil Upgrading, Anal. Chem. 2022, https://doi.org/10.1021/acs.analchem.2c00261

-
Anisha Haris, Yuko P. Y. Lam, Christopher A. Wootton, Alina Theisen, Bryan P. Marzullo, Pascal Schorr, Dietrich A. Volmer, and Peter B. O’Connor Differentiation of Dihydroxylated Vitamin D3 Isomers Using Tandem Mass Spectrometry J. Am. Soc. Mass Spectrom. 2022, 33, 6, 1022–1030. DOI: https://doi.org/10.1021/jasms.2c00085
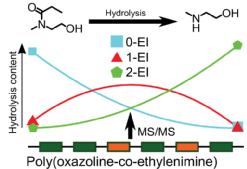
- Tomos E.Morgan, Thomas G.Floyd, Bryan P.Marzullo, Christopher A.Wootton, Mark P.Barrow, Anthony W. T.Bristow, SébastienPerrier, Peter B.O'Connor. Stochasticity of poly(2-oxazoline) oligomer hydrolysis determined by tandem mass spectrometry. Polym. Chem. 2022, DOI: 10.1039/d2py00437b.
-
Jingsha Xu, Meng Li, Bryan Marzullo, Christopher A. Wootton, Mark P. Barrow, and Peter B. O’Connor. Fine Structure in Isotopic Peak Distributions Measured Using Fourier Transform Ion Cyclotron Resonance Mass Spectrometry: A Comparison between an Infinity ICR Cell and a Dynamically Harmonized ICR Cell. J. Am. Soc. Mass Spectrom. 2022, 33, 8, 1499–1509. DOI: https://doi.org/10.1021/jasms.2c00093
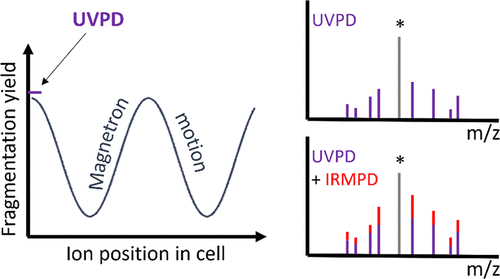
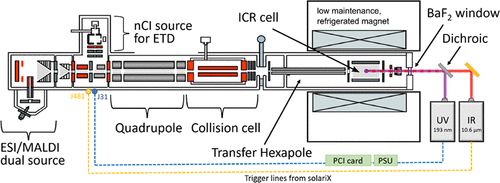
- Alina Theisen, Christopher A. Wootton, Anisha Haris, Tomos E. Morgan, Yuko P. Y. Lam, Mark P. Barrow, and Peter B. O’Connor, Enhancing Biomolecule Analysis and 2DMS Experiments by Implementation of (Activated Ion) 193 nm UVPD on a FT-ICR Mass Spectrometer, Analytical Chemistry, 2022 94, 45, 15631–15638, https://doi.org/10.1021/acs.analchem.2c02354
__
- Depraz Depland, Agathe; Stroganova, Iuliia; Wootton, Christopher; Rijs, Anouk. Developments in Trapped Ion Mobility Mass spectrometry to probe the early stages of peptide aggregation, JASMS, 2023, DOI :10.1021/jasms.2c00253Link opens in a new window
- Fanny C Liu, Mark E Ridgeway, Christopher A Wootton, Alina Theisen, Erin M Panczyk, Florian Meier, Melvin A Park, Christian Bleiholder. Top-Down Protein Analysis by Tandem-Trapped Ion Mobility Spectrometry/Mass Spectrometry (Tandem-TIMS/MS) Coupled with Ultraviolet Photodissociation (UVPD) and Parallel Accumulation/Serial Fragmentation (PASEF) MS/MS Analysis. JASMS 2023,
- Christopher A. Wootton*, Julien Maillard, Alina Theisen, Gregory F Brabeck, Carlos L Schat, Christopher P Rüger, Carlos Afonso*, Pierre Giusti.A Gated TIMS FTICR MS instrument to decipher isomeric content of complex organic mixtures. Analytical chemistry 2024, 96, 11343-11352, https://doi.org/10.1021/acs.analchem.4c01370

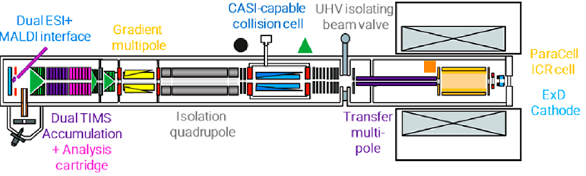
-
Jamie P. Butalewicz, Edwin E. Escobar, Christopher A. Wootton, Alina Theisen, Melvin A. Park, Erin H. Seeley, Jennifer S. Brodbelt, Conformational Characterization of Peptides and Proteins by 193 nm Ultraviolet Photodissociation in the Collision Cell of a Trapped Ion Mobility Spectrometry-Time-of-Flight Mass Spectrometer. Anal. Chem. 2024, 96, 41, 16154–16161. https://doi.org/10.1021/acs.analchem.4c02686Link opens in a new window
- Kas J. Houthuijs, Lara van Tetering, Jelle L. Schuurman, Christopher A. Wootton, Christoph R. Gebhardt, Mark E. Ridgeway , Giel Berden, Jonathan Martens, Jos Oomens. A trapped ion mobility enabled Fourier transform ion cyclotron resonance mass spectrometer for infrared ion spectroscopy at FELIX. IJMS 2024 DOI:https://doi.org/10.1016/j.ijms.2024.117323 Link opens in a new window

- Callan Littlejohn; Meng Li; Christopher A. Wootton; Mark P. Barrow; Peter B. O’Connor. Hyphenation of Trapped Ion Mobility to Two-Dimensional Mass Spectrometry for Protein Analysis in Complex Biomixtures. https://doi.org/10.1021/jasms.5c00292
- Tomos E. Morgan, Alina Theisen, Sean Ellacott, Anisha Haris, Christopher A. Wootton, Julia Y. Rho, Mark P. Barrow, Anthony W. T. Bristow, Sébastien Perrier, Peter B. O’Connor; Cyclic Peptide–Polymer Conjugate Characterization Using 193 nm Ultraviolet Photodissociation Tandem Mass Spectrometry, Analytical Chemistry, 2026, https://pubs.acs.org/doi/10.1021/acs.analchem.5c03375Link opens in a new window

- Fanny C. Liu, Jusung Lee, Kaira A. Mayberry, Mark E. Ridgeway, Christopher A. Wootton, Alina Theisen, Erin M. Panczyk, Benjamin J. Jones, Lea Nienhaus, Melvin A. Park, Christian Bleiholder* Direct observation of metastable fragment ions in ultraviolet photodissociation of Ubiquitin. JASMS, 2026, https://doi.org/10.1021/jasms.5c00432.
- Highlighted by the ACS Editor's choice program

Application notes/patents etc.:
- Patent for novel ionisation source, O'Connor P.B., Wootton C.A., Hussein H., Chiu C., Wong M.
- Application note "Monitoring the Electrochemical Reduction of Disulphide Bonds in Proteins for Enhanced MS and MS/MS Capabilities". Christopher A. Wootton and Cookson Chiu, Agnieszka Kraj, Thomas Lambert, Simon Lambert, and Peter B. O’Connor, in collaboration with ARC Sciences (UK) and Antec (Netherlands), please find online here or here; http://arcsciences.com/applications/protein_and_peptide.aspx
Poster Presentations:
- ASMS 2013 - Minneapolis, US
- EMPW 2013 - Leicester, UK
- BMSS 2013 - Eastbourne, UK
- Uppcon 2014 - Obergurgl, Austria - Awarded Poster Prize by The Analyst journal
- EFTMS 2014 - Paris, France
- Uppcon 2015 - California, US - Poster + 5 minute "lightning talk"
- IBBI 2016 - Oxford, UK - Awarded Poster Prize by the IBBI conference organisers
- Gratefully acknowledging travel funding from BMSS and IBBI conference organisers
- BMSS 2017 - Manchester, UK
- EMPW 2017 - Nottingham Trent, UK - Awarded best poster prize by the EMPW conference organisers
- ASMS 2018 - San Diego, US
- Uppcon 2018 - Leeds, UK
- IMSC 2018 - Florence, Italy
- EMPW 2018 - Lincoln, UK
- ASMS 2019 - Atlanta, US
- ASMS 2022 - Minneapolis, US
- BMSS 2024 - Warwick, UK
- CTDP 2025 - Hamburg, Germany (consortium for top down proteomics)
- ASMS 2026 - San Diego, US
Oral Presentations:
- EMPW 2014 - Leicester, UK - Awarded Oral presentation prize for best Early Career Researcher talk
- ASMS 2015 - St. Louis, US - Presented in the Top-down Protein Analysis session
- BMSS 2015 - Birmingham, UK - Presented in the Top-down Protein session
- EMPW 2015 - Univeristy of Warwick, UK
- Tandem Mass Spectrometry Workshop, Lake Louise, Canada - December 2015
- Awarded $2000 grant to present talk by Lake Louise conference organisers, which is gratefully acknowledged
- EFTMS 2016 - Matera, Italy - Presented in Nanodroplets, Ion sources, and clusters session
- Proteomics Methods Forum (PMF) 2016 - Warwick, UK - Presented in the Diverse Proteomics session
- BMSS 2016 - Eastbourne, UK - Presented in Fundamentals and Instrument Developments session
- MS&PG 2016 - Mass Spectrometry & Proteomic Congress 2016, London, UK - Presented a vendor-solutions talk after the keynote session
- Gratefully acknowledging the invitation and travel funding from ARC Sciences (UK) to discuss the ROXY ElectroChemical (EC) reduction of protein
disulphide bonds for enhanced MS and MS/MS capabilities in collaboration with Antec (The Netherlands).
- AI-SIG 2016 - Ambient Ionisation SIG meeting 2016 in Keele, UK
- ASMS 2017 - Indianapolis, US - Presented in the ionisation instrumentation session
- Kanpur workshop - Peptides, proteins, and metals in disease and therapy - Kanpur, India - Presented in the Advanced analytical methods for studies of protein modification and aggregation session
- Gratefully acknowledging funding from the British council Newton-Bhabha fund to travel to India and present our work
- EFTMS 2018 – Freising, Germany – Presented in the two-dimensional FTMS session
- Uppcon 2018 - Leeds, UK - presented on unusual effects of metal complexes in ECD MS/MS of biomolecules
- BMSS 2019 - Manchester, UK - presented on advances in 2DMS and 2DMS on a Q-ToF system
- EU FT-ICR MS school 2021, Lisbon - Invited Presentation on Paracell optimisation and FT-ICR harmonics for MRMS operation and on Top down protein analysis using MRMS
- BMSS 2021 - Sheffield, UK - Presented keynote talk in the Instrumentation development session; Multi-million resolving power MS and MS/MS on FT-ICR MS
- IBBI 2022 - Obergurgl, Austria - Presented vendor sponsored session Top down analysis using MRMS
- ASMS 2023 - Houston, US - Presented a new TIMS-FT-ICR MS instrument in the High resolution instrumentation session
- DGMS 2023 - Rostock, Germany - Presented developments of the new TIMS-FT-ICR MS instrument for complex mixture analysis
- EFTMS 2024 - Prague - Presented on TIMS FT-ICR MS analysis of complex mixtures and MALDI imaging with new instrumentation
- UppCon 2024 - Darmstadt, Germany, Presented on conformer selective ExD MS/MS and CIU of proteins using a novel gated TIMS FT-ICR MS system
- BMSS 2024 - Warwick, UK - Presented on recent developments of the new TIMS-FT-ICR MS instrument for complex mixture and biomolecule analysis
- NA FTMS 2025 - Lake Tahoe, US - Presented on extending the performance of prototype TIMS-FT-ICR MS instruments and MALDI imaging
- ASMS 2025 - Baltimore, US - Presented on Enhancing the dynamic range of TIMS-FT-ICR MS instruments to extend the frontiers of complex sample analysis. in the High resolution instrumentation session. Awarded the ASMS Ron Hites award for outstanding JASMS publication with Prof. C. Bleiholder.

 Leading the research group in FT-ICR MS (aka MRMS) fundamentals, instrument development, and applications thereof to a wide variety of analytical challenges.
Leading the research group in FT-ICR MS (aka MRMS) fundamentals, instrument development, and applications thereof to a wide variety of analytical challenges.